 Back
BackOrganic Chemistry Exam #2 – Step-by-Step Study Guidance
Study Guide - Smart Notes

Q1. Provide the correct IUPAC name for each compound shown.
Background
Topic: IUPAC Nomenclature of Alkanes and Cycloalkanes
This question tests your ability to systematically name organic molecules using IUPAC rules, focusing on identifying the parent chain, numbering, and naming substituents.
Key Terms and Concepts:
Parent Chain: The longest continuous chain of carbon atoms.
Substituents: Groups attached to the parent chain.
Numbering: Assign numbers to give substituents the lowest possible locants.
Prefixes: Use di-, tri-, etc., for multiple identical substituents.
Step-by-Step Guidance
Identify the longest continuous carbon chain in the structure (this will be your parent name).
Number the chain from the end nearest a substituent to give the lowest possible numbers to the substituents.
Identify and name each substituent (e.g., methyl, ethyl, tert-butyl, etc.).
Assign a number to each substituent based on its position on the parent chain.
Combine the substituent names and numbers in alphabetical order, followed by the parent name.
Try solving on your own before revealing the answer!
Q2. Draw the correct structure for 1-tert-butyl-4-methylcycloheptane.
Background
Topic: Drawing Structures from IUPAC Names
This question tests your ability to interpret IUPAC names and translate them into correct line-angle (skeletal) structures.
Key Terms and Concepts:
Cycloheptane: A seven-membered carbon ring.
tert-Butyl group: A tertiary butyl group (–C(CH3)3).
Methyl group: –CH3 substituent.
Step-by-Step Guidance
Draw a seven-membered ring to represent cycloheptane.
Number the ring so that position 1 is where you will attach the tert-butyl group.
Attach a tert-butyl group at carbon 1.
Attach a methyl group at carbon 4.
Try solving on your own before revealing the answer!
Q3. Label the following Newman projections as anti, eclipsed, or gauche.
Background
Topic: Conformational Analysis – Newman Projections
This question tests your understanding of the spatial arrangement of atoms/groups around a single bond and how to classify conformations as anti, eclipsed, or gauche.
Key Terms and Concepts:
Newman Projection: A way to visualize the conformation of a molecule by looking straight down a bond axis.
Anti: Largest groups are 180° apart.
Gauche: Largest groups are 60° apart.
Eclipsed: Groups on the front and back carbons overlap when viewed down the bond.
Step-by-Step Guidance
Identify the two largest groups on each Newman projection.
Determine the dihedral angle between these groups (0°, 60°, or 180°).
Classify each conformation as anti (180°), gauche (60°), or eclipsed (0°).
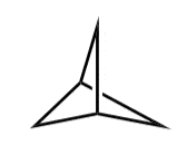
Try solving on your own before revealing the answer!
Q4. For each pair of structures, circle the most stable (lowest energy) and briefly explain your choice.
Background
Topic: Conformational Stability
This question tests your ability to compare the stability of different conformers, often based on steric hindrance and torsional strain.
Key Terms and Concepts:
Steric Hindrance: Repulsion between bulky groups.
Torsional Strain: Strain due to eclipsing interactions.
Equatorial vs. Axial: In cyclohexane, bulky groups prefer equatorial positions.
Step-by-Step Guidance
Examine each pair of structures for steric interactions (bulky groups close together).
Identify which structure has fewer steric or torsional interactions.
Briefly explain why the chosen structure is more stable (e.g., "bulky groups are equatorial").
Try solving on your own before revealing the answer!
Q5. Draw both chair conformations for the given structure and circle the lowest energy conformation.
Background
Topic: Cyclohexane Chair Conformations
This question tests your ability to draw and analyze the two possible chair conformations of a substituted cyclohexane and determine which is more stable.
Key Terms and Concepts:
Chair Conformation: The most stable conformation of cyclohexane.
Axial/Equatorial Positions: Substituents can be axial (up/down) or equatorial (out to the side).
1,3-Diaxial Interactions: Steric strain when bulky groups are axial.
Step-by-Step Guidance
Draw two chair conformations, flipping the positions of all substituents (axial to equatorial and vice versa).
Place the larger groups in the equatorial positions if possible.
Identify which conformation has fewer 1,3-diaxial interactions (bulky groups axial).
Try solving on your own before revealing the answer!
Q6. Draw a resonance structure for the free radical and show the interconversion using curved arrow notation.
Background
Topic: Resonance in Free Radicals
This question tests your understanding of resonance structures and the use of curved arrows to show electron movement, especially in radical species.
Key Terms and Concepts:
Resonance Structure: Alternative Lewis structures for the same molecule, showing delocalization of electrons.
Curved Arrow Notation: Shows movement of electrons (single-headed for radicals).
Step-by-Step Guidance
Identify the location of the unpaired electron in the given structure.
Draw an alternative structure where the unpaired electron is delocalized to another atom.
Use a single-barbed (fishhook) arrow to show the movement of the single electron.
Try solving on your own before revealing the answer!
Q7. Provide the major organic product and a mechanism for the reaction (CH3 + Br2, heat).
Background
Topic: Free Radical Halogenation Mechanism
This question tests your ability to predict the major product and outline the mechanism for a radical halogenation reaction, including initiation and propagation steps.
Key Terms and Concepts:
Initiation: Formation of radicals (e.g., Br2 → 2 Br•).
Propagation: Radicals react with substrate to form new radicals and products.
Termination: Two radicals combine to end the chain reaction.
Step-by-Step Guidance
Write the initiation step: homolytic cleavage of Br2 to form two Br radicals.
Write the propagation steps: abstraction of a hydrogen atom from CH3 by Br•, then reaction of the resulting methyl radical with Br2.
Draw the structure of the major organic product formed.
Use curved arrow notation (fishhook arrows) to show electron movement in each step.
Try solving on your own before revealing the answer!
Q8. Calculate the overall ΔH° (kcal/mol) for the reaction using the given bond dissociation energies.
Background
Topic: Thermochemistry – Bond Dissociation Energies
This question tests your ability to calculate the enthalpy change (ΔH°) for a reaction using bond dissociation energies (BDEs).
Key Terms and Formulas:
Bond Dissociation Energy (BDE): Energy required to break a bond.
ΔH° Calculation: ΔH° = Σ(BDE of bonds broken) – Σ(BDE of bonds formed)
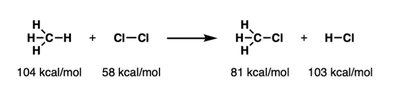
Step-by-Step Guidance
List all bonds broken in the reactants and sum their BDEs.
List all bonds formed in the products and sum their BDEs.
Subtract the total BDE of bonds formed from the total BDE of bonds broken to find ΔH°.
Try solving on your own before revealing the answer!
Q9. Which is the correct IUPAC name for the given molecule?
Background
Topic: IUPAC Nomenclature – Multiple Substituents
This question tests your ability to apply IUPAC rules to a complex branched alkane.
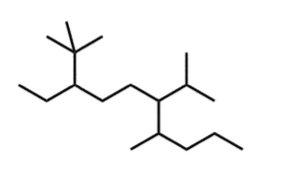
Key Terms and Concepts:
Identify the longest chain, number it, and name all substituents.
Apply alphabetical order and correct numbering for lowest set of locants.
Step-by-Step Guidance
Identify the longest continuous carbon chain in the structure.
Number the chain to give the substituents the lowest possible numbers.
Identify and name each substituent, then match the structure to the correct IUPAC name from the choices.
Try solving on your own before revealing the answer!
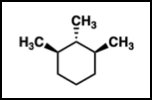
Q10. Which molecule is cis-1,3-dimethylcyclohexane?
Background
Topic: Stereochemistry of Cyclohexanes
This question tests your ability to recognize cis/trans isomerism in substituted cyclohexanes.

Key Terms and Concepts:
Cis: Both substituents on the same side of the ring (both up or both down).
Trans: Substituents on opposite sides of the ring.
Step-by-Step Guidance
Locate the 1 and 3 positions on the cyclohexane ring.
Determine which structures have both methyl groups on the same side (cis).
Match the correct structure to the answer choices.
Try solving on your own before revealing the answer!
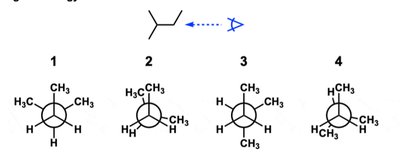
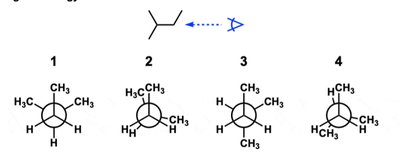
Q11. Which of the following would NOT be a Newman projection for the given line-angle drawing?
Background
Topic: Newman Projections and Line-Angle Structures
This question tests your ability to translate between line-angle drawings and Newman projections, identifying which projection does not match the structure.

Key Terms and Concepts:
Visualize the molecule down the bond indicated and compare the groups attached.
Step-by-Step Guidance
Draw the line-angle structure and identify the groups attached to each carbon in the bond of interest.
Compare each Newman projection to the actual arrangement of groups in the molecule.
Identify which projection does not match the structure.
Try solving on your own before revealing the answer!
Q12. Which cycloalkane would have the highest boiling point?
Background
Topic: Physical Properties of Cycloalkanes
This question tests your understanding of how molecular size and structure affect boiling points.
Key Terms and Concepts:
Boiling Point: Increases with molecular weight and surface area.
Cycloalkane Size: Larger rings generally have higher boiling points.
Step-by-Step Guidance
Compare the number of carbons in each cycloalkane.
Recall that larger rings have higher boiling points due to increased van der Waals forces.
Identify the largest cycloalkane in the list.
Try solving on your own before revealing the answer!
Q13. If the equilibrium constant (Keq) is 0.5, which statement must be true?
Background
Topic: Thermodynamics – Equilibrium and Gibbs Free Energy
This question tests your understanding of the relationship between Keq and ΔG° (Gibbs free energy).
Key Terms and Formulas:
Equilibrium Constant (Keq): Indicates the ratio of products to reactants at equilibrium.
Gibbs Free Energy (ΔG°): ΔG° = –RT ln Keq
Step-by-Step Guidance
Recall the formula ΔG° = –RT ln Keq.
Determine the sign of ΔG° when Keq < 1.
Relate the sign of ΔG° to whether products or reactants are favored.
Try solving on your own before revealing the answer!
Q14. Which compound cannot be formed through a termination reaction in the photochemical halogenation of ethane?
Background
Topic: Free Radical Mechanisms – Termination Steps
This question tests your understanding of which products can result from the combination of radicals in the termination step of a radical chain reaction.
Key Terms and Concepts:
Termination: Two radicals combine to form a stable molecule.
Consider which products can be formed by combining the possible radicals present in the reaction.
Step-by-Step Guidance
List the possible radicals formed during the reaction (e.g., Cl•, CH3CH2•).
Combine these radicals in all possible pairs to see which products can be formed.
Identify which product cannot be formed by any combination of these radicals.
Try solving on your own before revealing the answer!
Q15. Based on the reaction coordinate diagram for free radical bromination of ethane, choose the correct structure for the point indicated by an asterisk (*).
Background
Topic: Reaction Coordinate Diagrams and Transition States
This question tests your ability to interpret reaction coordinate diagrams and identify the structure at a specific point (e.g., transition state or intermediate).
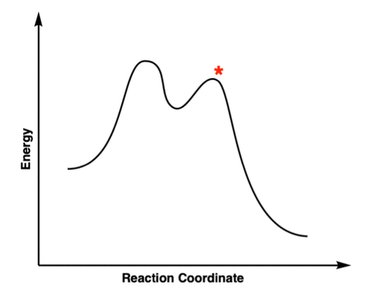
Key Terms and Concepts:
Transition State: Highest energy point along the reaction path.
Intermediate: Local energy minimum between transition states.
Step-by-Step Guidance
Identify whether the asterisk is at a peak (transition state) or valley (intermediate).
Recall the structure of the transition state or intermediate for free radical bromination.
Match the correct structure to the point indicated by the asterisk.
Try solving on your own before revealing the answer!
Q16. Which two reaction coordinates illustrate the Hammond Postulate?
Background
Topic: Hammond Postulate and Reaction Coordinate Diagrams
This question tests your understanding of the Hammond Postulate, which relates the structure of the transition state to the energies of reactants and products.
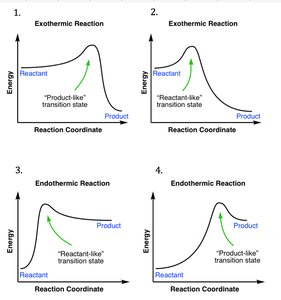
Key Terms and Concepts:
Hammond Postulate: The transition state resembles the species (reactant or product) to which it is closer in energy.
Exothermic reactions have early transition states; endothermic reactions have late transition states.
Step-by-Step Guidance
Examine each reaction coordinate diagram for the position of the transition state relative to reactants and products.
Identify which diagrams show a transition state that resembles the reactants (early) or products (late).
Select the two diagrams that best illustrate the Hammond Postulate.
Try solving on your own before revealing the answer!
Q17. Which of the following is a carbocation?
Background
Topic: Reactive Intermediates – Carbocations
This question tests your ability to recognize the structure and charge of a carbocation among various reactive intermediates.
Key Terms and Concepts:
Carbocation: A positively charged carbon atom with only six electrons in its valence shell.
Look for a carbon with a positive charge and three bonds.
Step-by-Step Guidance
Examine each structure for a carbon atom with a positive charge.
Eliminate structures with negative charges or unpaired electrons (radicals).
Identify the correct carbocation structure.
Try solving on your own before revealing the answer!
Q18. Which of the free radicals below is the most stable?
Background
Topic: Stability of Free Radicals
This question tests your understanding of the factors that stabilize free radicals, such as alkyl substitution and resonance.
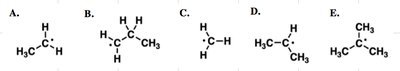
Key Terms and Concepts:
Radical Stability Order: Tertiary > Secondary > Primary > Methyl
Resonance stabilization increases radical stability.
Step-by-Step Guidance
Identify the degree of substitution for each radical (primary, secondary, tertiary).
Check for resonance stabilization in any of the radicals.
Determine which radical is most stabilized by alkyl groups or resonance.
Try solving on your own before revealing the answer!
Q19. Which reactive intermediate can best be described as both nucleophilic and electrophilic?
Background
Topic: Reactive Intermediates – Carbenes
This question tests your understanding of the dual reactivity of carbenes, which can act as both nucleophiles and electrophiles.
Key Terms and Concepts:
Carbene: A neutral species with a divalent carbon atom, containing a lone pair and an empty p orbital.
Carbenes can donate or accept electrons, making them both nucleophilic and electrophilic.
Step-by-Step Guidance
Recall the definitions of nucleophile (electron donor) and electrophile (electron acceptor).
Consider the electronic structure of each intermediate listed.
Identify which intermediate can act as both a nucleophile and an electrophile.
Try solving on your own before revealing the answer!
