 Back
BackOrganic Chemistry Practice Exam 3 – Step-by-Step Guidance
Study Guide - Smart Notes

Q1. What is the IUPAC name for the following compound?
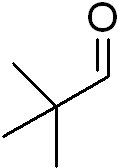
Background
Topic: Nomenclature of Aldehydes
This question tests your ability to apply IUPAC rules to name an aldehyde with branching.
Key Terms and Concepts:
Aldehyde: A compound containing a carbonyl group (C=O) bonded to at least one hydrogen.
IUPAC Naming: Find the longest carbon chain containing the aldehyde group, number from the carbonyl carbon, and name substituents as prefixes.
Step-by-Step Guidance
Identify the longest continuous carbon chain that contains the aldehyde group. The aldehyde carbon is always carbon 1.
Number the chain starting from the carbonyl carbon (the aldehyde group).
Identify and name any substituents attached to the main chain. In this case, look for alkyl groups attached to the chain.
Combine the substituent names and positions with the parent chain name, ending with "-al" for aldehydes.
Try solving on your own before revealing the answer!
Final Answer: 2,2-dimethylpropanal
The parent chain is three carbons (propanal), with two methyl groups on carbon 2.
Q2. Which of the following is the correct structure for benzophenone?
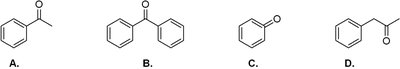
Background
Topic: Aromatic Ketones
This question tests your ability to recognize the structure of benzophenone, a common aromatic ketone.
Key Terms and Concepts:
Benzophenone: A ketone with two phenyl groups attached to the carbonyl carbon (diphenyl ketone).
Structure: The central carbonyl (C=O) is bonded to two benzene rings.
Step-by-Step Guidance
Look for a structure with a carbonyl group (C=O) directly attached to two benzene rings.
Eliminate any structures that have only one benzene ring or have the carbonyl in the wrong position.
Compare the remaining options to the definition of benzophenone.
Try solving on your own before revealing the answer!
Final Answer: Structure B
Benzophenone is diphenyl ketone, with two phenyl groups attached to the carbonyl carbon.
Q3. What is the IUPAC name for the following compound?
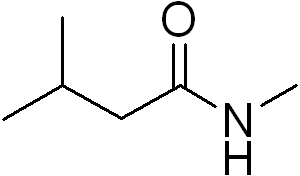
Background
Topic: Amide Nomenclature
This question tests your ability to name amides with alkyl substituents on both the nitrogen and the carbon chain.
Key Terms and Concepts:
Amide: A compound with a carbonyl group bonded to a nitrogen atom.
N-substitution: Alkyl groups attached to the nitrogen are indicated with an "N-" prefix.
Parent chain: The longest chain containing the carbonyl group is the base name.
Step-by-Step Guidance
Identify the parent chain containing the carbonyl group (amide carbon is position 1).
Number the chain from the carbonyl carbon.
Identify and name any substituents on the chain and on the nitrogen atom.
Combine the names, using "N-" for groups on the nitrogen, and end with "amide".
Try solving on your own before revealing the answer!
Final Answer: N-methyl 3-methylbutanamide
The parent chain is butanamide, with a methyl group on carbon 3 and a methyl group on the nitrogen.
Q5. Which of the following substrates cannot be used as an immediate precursor to synthesize an ester?
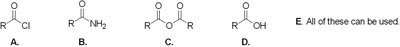
Background
Topic: Ester Synthesis
This question tests your understanding of which functional groups can be directly converted to esters.
Key Terms and Concepts:
Esterification: Esters can be synthesized from carboxylic acids, acid chlorides, and anhydrides, but not directly from all carboxylic acid derivatives.
Functional Groups: Recognize the reactivity of acid chlorides, amides, anhydrides, and carboxylic acids toward ester formation.
Step-by-Step Guidance
Review each structure and identify the functional group present.
Recall which functional groups can be directly converted to esters (e.g., via reaction with alcohols).
Identify any group that cannot be converted to an ester in a single step without prior transformation.
Try solving on your own before revealing the answer!
Final Answer: B (Amide)
Amides cannot be directly converted to esters without first being hydrolyzed to a carboxylic acid or another reactive intermediate.
Q14. What is the immediate product for the following reaction?
Background
Topic: Nucleophilic Addition to Carbonyls (Cyanohydrin Formation)
This question tests your understanding of the reaction between an aldehyde and cyanide ion in acidic conditions.
Key Terms and Concepts:
Cyanohydrin Formation: Aldehydes and ketones react with HCN (or NaCN/HCl) to form cyanohydrins.
Mechanism: Cyanide ion attacks the carbonyl carbon, followed by protonation of the oxygen.
Step-by-Step Guidance
Identify the nucleophile (CN-) and electrophile (carbonyl carbon of the aldehyde).
Show the nucleophilic attack of CN- on the carbonyl carbon, forming a tetrahedral intermediate.
Protonate the oxygen atom with HCl to yield the cyanohydrin product.
Try solving on your own before revealing the answer!
Final Answer: Cyclobutyl cyanohydrin
The product is a cyclobutyl group with a hydroxyl and a nitrile group on the same carbon.
