 Back
BackAtoms, Molecules, and Ions: Structured Study Notes
Study Guide - Smart Notes

Atoms, Molecules, and Ions
Atomic Theory of Matter
The concept of the atom as the fundamental building block of matter originated with ancient Greek philosophers and was formalized by John Dalton in the early 1800s. Dalton's atomic theory was based on several key laws:
Law of Constant Composition: Compounds have a definite composition; the relative number of atoms of each element in a compound is the same in any sample.
Law of Conservation of Mass: The total mass of substances present before and after a chemical process remains unchanged.
Law of Multiple Proportions: If two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers.
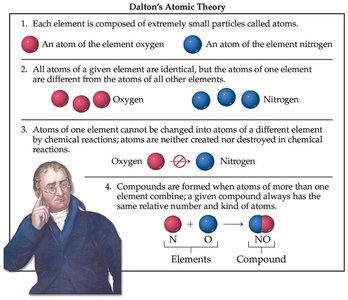
Dalton's Atomic Theory consists of four postulates:
1) Each element is composed of extremely small particles called atoms.
2) All atoms of a given element are identical in mass and properties, but differ from atoms of other elements.
3) Atoms are not changed into atoms of another element by chemical reactions; they are neither created nor destroyed.
4) Atoms of different elements combine to form compounds; a given compound always has the same relative number and kind of atoms.
Discovery of Subatomic Particles
Dalton considered atoms indivisible, but later experiments revealed that atoms are composed of smaller particles:
Electrons: Discovered via cathode ray experiments by J. J. Thomson.
Protons and Neutrons: Found in the nucleus, discovered through studies of radioactivity and nuclear scattering.
Radioactivity: Spontaneous emission of radiation, observed by Henri Becquerel and studied by Marie and Pierre Curie.

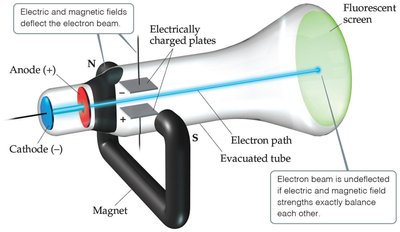
Cathode Rays and the Electron
Cathode rays are streams of negatively charged particles (electrons) observed in vacuum tubes. Thomson measured the charge-to-mass ratio of the electron:
Charge/mass ratio: C/g

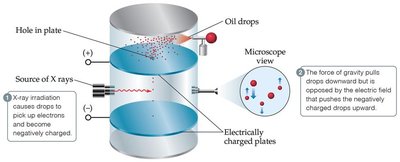
Millikan Oil-Drop Experiment
Robert Millikan determined the charge of the electron ( C) using the oil-drop experiment, allowing calculation of the electron's mass.
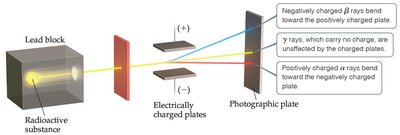
Radioactivity
Radioactivity is the spontaneous emission of high-energy radiation by atoms. Three types of radiation were discovered:
Alpha (α) particles: Positively charged
Beta (β) particles: Negatively charged
Gamma (γ) rays: Uncharged

Models of the Atom
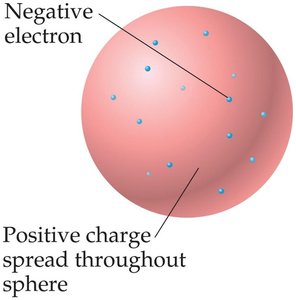
The "plum pudding" model proposed by Thomson depicted the atom as a sphere of positive charge with embedded electrons.
Discovery of the Nucleus
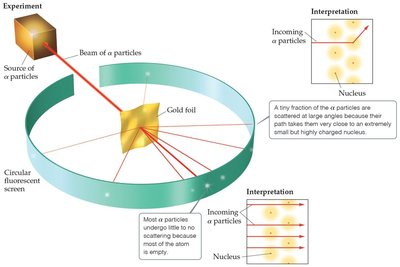
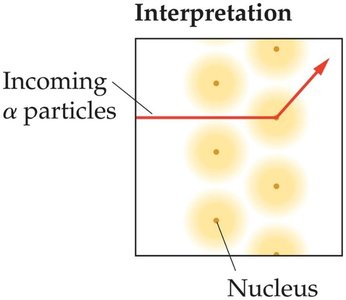
Ernest Rutherford's gold foil experiment showed that atoms have a small, dense, positively charged nucleus, disproving the plum pudding model.
Modern View of Atomic Structure
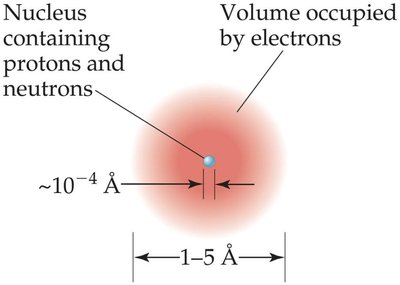
Atoms consist of a tiny nucleus containing protons and neutrons, surrounded by electrons occupying most of the atom's volume. Most of the atom is empty space.
Atomic size: 1–5 Å (100–500 pm)
Nucleus size: Å
Subatomic Particles
Protons and neutrons reside in the nucleus; electrons orbit the nucleus. Their properties are summarized below:
Particle | Charge | Mass (amu) |
|---|---|---|
Proton | +1 | 1.0073 |
Neutron | 0 | 1.0087 |
Electron | -1 | 5.486 × 10-4 |
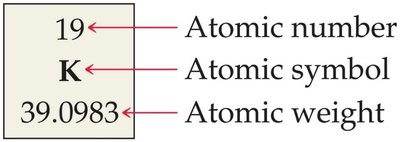
Atomic Number and Mass Number
The atomic number (Z) is the number of protons in the nucleus and defines the element. The mass number (A) is the sum of protons and neutrons.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons. For example, carbon has isotopes with mass numbers 11, 12, 13, and 14.
Symbol | Protons | Electrons | Neutrons |
|---|---|---|---|
11C | 6 | 6 | 5 |
12C | 6 | 6 | 6 |
13C | 6 | 6 | 7 |
14C | 6 | 6 | 8 |
Atomic Mass Unit (amu) and Atomic Weight
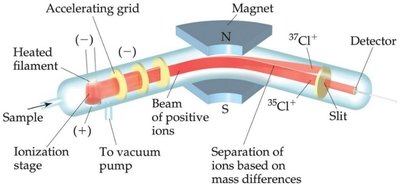
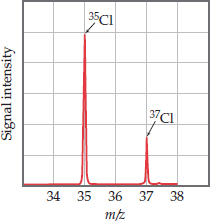
Atomic masses are measured in atomic mass units (amu), where 1 amu = g. Atomic weight is the weighted average of the masses of all isotopes of an element.
Atomic Weight Calculation
The atomic weight is calculated as:
The Periodic Table
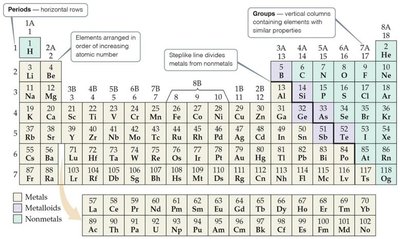
The periodic table organizes elements by atomic number. Rows are called periods, columns are groups. Elements in the same group have similar properties.
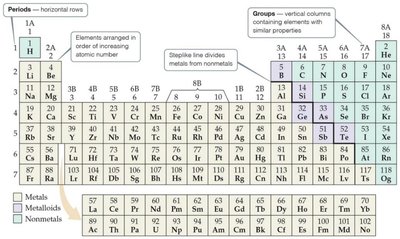
Periodicity and Groups
Elements in a row (period) show repeating patterns of properties. Groups (columns) are named, such as alkali metals (1A), alkaline earth metals (2A), halogens (7A), and noble gases (8A).
Group | Name | Elements |
|---|---|---|
1A | Alkali metals | Li, Na, K, Rb, Cs, Fr |
2A | Alkaline earth metals | Be, Mg, Ca, Sr, Ba, Ra |
6A | Chalcogens | O, S, Se, Te, Po |
7A | Halogens | F, Cl, Br, I, At |
8A | Noble gases | He, Ne, Ar, Kr, Xe, Rn |
Classification of Elements
Metals are on the left, nonmetals on the right, and metalloids along the steplike line. Metals are shiny, conduct heat and electricity, and are mostly solids. Nonmetals can be solid, liquid, or gas.


Molecules and Molecular Compounds
Chemical formulas indicate the number of atoms of each element in a molecule. Molecular compounds are composed of molecules and usually contain only nonmetals.
Diatomic Molecules
Seven elements naturally occur as diatomic molecules: H2, N2, O2, F2, Cl2, Br2, I2.
Types of Formulas
Empirical formula: Lowest whole-number ratio of atoms.
Molecular formula: Exact number of atoms of each element.
Structural formula: Shows how atoms are connected.
Ions and Ionic Compounds
Atoms or groups of atoms that gain or lose electrons become ions. Cations (positive) are formed by metals; anions (negative) by nonmetals. Ionic compounds are formed between metals and nonmetals.
Naming Inorganic Compounds
Chemical nomenclature includes rules for naming ions, ionic compounds, acids, and binary molecular compounds. Cations are named first, followed by anions. Polyatomic ions have specific names.
Common Cations and Anions
Charge | Formula | Name |
|---|---|---|
+1 | Na+ | Sodium ion |
+2 | Mg2+ | Magnesium ion |
-1 | Cl- | Chloride ion |
-2 | SO42- | Sulfate ion |
Polyatomic Ions
Groups of atoms that gain or lose electrons are polyatomic ions, such as ammonium (NH4+) and sulfate (SO42-).
Writing Inorganic Formulas
Formulas are written so that the compound is electrically neutral. The charge of the cation becomes the subscript of the anion and vice versa.
Patterns in Oxyanion Nomenclature
Oxyanions with fewer oxygens end in -ite; those with more end in -ate. Prefixes hypo- and per- are used for the least and most oxygens, respectively.
Inorganic Acid Nomenclature
Acids are named based on their anions: -ide becomes hydro-...-ic acid, -ite becomes -ous acid, and -ate becomes -ic acid.
Nomenclature of Binary Molecular Compounds
The element farther left or lower in the periodic table is named first. Prefixes denote the number of atoms (mono-, di-, tri-, etc.), and the second element ends in -ide.
Some Simple Organic Compounds
Organic chemistry studies carbon compounds. The simplest hydrocarbons are alkanes, named by the number of carbons (meth-, eth-, prop-, etc.) followed by -ane. Alcohols are named by replacing a hydrogen with an -OH group and ending with -ol.
Nomenclature Isomers: Alcohols
Isomers have the same chemical formula but different structures. For example, 1-propanol and 2-propanol differ in the position of the -OH group.
