 Back
BackStep-by-Step Guidance for Organic Chemistry Reaction Mechanisms and Product Prediction
Study Guide - Smart Notes

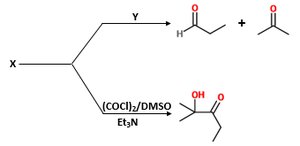
Q1. Identify “X” and “Y” in the following reaction sequence:
Background
Topic: Oxidative Cleavage and Swern Oxidation
This question tests your understanding of oxidative cleavage of diols (often with periodic acid or lead tetraacetate) and the Swern oxidation (using DMSO, oxalyl chloride, and triethylamine) for converting alcohols to carbonyl compounds.
Key Terms and Formulas:
Oxidative cleavage: Splitting a diol into two carbonyl compounds using reagents like HIO4 or Pb(OAc)4.
Swern oxidation: A method to oxidize primary and secondary alcohols to aldehydes and ketones using (COCl)2/DMSO/Et3N.
Step-by-Step Guidance
Examine the products formed in the first reaction. Notice that the molecule is split into two carbonyl compounds: an aldehyde and a ketone.
Recall that such a cleavage is characteristic of oxidative cleavage of a 1,2-diol (glycol) using reagents like HIO4 or Pb(OAc)4.
For the second reaction, recognize the use of (COCl)2/DMSO/Et3N, which is the Swern oxidation, converting a secondary alcohol to a ketone.
Deduce the structure of X: it must be a compound that, upon oxidation, gives the observed β-hydroxy ketone.
Determine the identity of Y: it should be the reagent that cleaves a 1,2-diol to give the observed aldehyde and ketone.
Try solving on your own before revealing the answer!
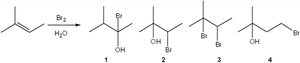
Q2. What is the major organic product obtained from the following reaction?
Background
Topic: Halohydrin Formation
This question tests your knowledge of the reaction between an alkene and bromine in water, which forms a bromohydrin via anti addition.
Key Terms and Formulas:
Bromohydrin formation: Addition of Br2 and H2O to an alkene yields a bromohydrin (a molecule with both Br and OH on adjacent carbons).
Regioselectivity: The OH group attaches to the more substituted carbon due to the stability of the bromonium ion intermediate.
Step-by-Step Guidance
Identify the structure of the starting alkene and note the positions where Br and OH can add.
Recall that the reaction proceeds via formation of a bromonium ion intermediate.
Water attacks the more substituted carbon of the bromonium ion, leading to anti addition of Br and OH.
Draw all possible products and match them to the given choices, paying attention to regiochemistry and stereochemistry.
Try solving on your own before revealing the answer!
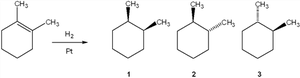
Q3. What is the major organic product obtained from the following reaction?
Background
Topic: Catalytic Hydrogenation of Alkenes
This question tests your understanding of syn addition of hydrogen to an alkene using a platinum catalyst, and how this affects stereochemistry in cyclic systems.
Key Terms and Formulas:
Catalytic hydrogenation: Addition of H2 across a double bond using a metal catalyst (Pt, Pd, or Ni), typically resulting in syn addition.
Syn addition: Both hydrogens add to the same face of the double bond.
Stereochemistry: In cyclic systems, syn addition can lead to cis or trans isomers depending on the substituents.
Step-by-Step Guidance
Identify the position of the double bond and the methyl group on the cyclohexene ring.
Recall that hydrogenation adds two hydrogens to the same face of the double bond (syn addition).
Determine the possible stereoisomers that can result from this addition.
Compare the structures of the products to the answer choices, focusing on the relative positions (cis/trans) of the methyl groups.
Try solving on your own before revealing the answer!
