 Back
BackOrganic Chemistry Exam 3 Study Guidance: Acidity, Basicity, Mechanisms, and Reaction Analysis
Study Guide - Smart Notes

Q1. In Figure 1, Group A, which hydrogen is most acidic? The best answer will provide the best one word rationale.
Background
Topic: Acidity in Organic Molecules
This question tests your understanding of how structural features (such as atom type, resonance, and induction) affect the acidity of hydrogens in organic compounds.
Key Terms:
Acidity: The tendency of a hydrogen atom to be released as a proton (H+).
Resonance: Delocalization of electrons that can stabilize the conjugate base.
Induction: Electron withdrawal through sigma bonds, affecting acidity.
Atom: The identity of the atom bonded to hydrogen (e.g., O, N, C) affects acidity.
Step-by-Step Guidance
Identify the hydrogens labeled Ha, Hb, and Hc in Group A and note their chemical environments.
Consider the atom to which each hydrogen is attached. Hydrogens attached to more electronegative atoms are generally more acidic.
Evaluate whether resonance stabilization is possible for the conjugate base formed after deprotonation. Resonance can greatly increase acidity.
Assess if induction effects are present, such as nearby electronegative groups that can stabilize the conjugate base.
Try solving on your own before revealing the answer!
Q2. In Figure 1, Group B, which answer correctly ranks the members from most acidic to least acidic?
Background
Topic: Ranking Acidity
This question tests your ability to compare acidity among different compounds based on their structure and substituents.
Key Terms:
Acidity trend: Influenced by resonance, induction, atom identity, and charge.
Ranking: Ordering compounds from most to least acidic.
Step-by-Step Guidance
Examine the structures labeled I, II, III, and IV in Group B.
Identify features that increase acidity, such as resonance stabilization or electronegative substituents.
Compare the ability of each structure to stabilize the conjugate base after deprotonation.
Arrange the compounds in order from most acidic to least acidic based on your analysis.
Try solving on your own before revealing the answer!
Q3. In Figure 1, Group C, which Nitrogen is least basic?
Background
Topic: Basicity of Nitrogen Atoms
This question tests your understanding of how structure and electron availability affect the basicity of nitrogen atoms in organic molecules.
Key Terms:
Basicity: The ability of a molecule to accept a proton (H+).
Resonance: Can decrease basicity if lone pairs are delocalized.
Electron availability: Nitrogens with lone pairs involved in resonance are less basic.
Step-by-Step Guidance
Identify the nitrogens labeled N1, N2, and N3 in Group C.
Determine if the lone pair on each nitrogen is available for protonation or involved in resonance.
Compare the basicity based on electron availability and resonance effects.
Decide which nitrogen is least basic based on your analysis.
Try solving on your own before revealing the answer!
Q4. Which leaving group (LG) would result in the lowest energy transition state? Refer to Figure 2.
Background
Topic: Leaving Group Ability and Transition State Stability
This question tests your understanding of how the nature of the leaving group affects the energy of the transition state in a reaction.
Key Terms:
Leaving Group (LG): The atom or group that departs with a pair of electrons during a reaction.
Transition State: The highest energy point along the reaction coordinate.
Stability: Better leaving groups stabilize the transition state, lowering its energy.
Step-by-Step Guidance
Review the possible leaving groups shown in Figure 2.
Recall that leaving group ability is related to how well the group can stabilize negative charge after departure.
Rank the leaving groups from best to worst based on their ability to stabilize charge.
Identify which leaving group would result in the lowest energy transition state.
Try solving on your own before revealing the answer!
Q5. Which solvent structure would you expect to best stabilize a transition state when the nucleophile/base is sodium propoxide? Refer to Figure 2.
Background
Topic: Solvent Effects on Reaction Mechanisms
This question tests your understanding of how solvent polarity and structure affect the stabilization of transition states, especially with charged nucleophiles/bases.
Key Terms:
Solvent: The medium in which the reaction occurs.
Polar Aprotic: Solvents that do not donate protons but can stabilize ions.
Transition State Stabilization: Solvents can lower activation energy by stabilizing charged intermediates.
Step-by-Step Guidance
Identify the solvent options provided in Figure 2.
Recall that polar aprotic solvents are best for stabilizing anionic nucleophiles like sodium propoxide.
Compare the structures of the solvents and their ability to stabilize the transition state.
Choose the solvent that would best stabilize the transition state.
Try solving on your own before revealing the answer!
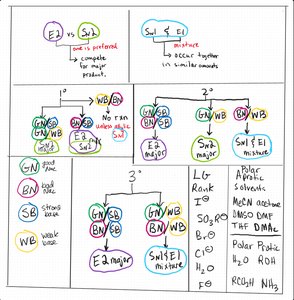
Q6. DMSO is an extremely polar solvent. Given the structure of DMSO, which nucleophile would you expect to react fastest when it is used as a solvent?
Background
Topic: Nucleophilicity in Polar Aprotic Solvents
This question tests your understanding of how polar aprotic solvents like DMSO affect the reactivity of nucleophiles.
Key Terms:
Nucleophile: A species that donates an electron pair to form a bond.
Polar Aprotic Solvent: Solvents like DMSO that do not hydrogen bond with nucleophiles, increasing their reactivity.
Nucleophilicity: The rate at which a nucleophile reacts.
Step-by-Step Guidance
Recall that polar aprotic solvents enhance the nucleophilicity of anions by not solvating them strongly.
Compare the nucleophiles listed (fluoride, fluorine, iodide, iodine) and their behavior in DMSO.
Consider the size and charge of each nucleophile and how DMSO affects their reactivity.
Identify which nucleophile would react fastest in DMSO.
Try solving on your own before revealing the answer!

Q7. Consider using a base such as NaOH on the substrate in Figure 2. When the LG is on a wedge, which product is NOT possible? Minor products should be considered as possible.
Background
Topic: Stereochemistry in Elimination Reactions
This question tests your understanding of how the spatial arrangement of the leaving group affects the possible products in elimination reactions.
Key Terms:
Leaving Group (LG): The group that departs during the reaction.
Wedge: Indicates the LG is oriented out of the plane (stereochemistry).
Elimination: Removal of a group to form a double bond.
Step-by-Step Guidance
Identify the stereochemistry of the substrate with the LG on a wedge.
Recall the requirements for anti-periplanar geometry in E2 eliminations.
Determine which products are possible based on the stereochemistry and mechanism.
Identify which product is NOT possible, considering minor products as well.
Try solving on your own before revealing the answer!
Q8. The elimination pathway is often favored at higher temperatures. Which answer best rationalizes why increasing temperature will generally increase the amount of elimination in a chemical reaction?
Background
Topic: Thermodynamics and Kinetics of Elimination Reactions
This question tests your understanding of how temperature affects the favorability of elimination versus substitution reactions.
Key Terms:
Elimination: Reaction pathway that forms a double bond.
Entropy: Measure of disorder; elimination increases entropy.
Activation Energy: Energy required to reach the transition state.
Step-by-Step Guidance
Recall that elimination reactions often result in more molecules (higher entropy).
Consider how increasing temperature affects the entropy term in the Gibbs free energy equation.
Compare the thermodynamic favorability of elimination versus substitution at higher temperatures.
Choose the rationale that best explains the effect of temperature on elimination.
Try solving on your own before revealing the answer!
Q9. Consider the following product of the reaction shown in Figure 2. Which Substrate/Reagent pairing would allow for the maximum formation of the ether shown below?
Background
Topic: Ether Formation and Reaction Optimization
This question tests your understanding of how substrate and reagent choice affects the yield of ether products.
Key Terms:
Ether: Compound with an oxygen atom connected to two alkyl or aryl groups.
Substrate: The molecule undergoing reaction.
Reagent: The chemical used to react with the substrate.
Step-by-Step Guidance
Identify the substrate and reagent options provided in Figure 2.
Recall the conditions that favor ether formation (e.g., Williamson ether synthesis).
Analyze which pairing would maximize ether formation based on mechanism and reactivity.
Choose the substrate/reagent pairing that would give the highest yield of the ether.
Try solving on your own before revealing the answer!
Q10. Which reaction(s) in Figure 3 do you expect to have a significant decrease in entropy?
Background
Topic: Entropy Changes in Reaction Mechanisms
This question tests your understanding of how reaction mechanisms affect entropy, especially when multiple reactants combine to form fewer products.
Key Terms:
Entropy: Measure of disorder; decreases when fewer molecules are formed.
Reaction Coordinate Diagram (RCD): Shows energy changes during a reaction.
Step-by-Step Guidance
Examine the reactions and their corresponding RCDs in Figure 3.
Identify reactions where two or more molecules combine to form one product (decrease in entropy).
Compare the mechanisms and products to determine which reaction(s) have significant entropy decrease.
Choose the correct answer based on your analysis.
Try solving on your own before revealing the answer!
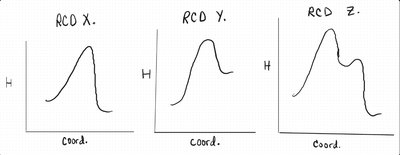
Q11. A student attempts to describe each reaction in Figure 3 in terms of the behavior of the basic component in each reaction. Which description(s) did the student get correct?
Background
Topic: Acid-Base Theory in Organic Reactions
This question tests your understanding of Lewis and Bronsted-Lowry acid/base definitions and their application to reaction mechanisms.
Key Terms:
Lewis Base: Donates an electron pair.
Bronsted-Lowry Base: Accepts a proton.
Step-by-Step Guidance
Review the descriptions for reactions M, N, and O in Figure 3.
Recall the definitions of Lewis and Bronsted-Lowry bases.
Analyze the role of the basic component in each reaction.
Determine which descriptions are correct based on the definitions.
Try solving on your own before revealing the answer!

Q12. A student attempts to match the RCDs in Figure 3 to the Reactions using fundamental principles of bond formation being exothermic, bond strength being related to the size of atoms within the bond, and a general understanding of how many mechanistic steps are required for a reaction to occur.
Background
Topic: Reaction Coordinate Diagrams and Mechanistic Steps
This question tests your ability to match reaction mechanisms to their energy profiles based on bond formation/breaking and number of steps.
Key Terms:
Reaction Coordinate Diagram (RCD): Graphical representation of energy changes during a reaction.
Exothermic: Releases energy; bond formation.
Mechanistic Steps: Number of steps in the reaction mechanism.
Step-by-Step Guidance
Review the guesses for matching reactions to RCDs.
Recall that exothermic steps lower the energy, while endothermic steps raise it.
Analyze the number of peaks in each RCD to infer the number of mechanistic steps.
Match the reactions to the RCDs based on your analysis.
Try solving on your own before revealing the answer!

Q13. A rate law investigation is conducted for Reaction O in Figure 3. Based on the results of the experiments, and the corresponding rates, what can be concluded about Reaction O?
Background
Topic: Rate Laws and Reaction Mechanisms
This question tests your ability to interpret experimental data to determine the rate law and mechanism of a reaction.
Key Terms:
Rate Law: Mathematical relationship between reactant concentrations and reaction rate.
Bimolecular: Involves two reactants in the rate-determining step.
Unimolecular: Involves one reactant in the rate-determining step.
Step-by-Step Guidance
Examine the experimental data for [H2O], [Electrophile], and rate.
Determine how changes in concentration affect the rate.
Analyze whether the rate depends on one or both reactants.
Conclude the type of rate-determining step and the reaction pathway.
Try solving on your own before revealing the answer!
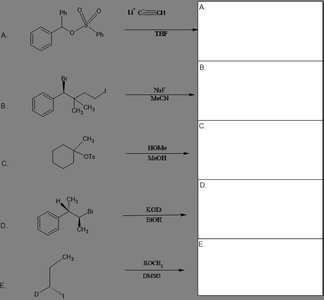
Q14. Predict the major organic products of each reaction below. Include stereochemistry when appropriate. The metals and leaving groups can be placed in the trash bin provided for 2 points extra.
Background
Topic: Reaction Products and Stereochemistry
This question tests your ability to predict the products of organic reactions, including stereochemical outcomes.
Key Terms:
Major Product: The most abundant product formed.
Stereochemistry: Spatial arrangement of atoms in the product.
Leaving Group: The group that departs during the reaction.
Step-by-Step Guidance
Identify the reactants and reagents for each reaction (A–E).
Determine the mechanism (e.g., substitution, elimination) based on the reagents and conditions.
Draw the expected product, including stereochemistry where relevant.
Consider removing metals and leaving groups for extra points.
Try solving on your own before revealing the answer!
Q15. Elimination can occur via an E1 or an E2 mechanism. The two reactions are shown below can enforce a particular mechanism according to the base being used. Draw the corresponding intermediate and/or chair conformation for the cyclic structures that explains the difference in outcomes. For the linear structure use a Newman projection alongside any intermediates to explain the difference in outcomes. Each answer will be supported by a few words and concepts to go alongside your diagrams.
Background
Topic: E1 vs E2 Elimination Mechanisms
This question tests your understanding of elimination mechanisms, intermediates, and stereochemical requirements.
Key Terms:
E1 Mechanism: Two-step elimination with carbocation intermediate.
E2 Mechanism: One-step elimination requiring anti-periplanar geometry.
Chair Conformation: 3D structure of cyclohexane rings.
Newman Projection: Visualizes bond angles and spatial relationships in linear molecules.
Step-by-Step Guidance
Identify which reaction enforces E1 and which enforces E2 based on the base used.
Draw the intermediate for E1 (carbocation) and the chair conformation for E2 (anti-periplanar arrangement).
For the linear structure, use a Newman projection to show the required geometry for E2.
Support your diagrams with brief explanations of the mechanistic differences.
Try solving on your own before revealing the answer!
Q16. A mechanism is shown below, with all of the intermediates accurately drawn. You will draw in the correct mechanistic arrows and identify the mechanistic pattern you are using. You can draw your answer on top of the structures below. The patterns should be placed on the equilibrium arrows. Here is a pattern bank: NA, R, LG, E, PT
Background
Topic: Mechanistic Arrow-Pushing and Pattern Identification
This question tests your ability to use arrow-pushing to show electron movement and identify mechanistic steps.
Key Terms:
Arrow-Pushing: Visual representation of electron movement.
Mechanistic Pattern: NA (nucleophilic attack), R (rearrangement), LG (leaving group), E (elimination), PT (proton transfer).
Step-by-Step Guidance
Review the intermediates and identify the electron movement required for each step.
Draw the correct arrows to show nucleophilic attack, rearrangement, leaving group departure, elimination, and proton transfer as needed.
Label each step with the appropriate mechanistic pattern from the pattern bank.
Place the pattern labels on the equilibrium arrows as instructed.
Try solving on your own before revealing the answer!
Q17. Three pKas are provided below. Please match the pKas to the structures shown and draw the requested conjugate species.
Background
Topic: pKa Matching and Conjugate Species Drawing
This question tests your ability to match pKa values to structures and draw the conjugate acid/base species.
Key Terms:
pKa: Measure of acidity; lower pKa means stronger acid.
Conjugate Species: The form of the molecule after gaining or losing a proton.
Step-by-Step Guidance
Review the structures and pKa values provided.
Match each pKa to the correct structure based on acidity trends.
Draw the conjugate acid or base for each structure as requested.
Check your work for accuracy in matching and drawing.
Try solving on your own before revealing the answer!
