 Back
BackOrganic Chemistry Study Notes: Structure, Bonding, Acids/Bases, Functional Groups, and Stereochemistry
Study Guide - Smart Notes

Structure and Bonding
Atomic Structure and Orbitals
Understanding atomic structure is fundamental to organic chemistry. Atoms consist of a nucleus (protons and neutrons) surrounded by electrons in orbitals. The arrangement of electrons determines chemical bonding and molecular geometry.
Key Point: Electrons occupy orbitals (s, p, d, f) with specific shapes and energies.
Key Point: The valence shell is the outermost electron shell, crucial for bonding.
Example: Carbon (C) has 6 electrons: 1s2 2s2 2p2.

Bonding Types
Covalent bonds form when atoms share electrons. Ionic bonds result from electron transfer. The type of bond affects molecular properties.
Key Point: Covalent bonds are common in organic compounds.
Key Point: Ionic bonds occur between metals and nonmetals.
Example: H2O (water) has covalent bonds; NaCl (salt) has ionic bonds.
Hybridization and Molecular Geometry
Hybridization describes the mixing of atomic orbitals to form new hybrid orbitals. This determines the geometry of molecules.
Key Point: sp3 hybridization leads to tetrahedral geometry (109.5° angles).
Key Point: sp2 hybridization leads to trigonal planar geometry (120° angles).
Key Point: sp hybridization leads to linear geometry (180° angles).
Example: Methane (CH4) is sp3 hybridized; ethene (C2H4) is sp2; acetylene (C2H2) is sp.


Acids and Bases; Functional Groups
Acid-Base Concepts
Acids donate protons (H+), bases accept protons. The strength of acids and bases is measured by their dissociation constants.
Key Point: Ka is the acid dissociation constant. Larger Ka means stronger acid.
Key Point: pKa is the negative logarithm of Ka; lower pKa means stronger acid.
Example: HCl is a strong acid; acetic acid (CH3COOH) is a weak acid.
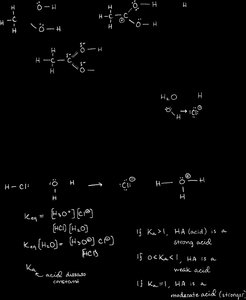
Functional Groups
Functional groups are specific groups of atoms within molecules that determine chemical reactivity and properties.
Key Point: Common functional groups include alcohols (–OH), amines (–NH2), carboxylic acids (–COOH), and halides (–Cl, –Br).
Key Point: Functional groups are used to classify organic compounds.
Example: Ethanol (CH3CH2OH) contains an alcohol group.
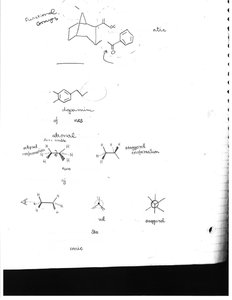
Structure and Stereochemistry of Alkanes
Alkane Structure and Nomenclature
Alkanes are saturated hydrocarbons with only single bonds. Their structure and naming follow IUPAC rules.
Key Point: Alkanes have the general formula CnH2n+2.
Key Point: Naming involves identifying the longest carbon chain and substituents.
Example: 2-methylpentane is an alkane with a methyl group on the second carbon.

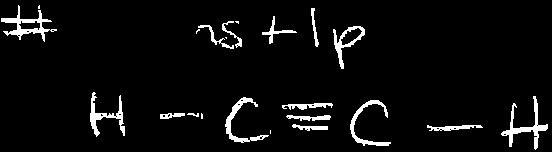
Conformations of Alkanes
Alkanes can adopt different conformations due to rotation around single bonds. The most stable conformation is the staggered form.
Key Point: Staggered conformation is more stable than eclipsed due to minimized electron repulsion.
Key Point: Conformational analysis is important for understanding reactivity.
Example: Ethane has staggered and eclipsed conformations.
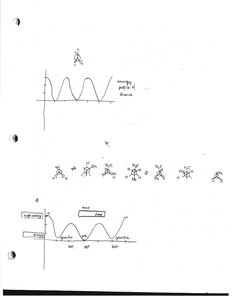

Stereochemistry
Isomerism and Chirality
Stereochemistry deals with the spatial arrangement of atoms. Isomers have the same formula but different structures. Chirality is a property of molecules that are non-superimposable on their mirror images.
Key Point: Enantiomers are mirror-image isomers.
Key Point: Diastereomers are non-mirror-image stereoisomers.
Example: Lactic acid has two enantiomers.

Newman Projections and Stereochemical Representations
Newman projections are used to visualize the spatial arrangement of atoms in molecules, especially for conformational analysis.
Key Point: Newman projections show the front and back atoms along a bond axis.
Key Point: Used to distinguish between staggered and eclipsed conformations.
Example: Butane's anti and gauche conformations.

Acids and Bases: Mechanisms and Stability
Acid-Base Mechanisms
Acid-base reactions involve proton transfer. The stability of the conjugate base determines acid strength.
Key Point: More stable conjugate bases make stronger acids.
Key Point: Resonance and electronegativity affect stability.
Example: Sulfuric acid (H2SO4) is strong due to resonance stabilization.

Functional Groups and Reactivity
Functional Group Classification
Functional groups are classified based on their chemical behavior. Understanding their reactivity is essential for predicting reactions.
Key Point: Alcohols, amines, carboxylic acids, and halides are common functional groups.
Key Point: Reactivity depends on electronic and steric factors.
Example: Alcohols can be oxidized to aldehydes or ketones.

Summary Table: Hybridization and Geometry
The following table summarizes hybridization, geometry, and bond angles for common atomic arrangements:
Hybridization | Geometry | Bond Angle |
|---|---|---|
sp3 | Tetrahedral | 109.5° |
sp2 | Trigonal planar | 120° |
sp | Linear | 180° |
Additional info:
Some content was inferred from context and standard organic chemistry knowledge to ensure completeness.
Images were included only when directly relevant to the explanation.
