 Back
BackChemical Foundations: Matter, Measurement, and Problem Solving
Study Guide - Smart Notes

Chemical Foundations
Matter and Its Composition
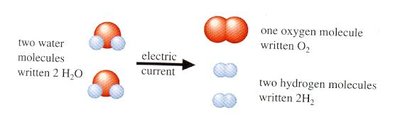
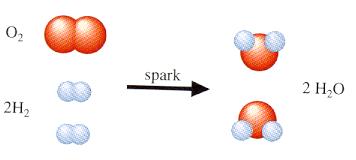
Matter is anything that takes up space, has mass, and exhibits inertia. It is composed of atoms, with only about 100 different types known. For example, water consists of one oxygen atom and two hydrogen atoms. Chemical reactions can rearrange these atoms to form new molecules, and many reactions are reversible.
Atoms: The basic units of matter, each element is defined by its unique type of atom.
Molecules: Combinations of atoms bonded together, such as H2O for water.
Example: Passing an electric current through water separates it into hydrogen and oxygen molecules.
Chemistry: Definition and Importance
Chemistry is the study of matter, energy, and the changes between them. Studying chemistry enhances problem-solving skills, improves safety (e.g., understanding lead poisoning), and deepens understanding of all scientific fields.
The Scientific Method
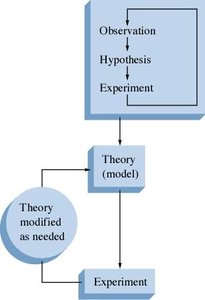
The scientific method is a systematic approach to investigating phenomena, acquiring new knowledge, or correcting and integrating previous knowledge. It consists of several fundamental steps:
Making Observations: Observations can be qualitative (descriptive, e.g., color, state) or quantitative (numerical, e.g., mass, temperature).
Formulating Hypotheses: A hypothesis is a possible explanation for an observation.
Performing Experiments: Experiments test hypotheses, producing new observations and potentially leading to new hypotheses.

Theories, Models, and Scientific Laws
A theory is a set of hypotheses assembled to explain why observed phenomena occur. A model is a representation used to explain natural phenomena, which may change as new evidence is found. A scientific law summarizes observed behavior, while a theory attempts to explain why it happens.
Law of Conservation of Mass: Mass of reactants equals mass of products.
Law of Conservation of Energy: Energy cannot be created or destroyed, only transformed.
Units of Measurement
Quantitative observations always consist of a number and a unit. The two major measurement systems are the English system and the Metric system. The SI (International System of Units) is based on the metric system and is used globally for scientific communication.
Physical Quantity | Name of Unit | Abbreviation |
|---|---|---|
Mass | kilogram | kg |
Length | meter | m |
Time | second | s |
Temperature | kelvin | K |
Electric current | ampere | A |
Amount of substance | mole | mol |
Luminous intensity | candela | cd |

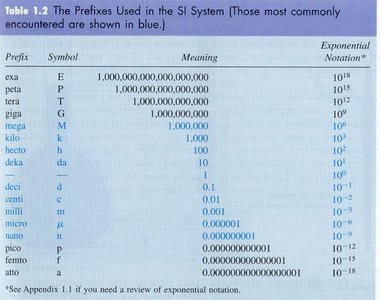
SI Prefixes
SI prefixes are used to denote multiples or fractions of units, facilitating the expression of very large or very small quantities.
Prefix | Symbol | Meaning | Exponential Notation |
|---|---|---|---|
kilo | k | 1,000 | 103 |
centi | c | 0.01 | 10-2 |
milli | m | 0.001 | 10-3 |
micro | μ | 0.000001 | 10-6 |
nano | n | 0.000000001 | 10-9 |
pico | p | 0.000000000001 | 10-12 |
mega | M | 1,000,000 | 106 |
Volume and Its Units
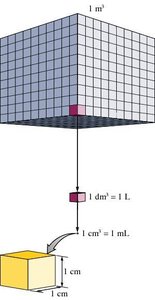
Volume is derived from length. For example, a cube with edges of 1 meter has a volume of 1 m3. A decimeter (dm) is 1/10 of a meter, so 1 dm3 = 1 liter (L), and 1 cm3 = 1 milliliter (mL).
Mass vs. Weight
Mass is a measure of the amount of matter in an object and is measured in grams (g) or kilograms (kg). Weight is the force exerted by gravity on an object and is measured in newtons (N). On Earth, mass and weight are often used interchangeably, but technically they are different.
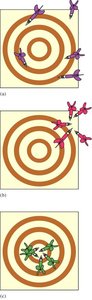
Precision and Accuracy
Accuracy refers to how close a measurement is to the true value, while precision refers to how reproducible measurements are. Errors can be random (indeterminate) or systematic (determinate).
Random Error: Equal probability of being high or low.
Systematic Error: Consistent deviation in one direction.
Significant Figures and Calculations
Significant figures reflect the precision of a measurement. Rules for determining significant figures:
Non-zero digits are always significant.
Zeros are significant if they are terminating and right of the decimal, or sandwiched between significant figures.
Exact numbers and constants have infinite significant figures.
Rules for calculations:
For multiplication and division, the answer should have the same number of significant figures as the term with the least significant figures.
For addition and subtraction, the answer should have the same number of decimal places as the term with the least decimal places.
Dimensional Analysis
Dimensional analysis is a method for converting units by multiplying by conversion factors. For example, to convert 2.85 cm to inches:
Conversion factor:
Calculation:
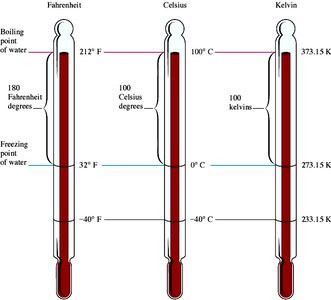
Temperature Scales
There are three main temperature scales: Fahrenheit (°F), Celsius (°C), and Kelvin (K). The relationships between them are:
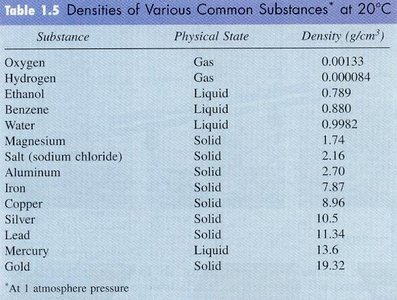
Density
Density is defined as mass per unit volume:
Units: g/cm3 or kg/m3
Substance | Physical State | Density (g/cm3) |
|---|---|---|
Water | Liquid | 0.9982 |
Ethanol | Liquid | 0.789 |
Gold | Solid | 19.32 |
Iron | Solid | 7.87 |
Oxygen | Gas | 0.00133 |
Classification of Matter
Matter can be classified by its physical state and composition:
States of Matter: Solid (rigid, definite shape and volume), Liquid (definite volume, takes shape of container), Gas (no definite volume, highly compressible).
Mixtures: Can be physically separated. Homogeneous mixtures (solutions) have indistinguishable parts; heterogeneous mixtures have distinguishable parts.
Pure Substances: Elements and compounds. Compounds can be separated into elements by chemical means (e.g., electrolysis).
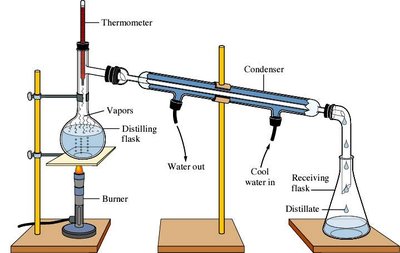
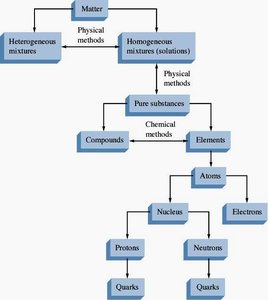

Summary Table: Classification of Matter
Type | Description | Separation Method |
|---|---|---|
Heterogeneous Mixture | Visibly distinguishable parts | Physical methods |
Homogeneous Mixture (Solution) | Indistinguishable parts | Physical methods |
Compound | Pure substance, can be separated into elements | Chemical methods |
Element | Pure substance, cannot be separated further | --- |
