6. Chemical Quantities & Aqueous Reactions
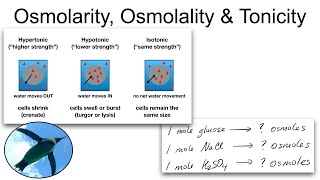
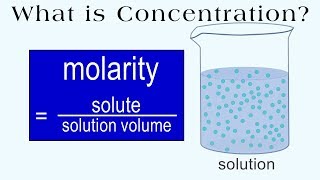
Molarity

Get help from an AI Tutor
Ask a question to get started.
Problem 57a
Textbook Question
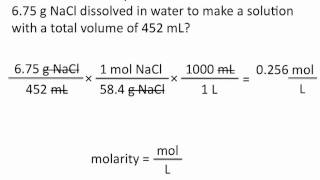
Textbook QuestionBrass is a substitutional alloy consisting of a solution of copper and zinc. A particular sample of red brass consisting of 80.0 % Cu and 20.0 % Zn by mass has a density of 8750 kg>m3. (b) What is the molarity of Zn in the solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
770
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos