 Back
BackStereoisomers and Chirality: Study Notes for Organic Chemistry
Study Guide - Smart Notes

Stereoisomers
Constitutional Isomers vs. Stereoisomers
Organic molecules can exhibit isomerism, which is the existence of compounds with the same molecular formula but different structures. Constitutional isomers have the same atoms but differ in how those atoms are connected. Stereoisomers have the same connectivity but differ in the spatial arrangement of their atoms.
Constitutional Isomers: Different connectivity of atoms.
Stereoisomers: Same connectivity, different spatial arrangement.
Geometric Isomers: A type of stereoisomerism arising from restricted bond rotation, commonly seen in cyclic molecules and alkenes (cis/trans isomers).
Chirality
Definition and Properties
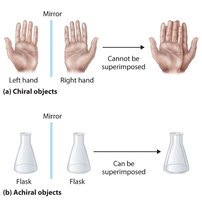
Chirality is a property describing the 'handedness' of an object or molecule. A chiral object is not superimposable on its mirror image, much like left and right hands. In contrast, achiral objects are superimposable on their mirror images.
Chiral: Not superimposable on mirror image (e.g., hands).
Achiral: Superimposable on mirror image (e.g., flasks).
Chirality Centers in Molecules
The most common cause of molecular chirality is the presence of a chirality center (chiral center or asymmetric center): a tetrahedral atom (usually carbon) bonded to four different groups.
If a carbon atom is attached to four distinct groups, it is a chiral center.
Molecules with chiral centers can exist in different configurations.
If a molecule has an internal plane of symmetry, it is achiral.
Enantiomers
Definition and Drawing
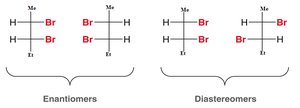
Enantiomers are pairs of stereoisomers that are nonsuperimposable mirror images of each other. Chiral molecules exist as pairs of enantiomers.
To draw an enantiomer, draw the mirror image and invert all chiral centers.
If a molecule has more than one chiral center, invert all centers to get the enantiomer.
Cahn-Ingold-Prelog Nomenclature for Chiral Molecules
Assigning Absolute Configuration (R/S)
Chiral centers are designated as R or S based on the Cahn-Ingold-Prelog priority rules:
Assign priorities to the four groups attached to the chiral center.
Orient the molecule so the lowest priority group is away from you.
Trace from priority 1 to 2 to 3:
If the path is clockwise, the configuration is R.
If the path is counterclockwise, the configuration is S.
Alternative method: If the lowest priority group is toward you, flip the assignment.
Fischer Projections
The Fischer projection is a convenient way to represent chiral centers. In Fischer projections:
Horizontal lines represent groups coming out of the plane (wedges).
Vertical lines represent groups going behind the plane (dashes).
For molecules with multiple chiral centers, the carbon backbone is drawn vertically.
To draw the enantiomer, switch all horizontal groups.
Physical Properties of Enantiomers: Optical Activity
Optical Activity and Polarimetry
Enantiomers share many physical properties, but differ in their interaction with plane-polarized light and other chiral molecules.
Optical Activity: Chiral compounds rotate plane-polarized light. The direction and degree of rotation is called specific rotation.
If light is rotated counterclockwise: Levorotary (l or -).
If light is rotated clockwise: Dextrorotary (d or +).
Specific rotation is not directly related to R/S configuration.
Achiral molecules and racemic mixtures (50:50 enantiomers) are optically inactive.
Diastereomers
Definition and Drawing
Diastereomers are stereoisomers that are not mirror images of each other. Molecules with more than one chiral center can have diastereomers.
To draw a diastereomer, invert one or more (but not all) chiral centers.
Enantiomers: invert all chiral centers.
2n Rule for Stereoisomers
The maximum number of stereoisomers for a molecule with n chiral centers is given by:
Some molecules have fewer stereoisomers due to symmetry (meso compounds).
Meso Compounds
Meso compounds have chiral centers but are achiral due to an internal plane of symmetry. For example, cis-1,2-dimethylcyclobutane is a meso compound.
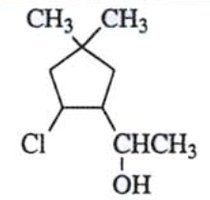
Worked Example: Stereoisomer Counting
How Many Stereoisomers?
To determine the number of stereoisomers for a molecule, count the chiral centers and apply the 2n rule, considering meso forms if present.
Optical Activity of Mixtures
Racemic Mixtures
A 50:50 mixture of two enantiomers is called a racemic mixture. Racemic mixtures are optically inactive because the optical rotations of the enantiomers cancel each other.
Always optically active: Pure chiral compounds.
Always optically inactive: Achiral compounds or racemic mixtures.
Optically active only at high temperature: Some conformationally immobile systems.
Summary Table: Types of Stereoisomers
Type | Mirror Image? | Superimposable? | Optical Activity |
|---|---|---|---|
Enantiomers | Yes | No | Active |
Diastereomers | No | No | Varies |
Meso Compounds | Yes | Yes | Inactive |
Racemic Mixture | Yes | No | Inactive |
Additional info: Academic context was added to clarify the definitions, examples, and applications of stereoisomers, chirality, enantiomers, diastereomers, meso compounds, and optical activity. The images included are directly relevant to the explanation of chirality, stereoisomer types, and stereoisomer counting.
