 Back
BackChapter 18: Free Energy and Thermodynamics – Study Notes
Study Guide - Smart Notes

Free Energy and Thermodynamics
Energy: Forms and Conservation
Energy is the capacity to supply heat or do work. It exists in two main forms: kinetic energy (energy of motion) and potential energy (stored energy). The Law of Conservation of Energy (First Law of Thermodynamics) states that energy cannot be created or destroyed, only converted from one form to another. The total energy of a system is the sum of its kinetic and potential energies.
Kinetic Energy (E_K): Energy due to motion.
Potential Energy (E_P): Energy due to position or composition.
Total Energy:
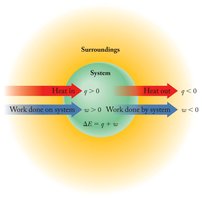
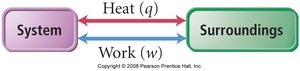
System and Surroundings
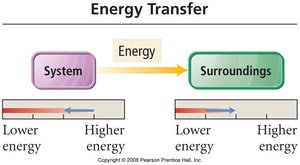
In thermodynamics, the system refers to the part of the universe under study (e.g., a reaction mixture), while the surroundings are everything else. Energy can be transferred between the system and surroundings as heat (q) or work (w).
Heat (q): Energy transferred due to temperature difference.
Work (w): Energy transferred when an object is moved by a force.
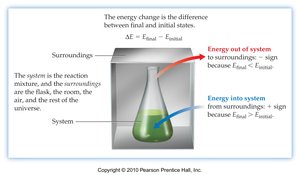
First Law of Thermodynamics
The First Law of Thermodynamics states that the total internal energy of an isolated system is constant. The change in internal energy () is given by:
Work done by the system:
At constant volume:
At constant pressure: (Enthalpy)
Signs of q and w:
Process | Sign |
|---|---|
Heat absorbed by system (endothermic) | q is (+) |
Heat released by system (exothermic) | q is (−) |
Work done on system (V↓) | w is (+) |
Work done by system (V↑) | w is (−) |
Enthalpy Changes
Enthalpy (H) is the heat content of a system at constant pressure. Important enthalpy changes include:
Enthalpy of Fusion (): Heat required to melt a substance.
Enthalpy of Vaporization (): Heat required to vaporize a substance.
Enthalpy of Sublimation (): Heat required to convert a solid directly to a gas.

Thermodynamics and Spontaneity
Spontaneous Processes
A spontaneous process proceeds without outside intervention. It may require an initial energy input but does not necessarily occur quickly. Spontaneity is determined by comparing the chemical potential energy before and after a reaction. If the system's free energy decreases, the process is thermodynamically favorable.
Spontaneous processes can be endothermic or exothermic.
Thermodynamics predicts if a process will occur, not how fast.
Entropy (S) and the Second Law of Thermodynamics
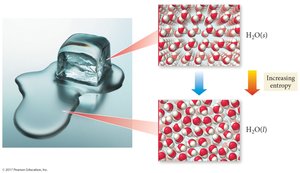
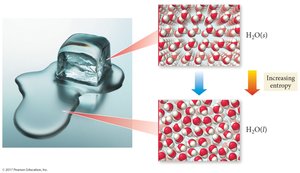
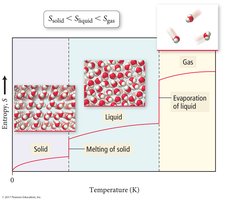
Entropy (S) is a measure of the disorder or randomness of a system. The Second Law of Thermodynamics states that the total entropy of the universe increases in any spontaneous process ().
Entropy increases with more freedom of motion and energy dispersal.
Mixing and heat dispersion increase entropy.
Spontaneous processes are irreversible and increase the universe's entropy.
Microstates and Probability
A microstate is a specific arrangement of the components of a system. The number of microstates (W) determines the entropy:
= Boltzmann constant ( J/K)
More microstates = higher entropy
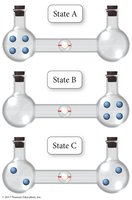
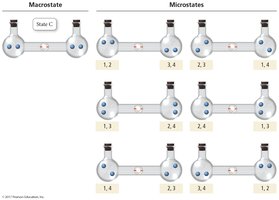
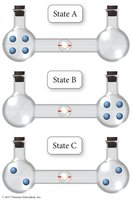
The Third Law of Thermodynamics
The Third Law of Thermodynamics states that the entropy of a perfect crystal at absolute zero (0 K) is zero (). At this temperature, there is only one microstate (), and all molecular motion stops.

Standard Molar Entropy (S°)
Standard molar entropy (S°) is the entropy of one mole of a substance at 1 bar pressure. Entropy increases with phase changes (solid < liquid < gas), molecular complexity, and mass.
Gases have higher entropy than liquids, which have higher entropy than solids:
Linear molecules and less branching increase S°.
Entropy increases with temperature, volume, and number of particles.
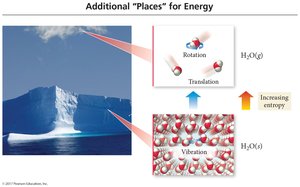
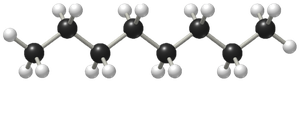
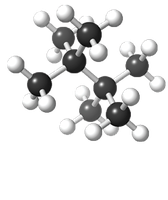
Predicting Entropy Changes
Entropy changes can be predicted by analyzing the number of particles and their states:
Formation of fewer gas molecules: decrease in entropy.
Formation of more gas molecules: increase in entropy.
System, Surroundings, and the Universe
The universe is the sum of the system and its surroundings. For a spontaneous process in an isolated system, .
Exothermic reactions can be spontaneous even if if
Temperature Dependence of Entropy Changes
The effect of heat transfer on entropy depends on the temperature at which it occurs. The higher the temperature, the smaller the impact of a given heat transfer on entropy change.
For surroundings:
Calculating Entropy and Free Energy Changes
(Gibbs Free Energy)
Spontaneity:
Summary Table: Predicting Entropy and Free Energy Changes
Process | Entropy Change (ΔS) | Spontaneity (ΔG) |
|---|---|---|
More gas molecules formed | Increase | More likely spontaneous |
Fewer gas molecules formed | Decrease | Less likely spontaneous |
Exothermic, ΔS > 0 | Increase | Spontaneous at all T |
Endothermic, ΔS < 0 | Decrease | Nonspontaneous at all T |
Key Equations
