 Back
BackOrganometallic Compounds: Structure, Reactivity, and Mechanisms
Study Guide - Smart Notes

Organometallic Compounds
Nomenclature
Organometallic compounds are chemical species in which a carbon atom is directly bonded to a metal. The metal is considered the “parent chain” in nomenclature, and the organic group is named as a substituent.
Organometallic compounds are metallic derivatives of organic molecules.
The metal atom is prioritized in naming conventions.
Carbanions and Bond Polarity
Organometallic compounds contain highly polar covalent bonds between carbon and metal atoms. The carbon atom in these bonds behaves as a carbanion, making it highly nucleophilic and reactive in organic synthesis.
Carbanions are carbon atoms with a negative charge, often stabilized by the metal.
These compounds are important sources of nucleophilic carbon.
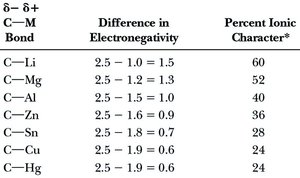
Table Purpose: This table compares the difference in electronegativity between carbon and various metals, and the resulting percent ionic character of the C–M bond. Higher ionic character correlates with greater reactivity and nucleophilicity of the carbon atom.
C–M Bond | Difference in Electronegativity | Percent Ionic Character |
|---|---|---|
C–Li | 1.5 | 60 |
C–Mg | 1.3 | 52 |
C–Al | 1.0 | 40 |
C–Zn | 0.9 | 36 |
C–Sn | 0.7 | 28 |
C–Cu | 0.6 | 24 |
C–Hg | 0.6 | 24 |
Preparation of Organolithium Compounds
Organolithium compounds are synthesized by reacting alkyl halides with alkali metals, typically lithium. The reaction is sensitive to the solvent, which can shift the equilibrium between covalent and ionic forms.
Common solvents: ethers and alkanes.
Organolithium compounds are in equilibrium with their ionic state.
Mechanism of Organometallic Formation
The formation of organometallic compounds generally follows a three-step mechanism:
Step 1: A radical electron resides in an antibonding orbital of the alkyl halide.
Step 2: The anion radical fragments, producing an alkyl radical and a halide ion.
Step 3: The alkyl radical rapidly combines with the alkali metal atom, forming the organometallic compound.
In apolar solvents, ions may combine as a crystal lattice precipitate.
Reactivity of sp2 Hybridized Carbons
Alkyl halides containing sp2 hybridized carbons (such as alkenes and aromatic rings) can also undergo organometallic formation, though their reactivity is generally lower than sp3 carbons.
Grignard Reagents
Grignard reagents are organomagnesium compounds, typically prepared by reacting alkyl halides with magnesium metal in ether solvents.
Common solvents: Diethyl ether (boiling point 35°C) and tetrahydrofuran (THF, boiling point 60°C).
Diethyl ether is used for more reactive reagents; THF is used for less reactive ones.
General reaction:
Mechanism of Grignard Formation
The mechanism for Grignard reagent formation is similar to that of organolithium compounds, involving radical intermediates and the combination of alkyl radicals with magnesium.
Factors Affecting Reactivity
The reactivity of alkyl halides in forming organometallic compounds depends on the halide and the hybridization of the carbon:
Alkyl iodides are most reactive; alkyl fluorides are least reactive.
Alkenyl and aryl carbons are reactive, but less so than sp3 carbons.
Organometallic compounds are strongly basic; aprotic solvents must be used to prevent unwanted reactions.
Alkyne Chemistry Review
Terminal alkynes can be deprotonated by strong bases (such as NH2-) to form nucleophilic alkynide ions. When sodium is included, the resulting sodium alkynide exhibits organometallic character.
Deprotonation reaction:
Alkynide ions are useful nucleophiles in organic synthesis.
Summary of Organometallic Compound Preparation
Three common types of organometallic compounds can be synthesized:
Organolithium compounds:
Organomagnesium halides (Grignard reagents):
Sodium alkynides:
Organometallic compounds are valuable sources of nucleophilic carbon for organic synthesis, enabling a wide range of transformations.
Additional info: The percent ionic character of the C–M bond influences the reactivity and stability of organometallic compounds. Higher ionic character generally increases nucleophilicity and basicity, making these compounds more reactive in organic reactions.
