 Back
Backlecture 5 Stereochemistry: Chirality, Stereoisomers, and Optical Activity
Study Guide - Smart Notes

Stereochemistry
Introduction to Stereochemistry
Stereochemistry is the study of the spatial arrangement of atoms in molecules and its effect on their chemical and physical properties. It is a fundamental concept in organic chemistry, especially in understanding isomerism and the behavior of chiral molecules.
Chirality and Achirality
Chirality: Definition and Analogy
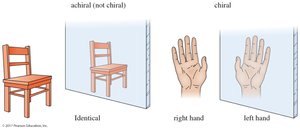
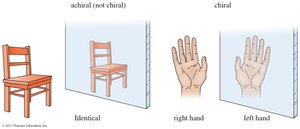
A molecule is chiral if it is not superimposable on its mirror image, much like how a right-hand glove does not fit the left hand. Chirality is often referred to as "handedness." Objects or molecules that are not superimposable on their mirror images are chiral.
Achirality
An achiral object or molecule is one that can be superimposed on its mirror image. For example, a simple chair is achiral, while human hands are chiral.
Stereoisomers
Enantiomers
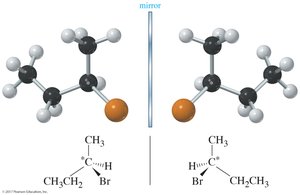
Enantiomers are stereoisomers that are nonsuperimposable mirror images of each other. Any chiral molecule must have an enantiomer. Enantiomers have identical physical properties except for the direction in which they rotate plane-polarized light and their interactions with other chiral substances.
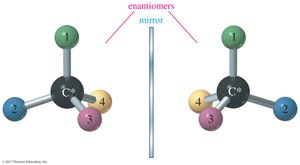
Chiral Centers and Stereocenters
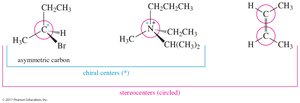
Chiral Carbon Atom (Asymmetric Carbon)
A chiral carbon atom (also called an asymmetric carbon) is a carbon atom bonded to four different groups. Its mirror image will be a different compound (an enantiomer).
Stereocenters
A stereocenter (or stereogenic atom) is any atom at which the interchange of two groups gives a stereoisomer. Asymmetric carbons and the double-bonded carbon atoms in cis-trans isomers are the most common types of stereocenters.
Achiral Compounds and Planes of Symmetry
Achiral Compounds
When the mirror images of a molecule can be superposed, the compound is achiral. This is often due to the presence of a plane of symmetry within the molecule.
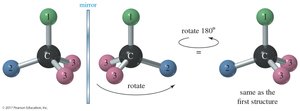
Planes of Symmetry
A molecule that has a plane of symmetry is achiral. The plane of symmetry divides the molecule into two mirror-image halves.
Cis and Trans Cyclic Compounds
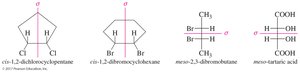
Cis-1,2-dichlorocyclohexane is achiral because it has an internal plane of symmetry.
Trans-1,2-dichlorocyclohexane does not have a plane of symmetry and is chiral, existing as a pair of enantiomers.
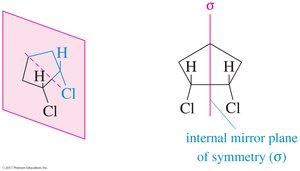
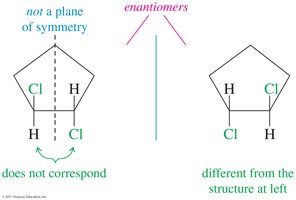
(R) and (S) Configuration: Cahn–Ingold–Prelog Convention
Assigning (R) and (S) Configuration
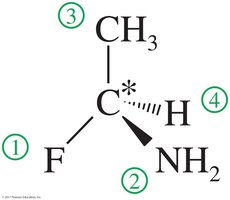
The Cahn–Ingold–Prelog (CIP) convention is used to assign absolute configurations to chiral centers. Each asymmetric carbon atom is assigned a letter, (R) or (S), based on its three-dimensional configuration.
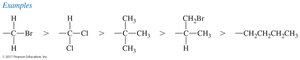
Step 1: Assign Priority – Assign a relative “priority” to each group bonded to the asymmetric carbon based on atomic number (higher atomic number = higher priority).
Breaking Ties – If two atoms have the same atomic number, move to the next atoms along the chain as tiebreakers.
Multiple Bonds – Treat double and triple bonds as if each were a bond to a separate atom.
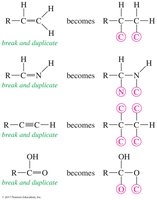
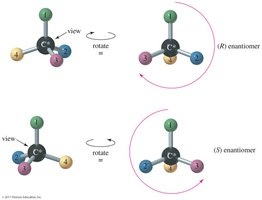
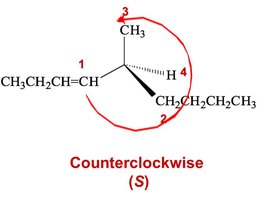
Step 2: Orient the Molecule – Rotate the molecule so that the lowest priority group is in the back. Draw an arrow from the highest to lowest priority group.
Clockwise = (R), Counterclockwise = (S)
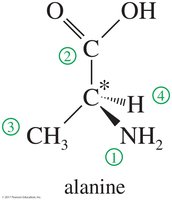
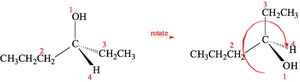
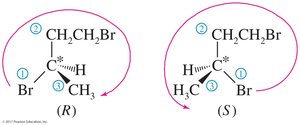
Solved Problem: 1,3-dibromobutane
Draw the enantiomers of 1,3-dibromobutane and label them as (R) and (S). The third carbon atom is asymmetric. Assign priorities and determine the configuration as shown.
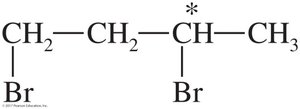
Properties of Enantiomers
Physical and Chemical Properties
Enantiomers have identical boiling points, melting points, and densities.
They rotate plane-polarized light in equal magnitude but opposite directions.
They interact differently with other chiral molecules (e.g., enzymes, taste, and scent receptors).
Optical Activity
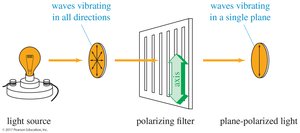
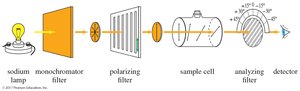
Polarized Light and Polarimeter
Plane-polarized light is composed of waves that vibrate in only one plane. A polarimeter is used to measure the rotation of plane-polarized light by chiral compounds.
Specific Rotation
The specific rotation is calculated using the formula:
where is the observed rotation, is the concentration in g/mL, and is the path length in decimeters.
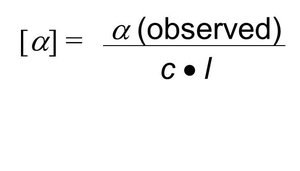
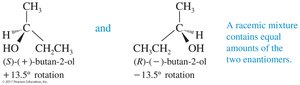
Racemic Mixtures and Enantiomeric Excess
Racemic Mixtures
A racemic mixture contains equal quantities of d- and l-enantiomers. Such mixtures are optically inactive because the rotations cancel each other out. Racemic mixtures may have different boiling and melting points compared to pure enantiomers.
Optical Purity and Enantiomeric Excess
Optical purity (o.p.) or enantiomeric excess (ee) is a measure of how much one enantiomer is present in excess over the racemic mixture. It is calculated as:

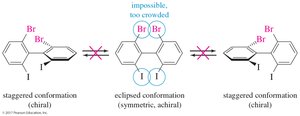
Chirality of Conformers and Conformational Isomers
Chirality of Conformers
If equilibrium exists between two chiral conformers, the molecule is not chiral. Chirality should be judged by the most symmetrical conformer.
Nonmobile Conformers
Some molecules, such as certain biphenyl derivatives, are conformationally locked and can be chiral if their conformers are nonsuperimposable mirror images.
Fischer Projections
Fischer Projection Rules
Fischer projections are flat representations of 3-D molecules, useful for visualizing stereochemistry. The chiral carbon is at the intersection of horizontal and vertical lines. Horizontal lines represent bonds coming out of the plane, and vertical lines represent bonds going behind the plane.
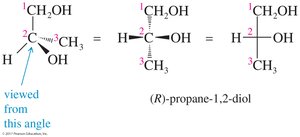
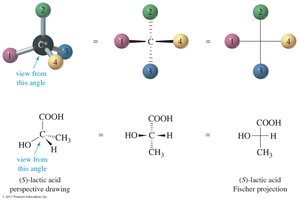
The carbon chain is on the vertical line, with the highest oxidized carbon at the top.
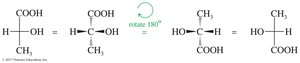
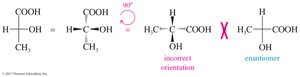
Rotation of 180° in the plane does not change the molecule, but rotation of 90° is not allowed.
Diastereomers
Cis-Trans Isomerism
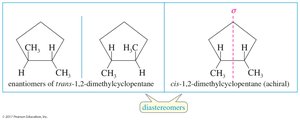
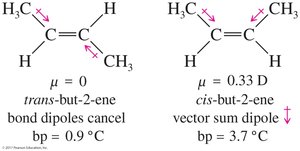
Diastereomers are stereoisomers that are not mirror images of each other. Cis-trans isomers on double bonds or rings are classic examples of diastereomers.
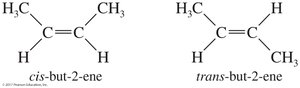
Multiple Chiral Centers
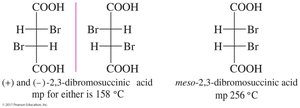
Molecules with two or more chiral centers can have enantiomers, diastereomers, or meso isomers. The maximum number of stereoisomers is , where is the number of chiral centers. Meso compounds have internal mirror planes and are achiral despite having chiral centers.
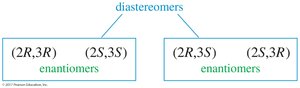
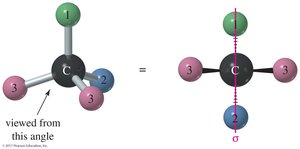
Meso Compounds
Meso compounds have a plane of symmetry and are achiral even though they contain chiral centers. If one image is rotated 180°, it can be superimposed on the other image.
Absolute and Relative Configuration
Definitions
Absolute configuration describes the exact spatial arrangement of atoms in a chiral molecule.
Relative configuration is the experimentally determined relationship between the configurations of two molecules, even if the absolute configuration is unknown.
Separation and Resolution of Enantiomers
Physical and Chemical Resolution
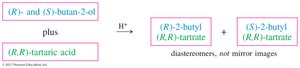
Enantiomers are difficult to separate due to their identical physical properties. Diastereomers, however, have different physical properties and can be separated more easily. Enantiomers can be converted into diastereomers for separation, a process known as resolution.
Historical Note: Louis Pasteur
Louis Pasteur accomplished the first artificial resolution of enantiomers by physically separating mirror-image crystals of racemic tartaric acid.

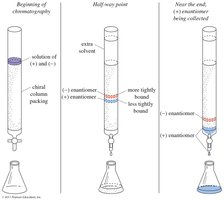
Chemical and Chromatographic Resolution
Reacting a racemic mixture with a pure chiral compound forms diastereomers, which can then be separated. Chromatographic techniques can also be used for resolution.
Summary Table: Types of Isomers
Type | Definition | Example |
|---|---|---|
Enantiomers | Nonsuperimposable mirror images | (R)- and (S)-lactic acid |
Diastereomers | Stereoisomers not related as mirror images | Cis- and trans-2-butene |
Meso Compounds | Achiral compounds with chiral centers and a plane of symmetry | Meso-tartaric acid |
Additional info: This guide covers the core concepts of stereochemistry, including chirality, stereoisomers, enantiomers, diastereomers, meso compounds, and the methods for assigning and resolving configurations. It is suitable for exam preparation in a college-level organic chemistry course.
