 Back
BackOrganic Chemistry: Lewis Structures, Hybridization, and Molecular Properties
Study Guide - Smart Notes

Q1. Which of the following is the most likely Lewis structure for C2H2?
Background
Topic: Lewis Structures
This question tests your ability to draw and recognize correct Lewis structures for organic molecules, specifically acetylene (C2H2).
Key Terms and Concepts:
Lewis Structure: A diagram showing all the valence electrons in a molecule, including bonds and lone pairs.
Octet Rule: Atoms (especially C, N, O, F) tend to form bonds until they are surrounded by 8 valence electrons.
Bonding: C2H2 is a simple alkyne, so expect a triple bond between the two carbons.
Step-by-Step Guidance
Count the total number of valence electrons for C2H2 (C = 4 each, H = 1 each).
Arrange the atoms: Carbon atoms are usually central, hydrogens are terminal.
Connect the atoms with single bonds first, then add multiple bonds as needed to satisfy the octet rule for carbon.
Check that all atoms (except H) have a complete octet and that the total number of electrons matches your count.
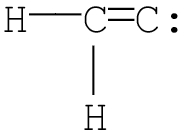
Try solving on your own before revealing the answer!
Q2. Which of the following Lewis structures is correct for CH2N2?
Background
Topic: Lewis Structures and Formal Charges
This question tests your ability to draw and evaluate Lewis structures for molecules containing multiple bonds and heteroatoms (N, in this case).
Key Terms and Concepts:
Lewis Structure: Shows all valence electrons, bonds, and lone pairs.
Formal Charge: Used to determine the most stable Lewis structure.
Octet Rule: Each atom (except H) should have 8 electrons around it if possible.
Step-by-Step Guidance
Count the total number of valence electrons for CH2N2 (C = 4, H = 1 each, N = 5 each).
Arrange the atoms: Carbon is usually central, hydrogens are terminal, and nitrogens are connected to carbon.
Draw single bonds first, then add multiple bonds or lone pairs to satisfy the octet rule and minimize formal charges.
Calculate formal charges for each atom to determine the most stable structure.
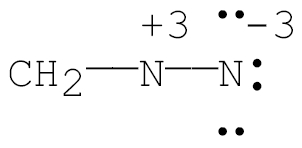
Try solving on your own before revealing the answer!
Q3. What are the formal charges on nitrogen and on the starred oxygen in the following species?
Background
Topic: Formal Charges in Lewis Structures
This question tests your ability to assign formal charges to atoms in a molecule based on their bonding and lone pairs.
Key Terms and Formulas:
Formal Charge Formula:
Step-by-Step Guidance
Identify the number of valence electrons for N and O (N = 5, O = 6).
Count the number of non-bonding electrons (lone pairs) on each atom.
Count the number of bonding electrons (shared in bonds) for each atom.
Apply the formal charge formula for both the nitrogen and the starred oxygen.
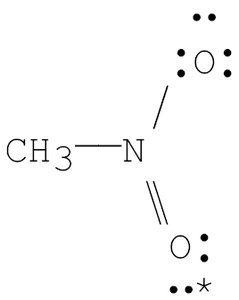
Try solving on your own before revealing the answer!
Q5. Which carbons in the following compound are sp hybridized?
Background
Topic: Hybridization
This question tests your understanding of how to assign hybridization states to carbon atoms in organic molecules, especially those involved in multiple bonds.
Key Terms and Concepts:
sp Hybridization: Carbon with two regions of electron density (usually a triple bond or two double bonds).
sp2 Hybridization: Carbon with three regions of electron density (usually a double bond).
sp3 Hybridization: Carbon with four regions of electron density (single bonds only).
Step-by-Step Guidance
Identify all the carbon atoms in the structure and number them as shown.
Look for carbons involved in triple bonds or with two regions of electron density—these are sp hybridized.
Assign the hybridization to each carbon based on its bonding pattern.
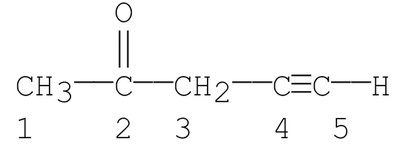
Try solving on your own before revealing the answer!
