 Back
BackChapter 3: Molecules and Compounds – Structured Study Notes
Study Guide - Smart Notes

Chapter 3: Molecules and Compounds
Introduction to Molecules and Compounds
Matter in nature exists in a vast diversity of substances, primarily due to the ability of elements to combine and form compounds. The properties of compounds are often dramatically different from those of the elements that compose them.
Element: A pure substance consisting of only one type of atom.
Compound: A substance formed when two or more elements combine in fixed, definite proportions.
Mixture: A combination of elements or compounds in variable proportions.
Example: Hydrogen and oxygen are both gases, but when combined in a fixed ratio, they form water, a liquid with unique properties.
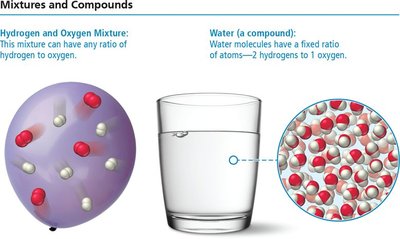
Mixtures vs. Compounds
Mixtures allow elements to combine in any proportion, whereas compounds require elements to combine in fixed ratios.
Mixture: Can have any ratio of hydrogen to oxygen.
Compound (Water): Always has two hydrogen atoms for every one oxygen atom.
Chemical Bonds
Atoms in compounds are held together by chemical bonds, which arise from attractions between charged particles (electrons and protons). Chemical bonds are classified as ionic or covalent.
Ionic Bonds: Occur between metals and nonmetals; involve the transfer of electrons.
Covalent Bonds: Occur between nonmetals; involve the sharing of electrons.
Ionic Bonds and Ionic Compounds
Ionic bonds result in the formation of ions: cations (positive, usually metals) and anions (negative, usually nonmetals). These ions are held together by electrostatic forces.
Ionic Compound: In the solid phase, forms a lattice structure of alternating cations and anions.
Example: Sodium chloride (NaCl) is formed by the transfer of an electron from sodium to chlorine.

Covalent Bonds and Molecular Compounds
Covalent bonds involve the sharing of electrons between two or more nonmetals, resulting in the formation of molecules.
Molecular Compound: Composed of molecules with covalently bonded atoms.
Representing Compounds: Chemical Formulas and Models
Chemical formulas indicate the elements present and the relative number of atoms or ions in a compound. There are three main types of chemical formulas:
Empirical Formula: Shows the simplest ratio of elements.
Molecular Formula: Shows the actual number of atoms of each element in a molecule.
Structural Formula: Shows how atoms are connected and bonded.
Molecular Models
Molecular models visually represent compounds. The ball-and-stick model shows atoms as balls and bonds as sticks, while the space-filling model shows atoms filling the space between each other.
Color Coding: Atoms are color-coded for clarity (e.g., hydrogen is white, carbon is black, oxygen is red).


Classification of Elements and Compounds
Elements can be atomic or molecular, and compounds can be molecular or ionic.
Atomic Elements: Exist as single atoms (e.g., Ne, Na).
Molecular Elements: Exist as molecules (e.g., O2, N2).
Molecular Compounds: Composed of molecules (e.g., H2O).
Ionic Compounds: Composed of formula units (e.g., NaCl).

Molecular Elements
Molecular elements exist as diatomic or polyatomic molecules. There are seven diatomic elements (H2, N2, O2, F2, Cl2, Br2, I2), and some polyatomic elements (e.g., P4, S8).

Molecular and Ionic Compounds
Molecular compounds are composed of covalently bonded nonmetals, while ionic compounds are composed of cations and anions bound by ionic bonds.
Molecular Compound: Smallest unit is a molecule.
Ionic Compound: Smallest unit is a formula unit.
Formulas and Names of Ionic Compounds
Ionic compounds always contain positive and negative ions. The sum of the charges must equal zero, and the formula reflects the smallest whole-number ratio of ions.
Type One Ionic Compounds: Metal has an invariant charge.
Type Two Ionic Compounds: Metal can form more than one type of cation; charge is specified with a Roman numeral.
Common Monoatomic Anions
Nonmetal | Symbol for Ion | Anion Name |
|---|---|---|
Fluorine | F- | Fluoride |
Chlorine | Cl- | Chloride |
Bromine | Br- | Bromide |
Iodine | I- | Iodide |
Oxygen | O2- | Oxide |
Sulfur | S2- | Sulfide |
Nitrogen | N3- | Nitride |
Phosphorus | P3- | Phosphide |
Polyatomic Ions
Polyatomic ions are groups of covalently bonded atoms with an overall charge. Many are oxyanions, containing oxygen and another element.
Name | Formula |
|---|---|
Acetate | C2H3O2- |
Carbonate | CO32- |
Nitrate | NO3- |
Sulfate | SO42- |
Phosphate | PO43- |
Ammonium | NH4+ |
Naming Molecular Compounds
Molecular compounds are named using prefixes to indicate the number of atoms present. The element with the smallest group number is named first.
Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, deca-
Example: NCl3 is nitrogen trichloride; AlCl3 is aluminum chloride (no prefix for ionic compounds).
Acids
Acids are molecular compounds that release hydrogen ions (H+) when dissolved in water. They are composed of hydrogen and one or more nonmetals.
Binary Acids: Composed of hydrogen and a nonmetal. Named with "hydro-" prefix, nonmetal base name, "-ic" ending, and "acid".
Oxyacids: Composed of hydrogen and an oxyanion. If the ion ends in "-ate", the acid name ends in "-ic"; if "-ite", ends in "-ous".
Formula Mass and Molar Mass
The formula mass (or molecular mass) is the sum of the masses of the atoms in a molecule or formula unit. The molar mass is the mass in grams of one mole of molecules or formula units.
Formula Mass:
Molar Mass:
Composition of Compounds
The chemical formula and molar masses of constituent elements indicate the relative quantities of each element in a compound. Mass percentage can be determined from the formula and experimental analysis.
Mass Percent Composition:
Determining Empirical and Molecular Formulas
The empirical formula is the simplest whole-number ratio of atoms in a compound. The molecular formula is a whole-number multiple of the empirical formula.
Steps to Find Empirical Formula:
Convert percentages to grams (assume 100 g sample).
Convert grams to moles using molar mass.
Write a pseudoformula with mole values as subscripts.
Divide all subscripts by the smallest number of moles.
If subscripts are not whole numbers, multiply to obtain whole numbers.
Molecular Formula: Requires empirical formula and molar mass.
Combustion Analysis
Combustion analysis is used to determine the empirical formula of organic compounds by burning a known mass and measuring the products.
All original C forms CO2, all H forms H2O, O is found by subtraction.
Organic Compounds and Hydrocarbons
Organic compounds are primarily composed of carbon and hydrogen, sometimes with oxygen, nitrogen, sulfur, and other elements. Hydrocarbons contain only carbon and hydrogen and are classified as alkanes, alkenes, or alkynes based on the types of bonds present.
Alkanes: Only single bonds (suffix -ane).
Alkenes: At least one double bond (suffix -ene).
Alkynes: At least one triple bond (suffix -yne).
Base Names: meth-, eth-, prop-, but-, pent-, hex-, hept-, oct-, non-, dec-
Common Hydrocarbons
Name | Molecular Formula | Common Uses |
|---|---|---|
Methane | CH4 | Primary component of natural gas |
Propane | C3H8 | LP gas for grills and outdoor stoves |
n-Butane | C4H10 | Common fuel for lighters |
n-Pentane | C5H12 | Component of gasoline |
Ethene | C2H4 | Ripening agent in fruit |
Ethyne | C2H2 | Fuel for welding torches |

Functionalized Hydrocarbons and Organic Families
Functional groups impart specific chemical properties to organic compounds. Families of organic compounds are classified by their functional groups.
Family Name | Ending | General Formula | Example Name | Occurrence/Use |
|---|---|---|---|---|
Alcohols | -ol | R–OH | Ethanol | Alcohol in beverages |
Ethers | ether | R–O–R' | Diethyl ether | Anesthetic; solvent |
Aldehydes | -al | R–CHO | Ethanal | Perfumes; flavors |
Ketones | -one | R–CO–R' | Propanone | Nail polish remover |
Carboxylic acids | acid | R–COOH | Acetic acid | Vinegar |
Esters | -ate | R–COO–R' | Methyl acetate | Solvent |
Amines | amine | R–NH2 | Ethyl amine | Smell of rotten fish |
