6. Chemical Quantities & Aqueous Reactions
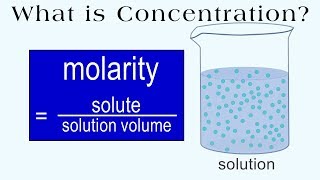
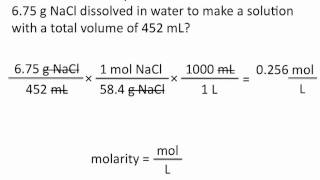
Molarity

Get help from an AI Tutor
Ask a question to get started.
Problem 101b
Textbook Question
Textbook QuestionHard water contains Ca2+, Mg2+, and Fe2+, which interfere with the action of soap and leave an insoluble coating on the insides of containers and pipes when heated. Water softeners replace these ions with Na+. Keep in mind that charge balance must be maintained. (a) If 1500 L of hard water contains 0.020 M Ca2+ and 0.0040 M Mg2+, how many moles of Na+ are needed to replace these ions?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1250
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos