 Back
BackAtoms, Elements, Chemical Reactions, and Molecular Structure: General Chemistry Study Guide
Study Guide - Smart Notes

Atoms & Elements
Atomic Structure
Atoms are the fundamental building blocks of matter, composed of subatomic particles: protons, neutrons, and electrons. Understanding atomic structure is essential for grasping chemical properties and reactions.
Atoms: Smallest unit of matter, indivisible by chemical means.
Subatomic Particles:
Protons: Positive charge, mass ≈ 1 amu.
Neutrons: Neutral charge, mass ≈ 1 amu.
Electrons: Negative charge, mass ≈ 0.0005 amu.
Atomic Number (Z): Number of protons in the nucleus; defines the element.
Mass Number (A): Sum of protons and neutrons.
Isotopes: Atoms of the same element with different numbers of neutrons.
Example: Carbon-12 and Carbon-13 are isotopes of carbon.

Valence Electrons
Valence electrons are the outermost electrons involved in chemical bonding. Their configuration determines an atom's reactivity.
Valence Electrons: Highest energy electrons in the outer shell.
Core Electrons: Electrons in inner shells, not involved in bonding.

Symbols and Notation
Chemical symbols and notation are used to represent elements and their atomic structure.
Element Symbol: One or two letters (e.g., C for carbon).
Notation: , where X = element symbol, Z = atomic number, A = mass number.
Atoms and Subatomic Particles
Changing Subatomic Particles
Changing the number of protons, neutrons, or electrons alters the identity or properties of an atom.
Changing Protons: Results in a new element.
Changing Neutrons: Creates isotopes.
Changing Electrons: Forms ions.
Atomic Mass and Isotopes
Atomic mass is the weighted average of isotopes based on their natural abundance.
Molecular Mass: Sum of masses of all atoms in a molecule.
Atomic Weight: Weighted average of isotopic masses.
Chemical Reactions
Types of Chemical Reactions
Chemical reactions involve the rearrangement of atoms to form new substances. There are several main types:
Synthesis: Two or more atoms combine to form a single compound.
Decomposition: Compound breaks down into two or more products.
Single Displacement: One element replaces another.
Double Displacement: Exchange of ions between compounds.
Combustion: Substance reacts with O2 to produce energy.
Physical vs. Chemical Changes
Physical changes alter the state or appearance of matter without changing its composition, while chemical changes result in new substances.
Physical Change: Melting, boiling, dissolving.
Chemical Change: Burning, rusting, reacting.
Quantum Theory and Electron Configuration
Quantum Numbers
Quantum numbers describe the properties of atomic orbitals and electrons.
Principal Quantum Number (n): Energy level, n = 1, 2, 3, ...
Azimuthal Quantum Number (l): Shape of orbital, l = 0 (s), 1 (p), 2 (d), 3 (f).
Magnetic Quantum Number (ml): Orientation in space, ml = -l to +l.
Spin Quantum Number (ms): Electron spin, ms = +1/2 or -1/2.

Electron Configuration
Electron configuration describes the arrangement of electrons in an atom's orbitals.
Aufbau Principle: Electrons fill lowest energy orbitals first.
Pauli Exclusion Principle: No two electrons in an atom can have the same set of quantum numbers.
Hund's Rule: Electrons occupy orbitals singly before pairing.
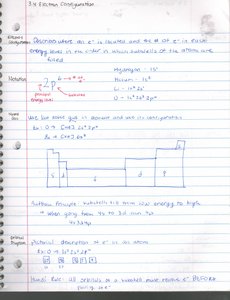
Bonding & Molecular Structure
Ionic and Covalent Bonds
Atoms form bonds to achieve stable electron configurations. Ionic bonds involve electron transfer, while covalent bonds involve electron sharing.
Ionic Bonds: Formed between metals and nonmetals; electrons are transferred.
Covalent Bonds: Formed between nonmetals; electrons are shared.
Bond Types Theory
Bond strength, polarity, and distribution of charges affect molecular properties.
Bond Energy: Energy required to break a bond.
Polarity: Unequal sharing of electrons creates dipoles.
Intermolecular Forces:
London Dispersion: Weak, temporary dipoles.
Dipole-Dipole: Attraction between polar molecules.
Hydrogen Bond: Strong dipole-dipole involving H bonded to N, O, or F.
Ion-Dipole: Attraction between ions and polar molecules.
Molecular Shapes & Valence Bond Theory
VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) theory predicts molecular geometry based on electron domain repulsion.
Linear: 2 electron domains, bond angle 180°.
Trigonal Planar: 3 electron domains, bond angle 120°.
Tetrahedral: 4 electron domains, bond angle 109.5°.
Trigonal Bipyramidal: 5 electron domains, bond angles 90°, 120°.
Octahedral: 6 electron domains, bond angle 90°.

Hybridization
Hybridization explains the mixing of atomic orbitals to form new, equivalent orbitals for bonding.
sp: Linear geometry.
sp2: Trigonal planar geometry.
sp3: Tetrahedral geometry.

Lewis Dot Structures
Drawing Lewis Structures
Lewis dot structures represent valence electrons and help predict molecular bonding and shape.
Octet Rule: Atoms tend to have eight electrons in their valence shell.
Formal Charge: Used to determine the most stable structure.

Periodic Properties
Periodic Trends
The periodic table organizes elements by increasing atomic number and reveals trends in properties.
Atomic Radius: Increases down a group, decreases across a period.
Ionization Energy: Increases across a period, decreases down a group.
Electronegativity: Increases across a period, decreases down a group.

Additional info:
Notes cover core general chemistry topics: atomic structure, chemical reactions, quantum theory, bonding, molecular shapes, and periodic properties.
Images included are directly relevant to the explanation and reinforce key concepts.
