 Back
BackStereoisomerism and Chirality: A Comprehensive Study Guide
Study Guide - Smart Notes

Chapter 3: Stereoisomerism and Chirality
Chirality: The Handedness of Molecules
Chirality is a fundamental concept in organic chemistry, describing molecules that are not superposable on their mirror images. This property is crucial for understanding molecular behavior, especially in biological systems.
Chiral Objects: Not superposable on their mirror images.
Achiral Objects: Superposable on their mirror images.
Plane of Symmetry: An imaginary plane dividing a molecule into two mirror-image halves.
Center of Symmetry: A point from which identical components are equidistant and opposite along any axis.
Missing Symmetries: Indicates chirality in molecules.
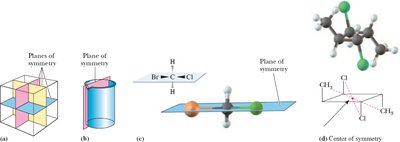
Example: A cube and a cylinder with planes of symmetry, and a molecule with a center of symmetry.
Stereoisomerism
Stereoisomers are compounds with the same molecular formula and connectivity but different spatial arrangements of atoms. This section explores the types and properties of stereoisomers.
Constitutional Isomers: Same formula, different connectivity.
Stereoisomers: Same formula and connectivity, different spatial orientation.
Configurational Isomers: Differ by the configuration of substituents; cannot be interconverted by rotation.
Cis/Trans Isomers: A type of configurational isomerism.
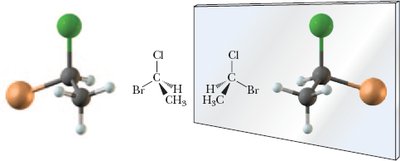
Example: A carbon atom with four different groups (-Cl, -H, -CH3, -Br) forms a chiral center.
Enantiomers and Diastereomers
Enantiomers are stereoisomers that are nonsuperposable mirror images, while diastereomers are not mirror images. These relationships are central to understanding molecular chirality.
Enantiomers: Nonsuperposable mirror images; relationship between pairs.
Diastereomers: Stereoisomers not related as mirror images; can be chiral or achiral.
Chiral Center: Typically a tetrahedral carbon with four different groups.
Multiple Stereocenters: Diastereomers arise when there are two or more stereocenters.
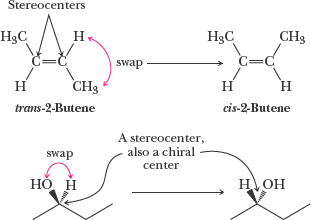
Example: Cis/trans-2-butene and 2-butanol demonstrate configurational isomerism and chirality.
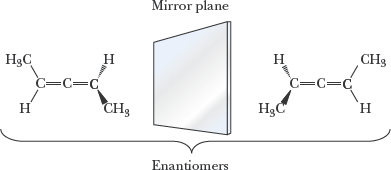
Example: Mirror images of trans-2-butene as enantiomers.
Chirality Beyond Carbon
Chiral centers are not limited to carbon; other elements such as silicon, phosphorus, and germanium can also form chiral centers.
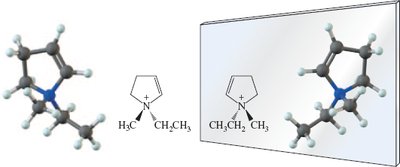
Example: Chiral centers in silicon compounds.
Conformational Isomers
Conformational isomers arise from rotations about single bonds. These isomers interconvert rapidly and are not considered configurational isomers.
Gauche and Anti Forms: Different spatial arrangements in butane.
Rapid Interconversion: Prevents isolation of enantiomers.
Atropisomers: Enantiomers lacking a chiral center, differing due to hindered rotation.

Example: Gauche and anti forms of butane.
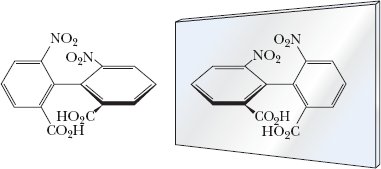
Example: Atropisomers in aromatic systems.
Naming Chiral Centers: The R, S System
The Cahn-Ingold-Prelog (R, S) system assigns absolute configuration to chiral centers based on priority rules.
Absolute Configuration: Specifies which enantiomer is present (right- or left-handed).
R Configuration: Clockwise order of priority.
S Configuration: Counterclockwise order of priority.
Priority Rules:
Assign priority based on atomic number.
If needed, examine atoms further from the chiral center.
Double/triple bonds treated as bonded to "phantom" atoms.
First point of difference determines priority.
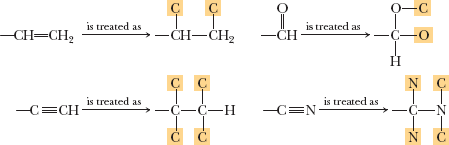
Example: Assigning priorities to groups in molecules with multiple bonds.
Multiple Stereocenters: Enantiomers and Diastereomers
Molecules with two or more chiral centers can have multiple stereoisomers. The number of possible stereoisomers is calculated using the 2n rule, where n is the number of chiral centers.
2,3,4-Trihydroxybutanal: Four stereoisomers possible.
Erythrose: Pair of enantiomers (2R,3R) and (2S,3S).
Threose: Pair of enantiomers (2R,3S) and (2S,3R).
Diastereomers: Non-mirror image stereoisomers.
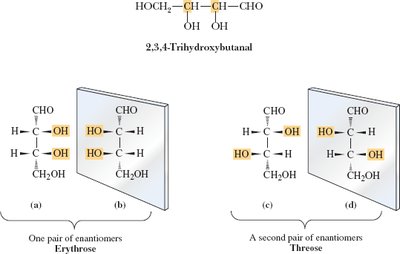
Example: Stereorepresentations of erythrose and threose.
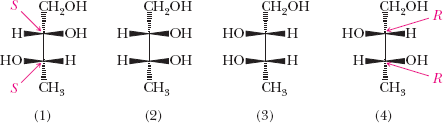
Example: Four stereoisomers of 1,2,3-butanetriol with R/S assignments.
Meso Compounds
Meso compounds are achiral molecules with two or more chiral centers and internal symmetry, reducing the number of stereoisomers.
Tartaric Acid: Has three stereoisomers: a pair of enantiomers and a meso compound.
Physical Properties: Enantiomers have identical properties in achiral environments.
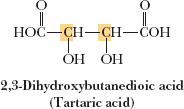
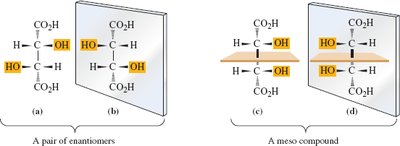
Example: Stereorepresentations of tartaric acid.
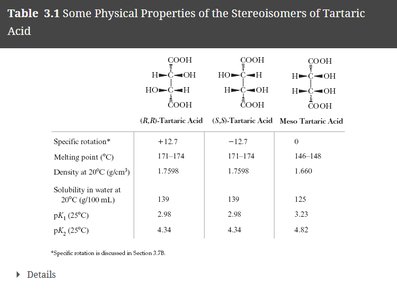
Property | (R,R)-Tartaric Acid | (S,S)-Tartaric Acid | Meso Tartaric Acid |
|---|---|---|---|
Specific rotation | +12.7 | -12.7 | 0 |
Melting point (°C) | 171-174 | 171-174 | 146-148 |
Density at 20°C (g/cm3) | 1.7598 | 1.7598 | 1.660 |
Solubility in water at 20°C (g/100 mL) | 139 | 139 | 125 |
pKa1 (25°C) | 2.98 | 2.98 | 2.98 |
pKa2 (25°C) | 4.34 | 4.34 | 4.82 |
Example: Comparison of physical properties of tartaric acid stereoisomers.
Fischer Projection Formulas
Fischer projections are two-dimensional representations of molecules, useful for visualizing stereochemistry. Groups on the right and left are in front, while those at the top and bottom are at the rear.
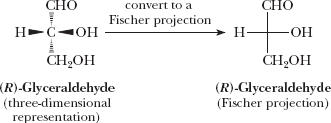
Example: Conversion of three-dimensional structure to Fischer projection.
Cyclic Molecules with Two or More Chiral Centers
Cyclic compounds such as cyclopentane and cyclohexane derivatives can have multiple chiral centers, leading to various stereoisomers.
2-Methylcyclopentanol: Two chiral centers, four possible stereoisomers.
1,2-Cyclopentanediol: Two chiral centers, only three stereoisomers due to meso compound.
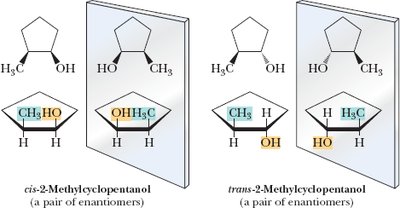
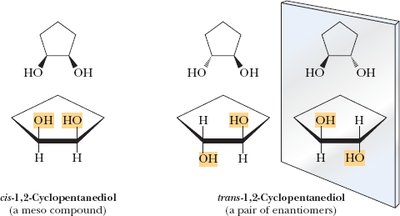
Example: Stereoisomers of cyclopentane derivatives.
Disubstituted Derivatives of Cyclohexane
Cyclohexane derivatives with two substituents can exhibit chirality depending on their spatial arrangement. Chair conformations are important for understanding their stereochemistry.
4-Methylcyclohexanol: Two stereocenters, but not chiral due to symmetry.
3-Methylcyclohexanol: Two chiral centers, four stereoisomers.
1,2-Cyclohexanediol: Three stereoisomers; cis isomer is meso, trans is a pair of enantiomers.
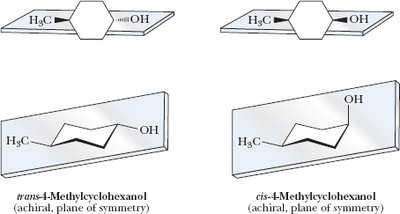
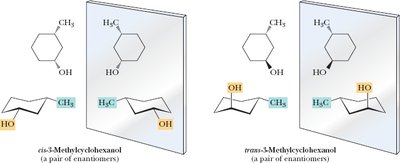
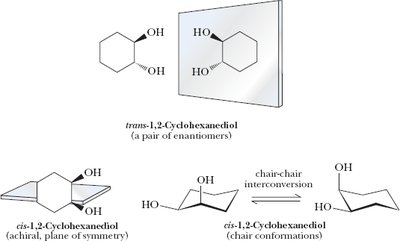
Example: Stereoisomers of cyclohexane derivatives.
Summary Flowchart: Classification of Isomers
A flowchart can help classify isomers based on their connectivity and spatial arrangement, guiding the identification of constitutional, configurational, and conformational isomers, as well as enantiomers, diastereomers, and meso compounds.
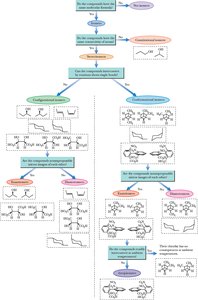
Example: Flowchart for systematic classification of isomers.
Additional info:
Chirality is essential in pharmaceuticals, as enantiomers can have drastically different biological effects.
Fischer projections are especially useful for carbohydrates and amino acids.
Meso compounds are optically inactive despite having chiral centers.
