 Back
BackOrganic Chemistry Study Guidance: Acidity, SN2, Addition-Elimination, pKa, Mechanisms, and Pharmaceutical Structure
Study Guide - Smart Notes

Q1A. Arrange the compounds below based on their increasing acidity.
Background
Topic: Acidity and pKa in Organic Chemistry
This question tests your understanding of how to compare the acidity of different organic compounds, which is fundamental for predicting reactivity and stability in organic reactions.
Key Terms and Formulas:
Acidity: The tendency of a compound to donate a proton (H+).
pKa: The negative logarithm of the acid dissociation constant; lower pKa means higher acidity.
Electronegativity: More electronegative atoms stabilize negative charge, increasing acidity.
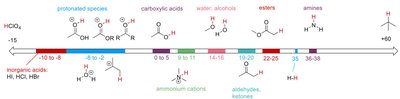
Step-by-Step Guidance
Identify the functional groups present in each compound (e.g., carboxylic acids, alcohols, amines).
Recall the general pKa ranges for these groups using the provided pKa scale.
Consider the effect of substituents (e.g., electronegative atoms, resonance) on acidity.
Arrange the compounds from lowest to highest pKa (highest to lowest acidity).
Try solving on your own before revealing the answer!
Q1B. A set of compounds will be submitted to SN2 reaction conditions using KOH as a reagent. Provide the correct structure of the expected product. Arrange the 4 compounds below according to their increasing rate in the SN2 reaction.
Background
Topic: SN2 Mechanism and Reactivity
This question tests your ability to predict the product of an SN2 reaction and to compare the rates of SN2 reactions for different substrates.
Key Terms and Formulas:
SN2 Reaction: Bimolecular nucleophilic substitution; rate depends on substrate structure.
Rate:
Steric hindrance: Less hindered (primary) substrates react faster than more hindered (secondary, tertiary).
Step-by-Step Guidance
Identify the leaving group and nucleophile (KOH provides OH-).
Draw the product for each compound after nucleophilic substitution.
Compare the degree of substitution (primary, secondary, tertiary) for each substrate.
Arrange the compounds in order of increasing SN2 rate based on steric hindrance.
Try solving on your own before revealing the answer!
Q1C. Arrange the compounds below according to their reactivity in addition-elimination reactions.
Background
Topic: Addition-Elimination Mechanism
This question tests your understanding of which functional groups are most reactive in nucleophilic addition-elimination reactions, such as acyl substitution.
Key Terms and Formulas:
Addition-Elimination: Common for carboxylic acid derivatives (e.g., esters, amides, anhydrides).
Reactivity order: Acyl chlorides > anhydrides > esters > amides.
Step-by-Step Guidance
Identify the functional group for each compound (e.g., acyl chloride, ester, amide).
Recall the general reactivity order for these groups.
Consider resonance and leaving group ability.
Arrange the compounds from least to most reactive in addition-elimination.
Try solving on your own before revealing the answer!
Q1D. Arrange the compounds according to their pKa constants.
Background
Topic: pKa Comparison
This question tests your ability to compare the acidity of compounds based on their pKa values, which is essential for predicting proton transfer reactions.
Key Terms and Formulas:
pKa: Lower pKa means stronger acid.
Functional group effects: Carboxylic acids, alcohols, amines, etc.

Step-by-Step Guidance
Identify the functional group for each compound.
Use the pKa scale to estimate the pKa for each compound.
Consider substituent effects (e.g., electron-withdrawing groups lower pKa).
Arrange the compounds from lowest to highest pKa.
Try solving on your own before revealing the answer!
Q2A–2D. In the following reactions, draw the correct organic product. The inorganic side products are not evaluated.
Background
Topic: Organic Reaction Products
These questions test your ability to predict the main organic product of a reaction, which is a key skill in organic synthesis.
Key Terms and Formulas:
Reaction mechanism: Not compulsory, but understanding helps predict products.
Functional group transformations: Recognize what changes occur in the reaction.
Step-by-Step Guidance
Identify the starting material and reagents for each reaction.
Recall the typical outcome for the reaction type (e.g., substitution, addition, elimination).
Draw the main organic product, focusing on the transformation of the functional group.
Check for regioselectivity or stereochemistry if relevant.
Try solving on your own before revealing the answer!
Q3. Provide a mechanism for the reaction below. (Any reasonable intermediate will get some partial score as long as it is generated by a meaningful reaction step).
Background
Topic: Reaction Mechanisms
This question tests your ability to propose a stepwise mechanism for an organic reaction, including intermediates and electron flow.
Key Terms and Formulas:
Mechanism: Sequence of steps showing how reactants convert to products.
Intermediates: Species formed during the reaction (e.g., carbocations, carbanions).
Arrow pushing: Use curved arrows to show electron movement.
Step-by-Step Guidance
Identify the starting material and product.
Determine the type of reaction (e.g., nucleophilic addition, elimination).
Draw the first step, showing electron movement with arrows.
Propose reasonable intermediates and subsequent steps.
Try solving on your own before revealing the answer!
Bonus Question: On the way from ADL1 to UMCG, an artist made the following stained glass. Please draw the correct structure to the rectangle on the right. To which class of pharmaceuticals does this molecule belong?
Background
Topic: Pharmaceutical Structure Identification
This question tests your ability to interpret a chemical structure from a visual representation and classify it according to pharmaceutical categories.
Key Terms and Formulas:
Structure drawing: Translate condensed formulas to line structures.
Pharmaceutical class: Identify based on functional groups and overall structure.

Step-by-Step Guidance
Carefully read the condensed formula in the stained glass.
Draw the structure step-by-step, starting from the leftmost group.
Identify key functional groups (e.g., amide, carboxylic acid, amine).
Compare the structure to known pharmaceutical classes (e.g., amino acids, peptides).
Try solving on your own before revealing the answer!
Final Answer
For Q1A: The correct order of acidity is based on functional group and substituent effects, as shown on the pKa scale.
For Q1B: The SN2 product is the compound with OH replacing the leaving group; the rate order is primary > secondary > tertiary.
For Q1C: Reactivity order is acyl chloride > anhydride > ester > amide.
For Q1D: pKa order follows the pKa scale provided.
For Q2A–2D: The main organic product is drawn based on the reaction type.
For Q3: The mechanism includes reasonable intermediates and electron flow.
Bonus: The structure in the stained glass is a peptide (amino acid sequence), belonging to the class of peptide pharmaceuticals.
