 Back
BackAtoms, Quantum Theory, and Periodicity: Foundations of General Chemistry
Study Guide - Smart Notes

Atoms and the Nuclear Model
The Structure of the Atom
The modern atomic model describes atoms as consisting of a dense, positively charged nucleus containing protons (and neutrons), with negatively charged electrons distributed around the nucleus. The atomic number (Z) is defined as the number of protons in the nucleus and determines the identity of the element.
Nucleus: Contains protons and neutrons; nearly all the atom's mass is concentrated here.
Electrons: Surround the nucleus in regions of space called orbitals.
Atomic Number (Z): Number of protons in the nucleus; unique for each element.
Electromagnetic Radiation and Light
Nature and Properties of Light
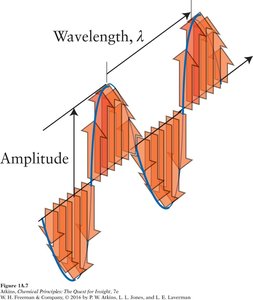
Light is a form of electromagnetic radiation, which consists of oscillating electric and magnetic fields that propagate through space. All electromagnetic radiation transfers energy and can be described by its wavelength (λ), frequency (ν), and amplitude.
Wavelength (λ): The distance between successive peaks of a wave (measured in meters, nm, etc.).
Frequency (ν): The number of wave cycles per second (measured in Hertz, Hz).
Amplitude: The height of the wave, related to the intensity of the radiation.
Speed of Light (c): All electromagnetic waves travel at the same speed in a vacuum: .
Relationship:
The Electromagnetic Spectrum
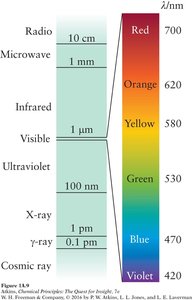
The electromagnetic spectrum includes all types of electromagnetic radiation, from radio waves to gamma rays. The visible spectrum is only a small part, with colors corresponding to specific wavelengths and frequencies.
Visible Light: Wavelengths from about 400 nm (violet) to 700 nm (red).
Color and Wavelength: The color of visible light depends on its wavelength and frequency.
Atomic Spectra and the Hydrogen Atom
Atomic Emission and Absorption Spectra
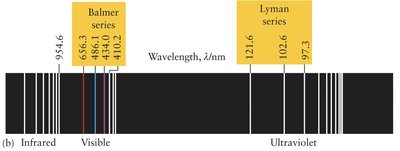
When atoms absorb energy, electrons are excited to higher energy levels. As they return to lower energy levels, they emit light at specific wavelengths, producing a line spectrum unique to each element. For hydrogen, these lines can be described by the Rydberg formula.
Emission Spectrum: Bright lines on a dark background, produced when excited electrons fall to lower energy levels.
Absorption Spectrum: Dark lines on a continuous spectrum, produced when electrons absorb specific wavelengths and are excited to higher levels.
Rydberg Formula: Describes the wavelengths of spectral lines in hydrogen: , where is the Rydberg constant, .
Quantum Theory and the Nature of Light
Quanta, Photons, and the Photoelectric Effect
Max Planck proposed that energy is exchanged in discrete packets called quanta. Albert Einstein extended this idea, describing light as consisting of particles called photons. The energy of a photon is proportional to its frequency:
Photon Energy: , where is Planck's constant ().
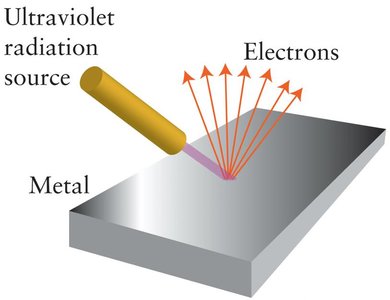
Photoelectric Effect: The ejection of electrons from a metal surface when exposed to light of sufficient frequency, demonstrating the particle nature of light.
Wave-Particle Duality
Light exhibits both wave-like and particle-like properties. Diffraction and interference patterns demonstrate its wave nature, while the photoelectric effect demonstrates its particle nature. Louis de Broglie proposed that matter (such as electrons) also exhibits wave-like properties, with a wavelength given by:
de Broglie Wavelength: , where is mass and is velocity.

The Uncertainty Principle
Werner Heisenberg's uncertainty principle states that it is impossible to simultaneously know both the exact position and momentum of a particle. This principle is fundamental to quantum mechanics and limits our ability to describe electrons as having precise orbits.
Uncertainty Principle:
Wavefunctions and Quantum Numbers
The Schrödinger Equation and Atomic Orbitals
Erwin Schrödinger developed a mathematical equation whose solutions, called wavefunctions (Ψ), describe the probability of finding an electron in a particular region of space. The square of the wavefunction, , gives the probability density. Nodes are regions where the probability density is zero.
Wavefunction (Ψ): Mathematical function describing the electron's behavior.
Probability Density: gives the likelihood of finding an electron at a specific location.
Node: A point or plane where and the probability density is zero.
Quantum Numbers
Each electron in an atom is described by a set of four quantum numbers:
Principal Quantum Number (n): Indicates the main energy level (shell);
Angular Momentum Quantum Number (l): Indicates the subshell (shape of orbital);
Magnetic Quantum Number (m_l): Indicates the orientation of the orbital;
Spin Quantum Number (m_s): Indicates the spin of the electron; or
Atomic Orbitals and Their Shapes
Atomic orbitals are regions in space where electrons are likely to be found. The shapes and orientations of orbitals are determined by the quantum numbers:
s orbitals (l = 0): Spherical shape
p orbitals (l = 1): Dumbbell shape, three orientations (p_x, p_y, p_z)
d orbitals (l = 2): More complex shapes, five orientations
f orbitals (l = 3): Even more complex, seven orientations
The number of orbitals in a shell is , and the maximum number of electrons in a shell is .
Many-Electron Atoms and Electron Configuration
Orbital Energies and the Aufbau Principle
In many-electron atoms, electrons experience both attraction to the nucleus and repulsion from other electrons. The effective nuclear charge (Z_eff) is less than the actual nuclear charge due to shielding. Electrons fill orbitals in order of increasing energy, following the Aufbau (building-up) principle.
Pauli Exclusion Principle: No more than two electrons can occupy the same orbital, and they must have opposite spins.
Hund's Rule: Electrons fill degenerate orbitals singly first, with parallel spins, before pairing up.
Electron Configuration: Notation showing the distribution of electrons among orbitals (e.g., 1s2 2s2 2p6).
Noble Gas Configuration: Shorthand notation using the previous noble gas to represent core electrons.
Periodicity and Trends in the Periodic Table
Structure of the Periodic Table
The periodic table is organized into blocks (s, p, d, f) based on electron configurations. Main group elements are in the s- and p-blocks, transition metals in the d-block, and lanthanides/actinides in the f-block.
Trends in Atomic Properties
Atomic Radius: Increases down a group (due to higher n), decreases across a period (due to increasing Z_eff).
Ionic Radius: Cations are smaller, anions are larger than their parent atoms. Isoelectronic species have different radii depending on nuclear charge.
Ionization Energy (I): Energy required to remove an electron. Increases up a group and across a period. Successive ionization energies increase for each electron removed.
Electron Affinity (Eea): Energy change when an electron is added to a neutral atom. Generally becomes more positive up a group and across a period, with exceptions.
General Properties of the Elements
s-block: Reactive metals, tend to lose electrons.
p-block: Tend to gain electrons, include nonmetals and metalloids.
d-block: Transition metals, often form cations with multiple charges.
Understanding periodic trends allows prediction of chemical and physical properties of elements, such as reactivity, ion formation, and bonding behavior.
