 Back
BackStudy Notes: Carbonyl Compounds and Their Reactions
Study Guide - Smart Notes

Carbonyl Compounds: Structure and Properties
Structure of Carbonyl Group
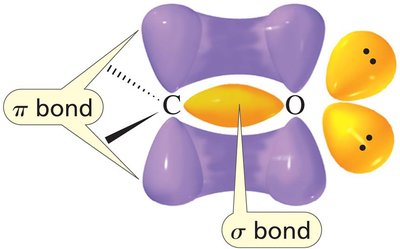
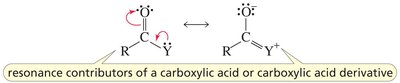
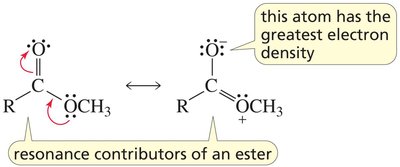
The carbonyl group (C=O) is a fundamental functional group in organic chemistry, present in aldehydes, ketones, carboxylic acids, and their derivatives. The carbonyl carbon is sp2 hybridized, resulting in a planar structure and significant reactivity due to the polarization of the C=O bond.
Key Point: The carbonyl carbon is electrophilic, making it susceptible to nucleophilic attack.
Key Point: The oxygen atom is more electronegative, creating a dipole moment.
Example: The carbonyl group in acetone (CH3COCH3) is highly reactive toward nucleophiles.
Physical Properties of Carbonyl Compounds
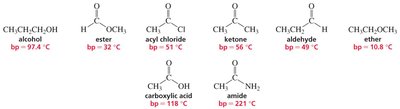
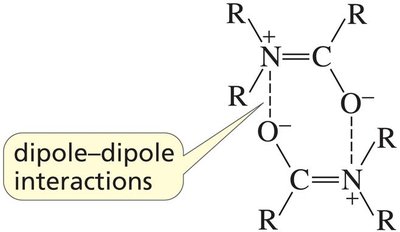
Carbonyl compounds exhibit a range of physical properties, such as boiling points and solubility, depending on their ability to form hydrogen bonds and dipole-dipole interactions.
Key Point: Amides and carboxylic acids have the highest boiling points due to strong hydrogen bonding.
Key Point: Esters, ketones, and aldehydes have lower boiling points, as they lack strong hydrogen bonding.
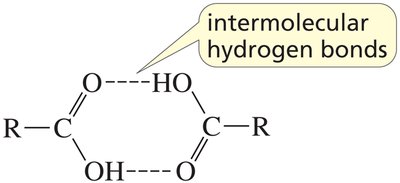
Example: Carboxylic acids can form dimers via intermolecular hydrogen bonds, increasing their boiling points.
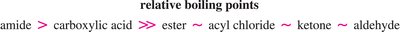
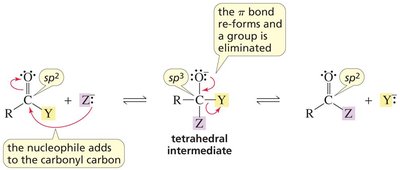
Nucleophilic Acyl Substitution Reactions
General Mechanism
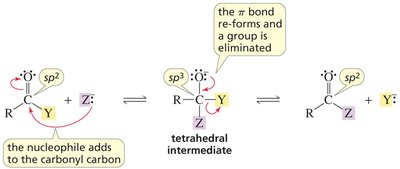
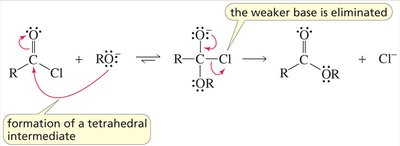
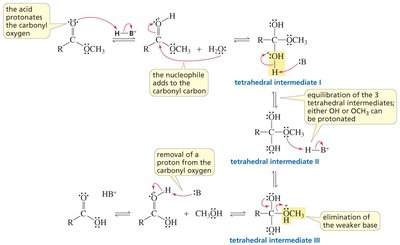
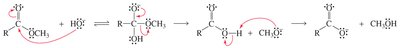
Nucleophilic acyl substitution is a key reaction for carboxylic acid derivatives. The nucleophile attacks the carbonyl carbon, forming a tetrahedral intermediate, which then eliminates a leaving group.
Key Point: The nature of the leaving group determines the product and the reaction's direction.
Key Point: The weakest base is eliminated from the intermediate.
Example: Conversion of acyl chloride to ester by methanol.
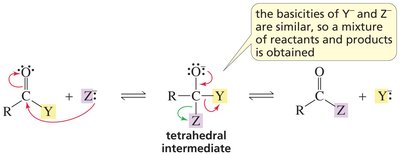
Base Strength and Reaction Outcome
The relative basicity of the incoming nucleophile and the leaving group affects the reaction outcome:
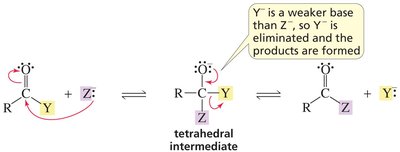
Key Point: If the nucleophile is a stronger base than the leaving group, the product forms.
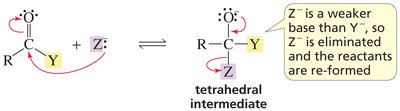
Key Point: If the nucleophile is a weaker base, the reactants are reformed.
Key Point: If both have similar basicity, a mixture results.
Bond Breaking in Nucleophilic Attack
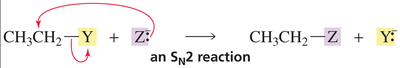
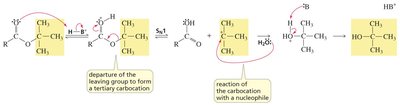
When a nucleophile attacks a carbonyl compound, the pi bond breaks, forming a tetrahedral intermediate. In contrast, nucleophilic attack on alkyl halides breaks the sigma bond.
Key Point: The pi bond in carbonyls is more reactive than the sigma bond in alkyl halides.
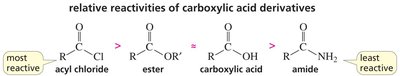
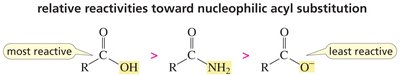
Relative Reactivity and Basicity of Leaving Groups
The reactivity of carboxylic acid derivatives depends on the basicity of the leaving group. Weaker bases are better leaving groups, making the compound more reactive.
Key Point: Acyl chlorides are most reactive; amides are least reactive.
Key Point: The order of leaving group basicity: Cl- < OR- ≈ OH- < NH2-.

Carboxylic Acid Derivatives: Synthesis and Reactions
Acyl (Acid) Chlorides
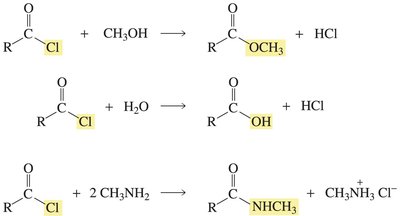
Acyl chlorides are highly reactive carboxylic acid derivatives, easily converted to esters, amides, and other derivatives via nucleophilic acyl substitution.
Key Point: Acyl chlorides react with alcohols, water, and amines to form esters, carboxylic acids, and amides, respectively.
Example: Acetyl chloride reacts with methanol to form methyl acetate.
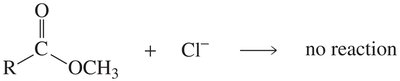
Carboxylic Acids and Esters
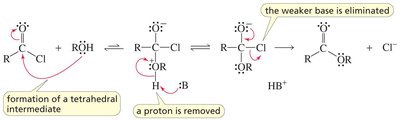
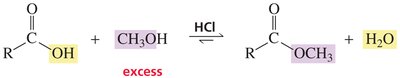
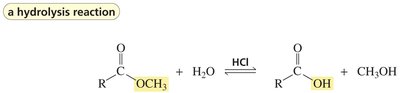
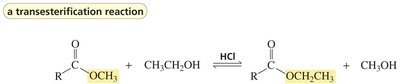
Carboxylic acids can react with alcohols to form esters via Fischer esterification, a reversible acid-catalyzed process. Esters can be hydrolyzed back to acids and alcohols.
Key Point: Acid-catalyzed ester hydrolysis is reversible; base-promoted hydrolysis is irreversible.
Example: Hydrolysis of methyl acetate yields acetic acid and methanol.
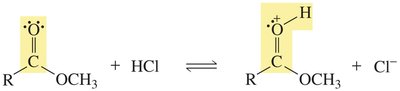

Aminolysis of Esters
Esters react with amines in aminolysis reactions to form amides and alcohols. This is an important method for synthesizing amides.
Key Point: Aminolysis requires heating and often proceeds via nucleophilic acyl substitution.
Example: Ethyl acetate reacts with methylamine to form N-methylacetamide and ethanol.
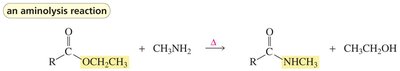
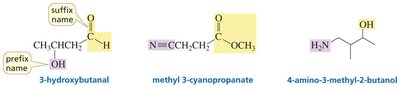
Naming Carbonyl Compounds
Functional Group Priority
Carbonyl-containing functional groups are ranked by priority for nomenclature. Carboxylic acids have the highest priority, followed by esters, acid halides, amides, nitriles, aldehydes, and ketones.
Key Point: The suffix and prefix names depend on the functional group present.
Example: An aldehyde is named with the suffix "-al" and the prefix "formyl" or "oxo".
Priority | Class | Suffix Name | Prefix Name |
|---|---|---|---|
1 | Carboxylic acid | -oic acid | Carboxy |
2 | Ester | -oate | Alkoxycarbonyl |
3 | Acid halide | -oyl halide | |
4 | Amide | -amide | Amido |
5 | Nitrile | -nitrile | Cyano |
6 | Aldehyde | -al | Formyl/Oxo |
7 | Ketone | -one | Oxo |
8 | Alcohol | -ol | Hydroxy |
9 | Amine | -amine | Amino |
10 | Alkene | -ene | Alkenyl |
11 | Alkyne | -yne | Alkynyl |
12 | Alkane | -ane | Alkyl |
13 | Ether | n/a | Alkoxy |
14 | Alkyl halide | n/a | Halo |
Naming Compounds with Multiple Functional Groups
When a compound contains two or more functional groups, the group with the highest priority determines the suffix, while others are named as prefixes.
Key Point: The IUPAC system ensures systematic naming for clarity.
Example: 3-hydroxybutanal (aldehyde and alcohol), methyl 3-cyanopropanoate (ester and nitrile).
Summary Table: Reactivity and Properties of Carboxylic Acid Derivatives
Derivative | Leaving Group | Relative Reactivity | Relative Boiling Point |
|---|---|---|---|
Acyl chloride | Cl- | Most reactive | Low |
Ester | OR- | Moderate | Moderate |
Carboxylic acid | OH- | Moderate | High |
Amide | NH2- | Least reactive | Highest |

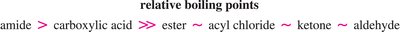

Key Spectroscopic Feature: IR Spectroscopy of Carbonyls
IR Absorption of Carbonyl Group
Carbonyl compounds exhibit a strong IR absorption near 1700 cm-1, which is diagnostic for the C=O bond.
Key Point: A sharp spike at 1700 cm-1 in IR spectra indicates the presence of a carbonyl group.
Example: IR spectrum of acetone shows a strong absorption at 1700 cm-1.

Summary
Carbonyl compounds are central to organic chemistry, with diverse reactivity and properties. Understanding their structure, nomenclature, and reaction mechanisms is essential for mastering organic synthesis and analysis.
