 Back
BackRadical Reactions in Organic Chemistry: Structure, Stability, and Halogenation Mechanisms
Study Guide - Smart Notes

Radical Reactions
Introduction to Radicals
Radicals are highly reactive intermediates in organic chemistry, characterized by a single unpaired electron. Their reactivity stems from the atom's lack of a complete octet, making them eager to react and achieve stability. Radicals are electrically neutral and are denoted by a single dot next to the atom symbol. Even a hydrogen atom (H•) is technically a radical.
Definition: A radical is an atom or molecule with one unpaired electron.
Notation: The unpaired electron is shown as a dot (•).
Reactivity: Radicals are extremely reactive due to their incomplete octet.
Types of Bond Cleavage
Homolytic vs. Heterolytic Cleavage
Bond cleavage in organic molecules can occur in two distinct ways: homolytic and heterolytic. These processes determine the nature of the products formed and the electron distribution.
Homolytic Cleavage: Each atom takes one electron from the bond, forming two radicals. This is depicted using two half-headed curved arrows (fishhooks).
Heterolytic Cleavage: Both electrons from the bond go to one atom, forming a cation and an anion. This is shown with a full-headed curved arrow.
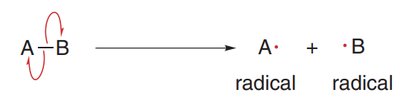
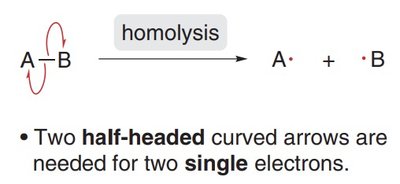
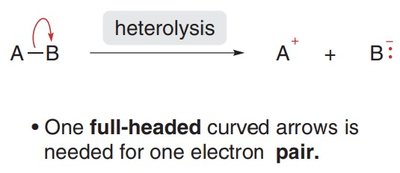
Radical Structure
Hybridization and Geometry of Carbon Radicals
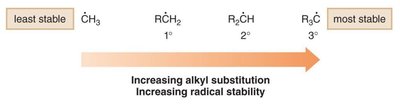
Carbon radicals are typically sp2-hybridized and exhibit a trigonal planar geometry. The unpaired electron resides in an unhybridized p orbital, perpendicular to the plane of the molecule. The classification of radicals as primary (1°), secondary (2°), or tertiary (3°) depends on the number of alkyl groups attached to the radical center.
sp2 Hybridization: Carbon radicals are planar, with bond angles of approximately 120°.
Classification:
Primary (1°): One alkyl group
Secondary (2°): Two alkyl groups
Tertiary (3°): Three alkyl groups
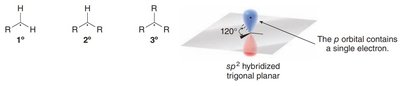
Radical Stability
Factors Affecting Radical Stability
The stability of carbon radicals increases with greater alkyl substitution. Tertiary radicals are the most stable, followed by secondary, primary, and methyl radicals. This trend is due to hyperconjugation and inductive effects from alkyl groups. Notably, secondary and primary radicals do not undergo rearrangement.
Stability Order: 3° > 2° > 1° > methyl
Reason: Alkyl groups stabilize radicals via hyperconjugation and electron donation.
Rearrangement: 2° and 1° radicals do not rearrange.
Formation of Radicals
Methods of Radical Generation
Radicals are typically formed by adding energy to break bonds. Common methods include exposure to light (hν), heat (Δ), or the use of radical initiators such as peroxides (RO–OR). Peroxides have weak O–O bonds that readily undergo homolysis to generate radicals.
Light (hν): Photochemical cleavage of bonds.
Heat (Δ): Thermal cleavage of bonds.
Radical Initiators: Peroxides (RO–OR) are commonly used.
Radical Reactions
General Reactivity of Radicals
Radicals seek to achieve an octet and react with σ-bonds, π-bonds, or other radicals. Key reactions include halogenation of alkanes, halogenation at allylic carbons, addition to π-bonds (e.g., HBr addition), polymerization, and radical-radical coupling.
Halogenation of Alkanes: Conversion of alkanes to alkyl halides.
Halogenation at Allylic Carbons: Selective functionalization at allylic positions.
Addition to π-Bonds: Radicals can add to double bonds.
Polymerization: Initiation of chain reactions in polymer synthesis.
Radical Coupling: Two radicals combine to form a stable bond.
Radical Halogenation of Alkanes
Mechanism and Scope
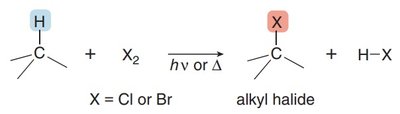
Radical halogenation is a substitution reaction where a halogen atom replaces a hydrogen atom in an alkane. This reaction is effective with chlorine (Cl2) and bromine (Br2) in the presence of light or heat. The process involves three main steps: initiation, propagation, and termination.
Initiation: Formation of radicals by homolysis.
Propagation: Radicals react with reactants to form new radicals and products.
Termination: Two radicals combine to form a stable molecule.
Product Mixtures and Selectivity
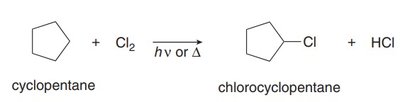
Halogenation can occur at any hydrogen atom in the molecule, often resulting in a mixture of alkyl halide products. Monohalogenation leads to different products depending on the structure of the alkane.
Product Diversity: Multiple products possible due to different hydrogen types.
Example: Halogenation of cyclohexane and cyclopentane.
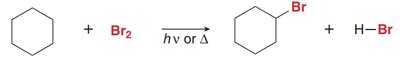
Mechanism of Radical Halogenation
Stepwise Mechanism
The radical halogenation mechanism consists of three distinct steps:
Initiation: Homolysis of the weakest bond (usually X–X) by light or heat forms two radicals.
Propagation:
Step 1: Halogen radical abstracts a hydrogen atom, forming HX and a carbon radical.
Step 2: Carbon radical abstracts a halogen atom from X2, forming the alkyl halide and regenerating the halogen radical.
Termination: Two radicals combine to form a stable bond, often leading to side-products.
Energetics and Rate-Determining Step
Energy Profile and Thermodynamics
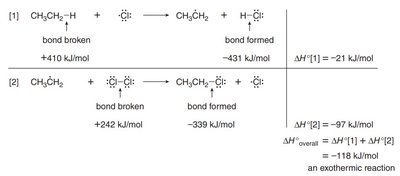
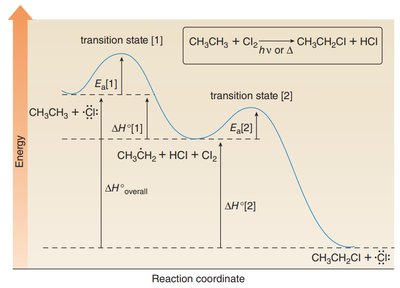
The first propagation step, where the hydrogen atom is abstracted to form a carbon radical, is the rate-determining step (RDS) and is exothermic. The overall reaction is also exothermic, as shown by the enthalpy changes for bond breaking and formation.
Bond Energies: The energy required to break and form bonds determines the reaction's thermodynamics.
Exothermicity: The sum of enthalpy changes for both propagation steps is negative, indicating an exothermic process.
Bond Strength and Reactivity
Effect of C–H Bond Strength on Halogenation
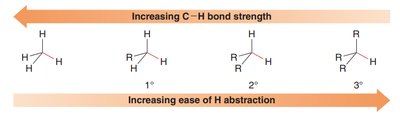
Weaker C–H bonds are more susceptible to radical halogenation. The ease of hydrogen abstraction increases as the bond strength decreases, which correlates with the degree of alkyl substitution.
Bond Strength Trend: 3° < 2° < 1° < methyl
Ease of Abstraction: Tertiary hydrogens are most easily abstracted.
Summary Table: Radical Stability and Reactivity
Comparison of Radical Types
Radical Type | Stability | Ease of H Abstraction |
|---|---|---|
Methyl (Me) | Least stable | Hardest |
Primary (1°) | Low | Hard |
Secondary (2°) | Moderate | Moderate |
Tertiary (3°) | Most stable | Easiest |
Additional info: The notes above expand on the original content by providing definitions, mechanistic details, and academic context for radical reactions, their structure, stability, and halogenation mechanisms. All images included directly reinforce the explanations and are strictly relevant to the adjacent paragraphs.
