 Back
BackChapter 13: Solutions – Structure, Properties, and Colligative Effects
Study Guide - Smart Notes

Solutions and Their Importance
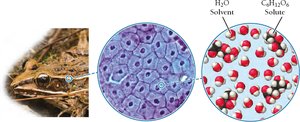
Antifreeze in Frogs: Biological Application of Solutions
Wood frogs survive freezing temperatures by flooding their blood with glucose, forming a concentrated solution that lowers the freezing point of their blood. This adaptation prevents cellular damage caused by ice formation, demonstrating the practical importance of solution chemistry in nature.
Key Point: Concentrated solutions have lower freezing points than pure liquids.
Example: Wood frogs use glucose as an antifreeze to survive winter hibernation.
Fundamentals of Solutions
Definition and Types of Solutions
A solution is a homogeneous mixture of two or more substances. The major component is the solvent, and the minor component is the solute. Solutions can involve different phases, such as solid-liquid, gas-liquid, or gas-gas combinations.
Key Point: Solutions are uniform throughout and can be composed of various phases.
Example: Salt water (solid in liquid), air (gas in gas), soda (gas in liquid).
Solubility and Miscibility
Solubility is the maximum amount of solute that can dissolve in a given amount of solvent. Two liquids that dissolve in each other are miscible; those that do not are immiscible. Solubility depends on temperature, pressure, and the nature of intermolecular forces.
Key Point: "Likes dissolve in likes" – polar substances dissolve in polar solvents, nonpolar in nonpolar.
Example: Alcohol and water are miscible; oil and water are immiscible.
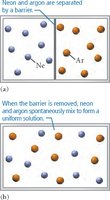
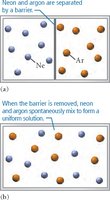
Nature’s Tendency Toward Mixing: Entropy
Spontaneous Mixing and Entropy
Mixing occurs spontaneously due to the increase in entropy, which is the measure of energy dispersal in a system. Even when potential energy changes are negligible, entropy drives the mixing of substances.
Key Point: Entropy increases as substances mix, favoring solution formation.
Example: Mixing of neon and argon gases in a container.
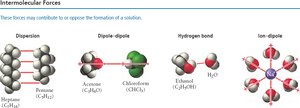
Intermolecular Forces and Solution Formation
Types of Intermolecular Forces
Solution formation depends on the balance of intermolecular forces: dispersion, dipole-dipole, hydrogen bonding, and ion-dipole interactions. The relative strengths of solute-solute, solvent-solvent, and solute-solvent interactions determine whether a solution will form.
Key Point: Energy is required to break solute-solute and solvent-solvent attractions (endothermic), but energy is released when new solute-solvent attractions form (exothermic).
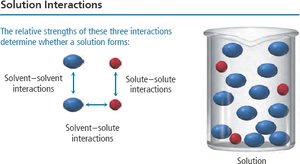
Solubility: Molecular Structure and Polarity
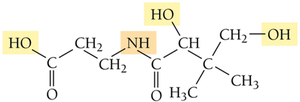
Solubility of Vitamins: Fat-Soluble vs. Water-Soluble
The solubility of vitamins depends on their molecular structure and polarity. Water-soluble vitamins have polar bonds and can hydrogen-bond with water, while fat-soluble vitamins are dominated by nonpolar bonds.
Key Point: Polar molecules are water-soluble; nonpolar molecules are fat-soluble.
Example: Vitamin C is water-soluble due to its polar bonds.
Energetics of Solution Formation
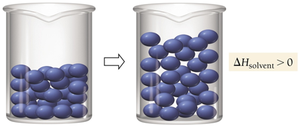
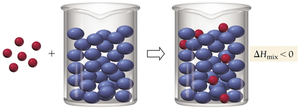
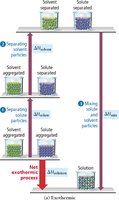
Enthalpy of Solution
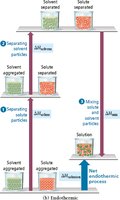
Three steps are involved in solution formation: (1) separating solute particles (endothermic), (2) separating solvent particles (endothermic), and (3) mixing solute and solvent particles (exothermic). The overall enthalpy change () depends on the relative sizes of these energy changes.
Key Point: If the energy released in mixing is greater than the energy required to separate particles, the process is exothermic; otherwise, it is endothermic.
Heats of Hydration
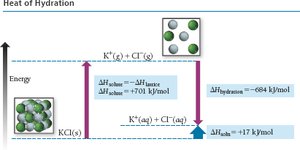
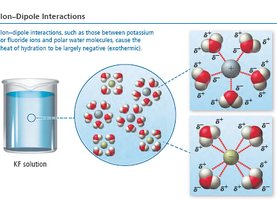
For ionic compounds in water, the heat of hydration combines the energy required to overcome water-water attractions and the energy released in forming ion-water attractions. Lattice energy (ion-ion attraction) and ion-dipole interactions are key.
Key Point: The heat of solution is the difference between the heat of hydration and lattice energy.
Solution Equilibrium and Saturation
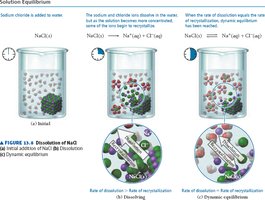
Dynamic Equilibrium and Saturation
When a solution reaches equilibrium, the rate of solute dissolving equals the rate of solute re-forming. A saturated solution contains the maximum amount of dissolved solute; an unsaturated solution can dissolve more solute; a supersaturated solution contains more solute than equilibrium allows.
Key Point: Saturation depends on temperature and pressure.

Purification by Recrystallization
Recrystallization is a method for purifying solids by dissolving them in hot solvent and allowing them to crystallize as the solution cools, leaving impurities behind.
Example: Formation of rock candy from recrystallized sugar.

Temperature and Pressure Effects on Solubility
Solubility of Solids and Gases
Solubility of solids generally increases with temperature, while solubility of gases decreases with temperature. The solubility of gases increases with pressure, described by Henry’s Law:
Henry’s Law:
Key Point: Higher partial pressure increases gas solubility.
Expressing Solution Concentration
Concentration Units
Several units are used to express concentration:
Molarity (M): Moles of solute per liter of solution ()
Molality (m): Moles of solute per kilogram of solvent ()
Percent by mass:
Parts per million (ppm):
Mole fraction ():
Colligative Properties
Vapor Pressure Lowering
The vapor pressure of a solvent is lowered when a nonvolatile solute is added. Raoult’s Law describes this effect:
Raoult’s Law:
Key Point: The vapor pressure of the solution is proportional to the mole fraction of the solvent.
Deviations from Raoult’s Law
Non-ideal solutions show deviations from Raoult’s Law when solute-solvent interactions are stronger or weaker than the sum of solute-solute and solvent-solvent interactions.
Freezing Point Depression and Boiling Point Elevation
Adding solute lowers the freezing point and raises the boiling point of a solvent. The changes are proportional to the molal concentration of solute particles:
Freezing Point Depression:
Boiling Point Elevation:
Osmosis and Osmotic Pressure
Osmosis
Osmosis is the flow of solvent from a region of low solute concentration to high solute concentration through a semipermeable membrane. The pressure required to stop this flow is the osmotic pressure ():
Osmotic Pressure Equation:
Van’t Hoff Factor and Electrolyte Solutions
Van’t Hoff Factor (i)
The van’t Hoff factor () accounts for the number of particles produced by dissociation of ionic compounds. It affects colligative properties:
Key Point:
Colligative Properties and Medical Solutions
Osmotic Effects on Cells
Medical solutions are classified as isosmotic, hyperosmotic, or hyposmotic relative to cell contents. Isosmotic solutions cause no net water flow; hyperosmotic solutions cause cells to shrivel; hyposmotic solutions cause cells to swell.
Key Point: Osmotic pressure differences drive water movement across cell membranes.
Summary Table: Types of Intermolecular Forces
Type | Example | Relative Strength |
|---|---|---|
Dispersion | Pentane (C5H12) | Weak |
Dipole-dipole | Acetone (C3H6O) | Moderate |
Hydrogen bond | Water (H2O) | Strong |
Ion-dipole | Na+ in water | Very strong |
Summary Table: Colligative Properties
Property | Effect of Solute | Equation |
|---|---|---|
Vapor Pressure Lowering | Decreases | |
Freezing Point Depression | Lowers | |
Boiling Point Elevation | Raises | |
Osmotic Pressure | Increases |
