 Back
BackPericyclic Reactions: Applications in Reserpine Synthesis and Precalciferol Chemistry
Study Guide - Smart Notes

Pericyclic Reactions in Organic Synthesis
Introduction to Pericyclic Reactions
Pericyclic reactions are a class of organic reactions that proceed via concerted cyclic rearrangements of bonding electrons. These reactions are governed by orbital symmetry and are fundamental in the construction of complex molecular architectures in organic synthesis.
Definition: Pericyclic reactions involve the redistribution of electrons through a cyclic transition state, without the formation of intermediates.
Types: Common types include cycloadditions, electrocyclic reactions, sigmatropic rearrangements, and group transfer reactions.
Significance: These reactions are stereospecific and regioselective, making them valuable tools in the synthesis of natural products and pharmaceuticals.
Application: Reserpine Synthesis
Key Steps in the Synthetic Sequence
The synthesis of Reserpine, a complex indole alkaloid, utilizes pericyclic reactions to construct its polycyclic framework. The following steps highlight the use of cycloaddition and rearrangement reactions in the synthetic route.
Diels-Alder Reaction: The initial step involves a [4+2] cycloaddition (Diels-Alder reaction) between a diene and a dienophile, forming a six-membered ring in a stereospecific manner.
Endo Transition State: The reaction proceeds via the endo transition state, favoring the formation of the observed stereochemistry.
Functional Group Transformations: Subsequent steps include reduction, bromination, and nucleophilic substitution, which further elaborate the molecular structure.
Regio- and Stereochemistry: The pericyclic nature of the key steps ensures control over the regiochemistry and stereochemistry of the product.
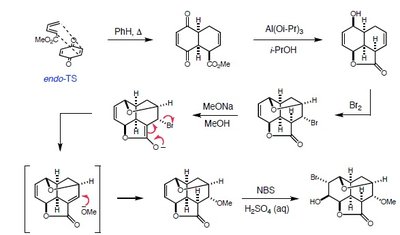
Mechanistic Details
Concerted Mechanism: The Diels-Alder reaction proceeds via a concerted mechanism, with simultaneous bond formation and electron movement.
Transition State: The endo transition state is stabilized by secondary orbital interactions, leading to the observed product.
Subsequent Steps: The sequence includes nucleophilic attack, elimination, and oxidation, each step carefully controlled to preserve the stereochemistry set in the cycloaddition.
Application: Precalciferol and Vitamin D Chemistry
Photochemical and Thermal Pericyclic Reactions
The transformation of Precalciferol to Vitamin D and related compounds involves pericyclic reactions, specifically electrocyclic ring openings and closings, which are triggered by light (photochemical) or heat (thermal) activation.
Electrocyclic Reactions: These reactions involve the conversion of a π-system to a σ-bond (or vice versa) through a concerted cyclic rearrangement.
Photochemical Activation: UV light induces the ring opening of Precalciferol, leading to the formation of Vitamin D derivatives.
Thermal Activation: Heat can also drive electrocyclic reactions, but the stereochemical outcome depends on the number of π electrons and the activation mode (conrotatory vs. disrotatory).
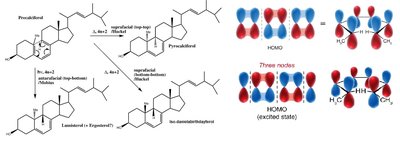
Orbital Symmetry and Stereochemistry
Woodward-Hoffmann Rules: The stereochemistry of electrocyclic reactions is predicted by the Woodward-Hoffmann rules, which relate the number of π electrons and the mode of activation to the reaction pathway.
HOMO Analysis: The highest occupied molecular orbital (HOMO) of the reacting system determines whether the reaction proceeds via a conrotatory or disrotatory mechanism.
Photochemical vs. Thermal:
Thermal reactions of 4n π systems proceed via conrotatory motion; 4n+2 π systems via disrotatory motion.
Photochemical activation reverses these preferences.
Examples and Applications
Vitamin D Synthesis: The conversion of 7-dehydrocholesterol to Precalciferol and then to Vitamin D3 is a classic example of photochemically induced electrocyclic ring opening.
Biological Relevance: These transformations are essential for the biosynthesis of Vitamin D in the skin upon exposure to sunlight.
Summary Table: Types of Pericyclic Reactions
Type | Example | Activation | Key Feature |
|---|---|---|---|
Cycloaddition | Diels-Alder (Reserpine synthesis) | Thermal | Forms rings, stereospecific |
Electrocyclic | Precalciferol to Vitamin D | Thermal/Photochemical | Ring opening/closing, stereospecific |
Sigmatropic | Cope, Claisen rearrangements | Thermal | Group migration, concerted |
Key Concepts and Equations
General Equation for Diels-Alder Reaction:
Electrocyclic Ring Opening:
Woodward-Hoffmann Selection Rule:
Additional info: The images provided illustrate the mechanistic steps in the Reserpine synthesis (image_1) and the photochemical/thermal transformations of Precalciferol, including molecular orbital diagrams (image_2), which are central to understanding pericyclic reaction mechanisms.
