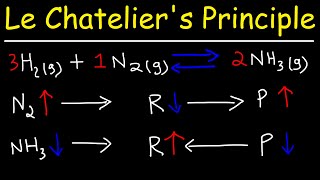
16. Chemical Equilibrium
Le Chatelier's Principle

Get help from an AI Tutor
Ask a question to get started.
Problem 77
Textbook Question
Textbook QuestionConsider the hypothetical reaction A1g2 Δ 2 B1g2. A flask is charged with 0.75 atm of pure A, after which it is allowed to reach equilibrium at 0 C. At equilibrium, the partial pressure of A is 0.36 atm. (c) What could we do to maximize the yield of B?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
336
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos