 Back
BackChapter 17: Acids and Bases – Properties, Strength, and Reactions
Study Guide - Smart Notes

Acids and Bases
Properties of Acids
Acids are a fundamental class of compounds in chemistry, characterized by their ability to donate protons (H+) and exhibit distinctive physical and chemical properties.
Sour taste: Many acids, such as citric acid in lemons, have a sour flavor.
Ability to dissolve many metals: Acids can react with metals to produce hydrogen gas.
Ability to neutralize bases: Acids react with bases to form water and a salt.
Litmus test: Acids turn blue litmus paper red.
Types of Acids
Acids can be classified based on their composition and structure:
Binary acids: Composed of hydrogen and one other nonmetal element (e.g., HCl).
Oxyacids: Contain hydrogen, oxygen, and another element (e.g., H2SO4, HNO3).
Carboxylic acids: Organic acids containing the –COOH group (e.g., acetic acid).

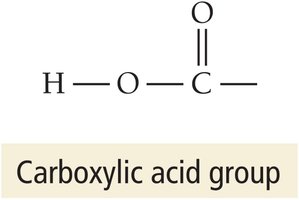
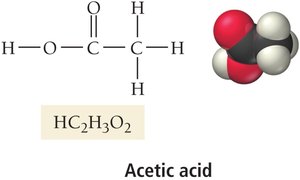
Acidic Hydrogen in Carboxylic Acids
In carboxylic acids, only the hydrogen atom attached to the –COOH group is acidic and can be donated as a proton.
Properties of Bases
Bases are substances that accept protons or donate hydroxide ions (OH-) in solution. They have distinct properties:
Bitter taste: Many bases taste bitter (e.g., caffeine, quinine).
Slippery feel: Bases feel slippery to the touch (e.g., soap).
Litmus test: Bases turn red litmus paper blue.
Neutralize acids: Bases react with acids to form water and a salt.

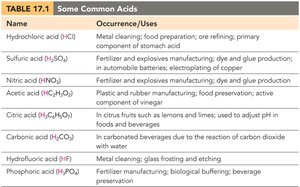
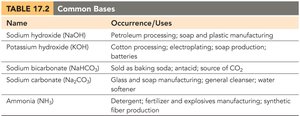
Common Acids and Bases
Acids and bases are widely used in industry and daily life. The following tables summarize some common examples and their uses:
Name | Occurrence/Uses |
|---|---|
Hydrochloric acid (HCl) | Metal cleaning; food preparation; ore refining; stomach acid |
Sulfuric acid (H2SO4) | Fertilizer and explosives manufacturing; batteries; copper electroplating |
Nitric acid (HNO3) | Fertilizer and explosives manufacturing; dye and glue production |
Acetic acid (HC2H3O2) | Plastic and rubber manufacturing; food preservation; vinegar |
Citric acid (H3C6H5O7) | Citrus fruits; flavoring agent |
Carbonic acid (H2CO3) | Carbonated beverages |
Hydrofluoric acid (HF) | Glass etching and frosting |
Phosphoric acid (H3PO4) | Fertilizer manufacturing; beverage preservation |

Name | Occurrence/Uses |
|---|---|
Sodium hydroxide (NaOH) | Petroleum processing; soap and plastic manufacturing |
Potassium hydroxide (KOH) | Cotton processing; soap production; batteries |
Sodium bicarbonate (NaHCO3) | Baking soda; antacid; CO2 source |
Sodium carbonate (Na2CO3) | Glass and soap manufacturing; water softener |
Ammonia (NH3) | Detergent; fertilizer; synthetic fiber production |
Definitions of Acids and Bases
There are several definitions for acids and bases, each with its own scope:
Arrhenius definition: An acid produces H+ ions in aqueous solution; a base produces OH- ions.
Brønsted-Lowry definition: An acid is a proton (H+) donor; a base is a proton acceptor. This definition is more general and applies to a wider range of reactions.



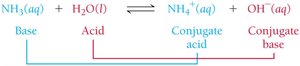
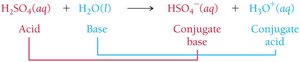
Brønsted-Lowry Acid-Base Reactions and Conjugate Pairs
In Brønsted-Lowry theory, acid-base reactions involve the transfer of a proton from the acid to the base, forming conjugate acid-base pairs.
Conjugate acid: The species formed when a base gains a proton.
Conjugate base: The species formed when an acid loses a proton.
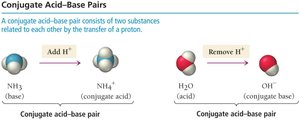
Amphoteric Substances
Some substances, such as water, can act as either an acid or a base depending on the reaction. These are called amphoteric substances.

Acid and Base Strength
The strength of an acid or base is determined by its degree of ionization in water:
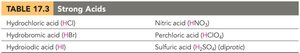
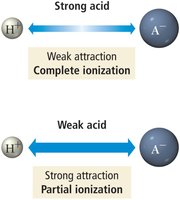
Strong acids: Ionize completely in water (100% ionization). Examples include HCl, HBr, HI, HNO3, HClO4, and H2SO4 (first ionization).
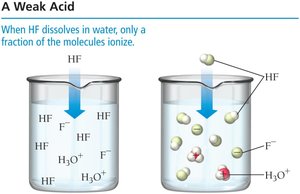
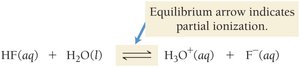
Weak acids: Only partially ionize in water.
Strong bases: Ionize completely in water (e.g., NaOH, KOH).
Weak bases: Only partially ionize in water (e.g., NH3).
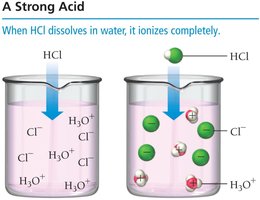
Acid Ionization Constant (Ka)
The strength of a weak acid is quantified by its acid ionization constant, Ka:
Larger Ka: Stronger acid (greater ionization).
Smaller Ka: Weaker acid (less ionization).
The general expression for the ionization of a weak acid HA is:
Autoionization of Water and the Ion Product (Kw)
Water can react with itself to form hydronium and hydroxide ions, a process called autoionization:
The equilibrium constant for this process is the ion product of water, Kw:
at 25°C

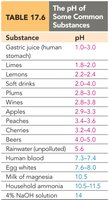
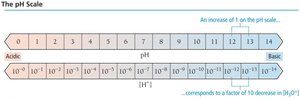
pH and pOH
The acidity or basicity of a solution is commonly expressed using the pH and pOH scales:
pH:
pOH:
Relationship: at 25°C
Interpretation: pH < 7 is acidic, pH > 7 is basic, pH = 7 is neutral.
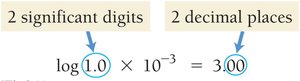
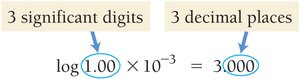

Percent Ionization
Percent ionization is another way to express acid strength, defined as the percentage of acid molecules that ionize in solution:
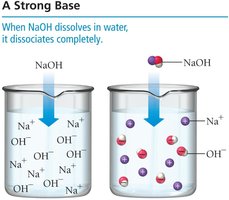
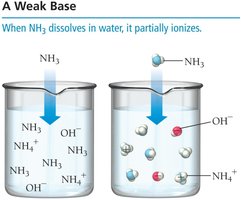
Strong and Weak Bases
Strong bases, such as NaOH and KOH, dissociate completely in water, while weak bases, such as ammonia (NH3), only partially ionize.

Acid-Base Properties of Ions and Salts
Salts are ionic compounds that can affect the pH of a solution depending on the acid-base properties of their constituent ions:
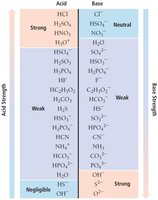
Anions: Conjugate bases of strong acids are neutral; conjugate bases of weak acids are basic.
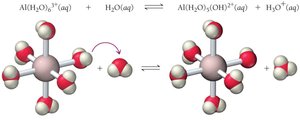
Cations: Conjugate acids of weak bases are weakly acidic; small, highly charged metal cations are weakly acidic; alkali and alkaline earth metal cations are neutral.
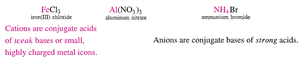

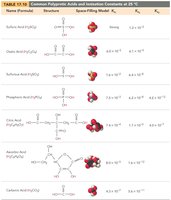
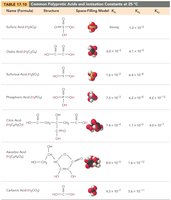
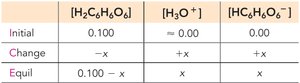
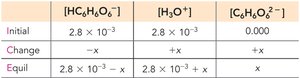
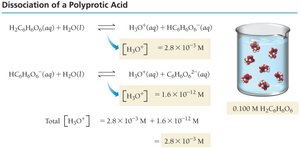
Polyprotic Acids
Polyprotic acids can donate more than one proton per molecule, ionizing in steps. The first ionization usually has the greatest effect on pH.
Acid Strength and Molecular Structure
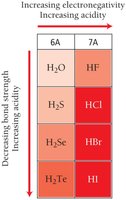
The strength of an acid is influenced by its molecular structure:
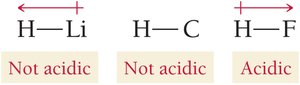
Binary acids: Acidity increases with bond polarity and decreases with bond strength. For example, H–F is a weak acid due to strong bond strength, while H–Cl and H–Br are strong acids.
Oxyacids: Acidity increases with the electronegativity of the central atom and the number of oxygen atoms attached.
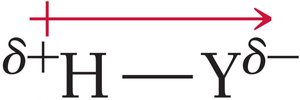

Lewis Acids and Bases
The Lewis definition broadens the concept of acids and bases:
Lewis acid: Electron pair acceptor (often electron-deficient species).
Lewis base: Electron pair donor (species with lone pairs).
This definition includes many reactions not covered by the Brønsted-Lowry theory.
