 Back
BackEssentials: Units, Measurements, and Problem Solving – Study Notes
Study Guide - Smart Notes

Essentials: Units, Measurements, and Problem Solving
Measurement Types: Qualitative and Quantitative
Measurements in chemistry are fundamental for describing and analyzing matter. They are categorized as qualitative or quantitative:
Qualitative observations: Descriptive, non-numerical, such as changes in color or physical state.
Quantitative observations: Numerical measurements obtained from instruments or glassware, with varying precision and accuracy. Examples include mass, volume, and counted values (e.g., number of cats per household).
The type of measurement determines the statistical methods used in data analysis.
What Are Measurements?
All measurements consist of two parts:
Number: Indicates the precision of the instrument used (e.g., 25.0 centimeters).
Unit: Specifies the scale (e.g., meters, kilograms). Units may be from the International System of Units (SI), metric, or English system.
Standard Units of Measure (SI)
The SI system is the standard for scientific measurements:
Length: meter (m)
Mass: kilogram (kg)
Time: second (s)
Temperature: kelvin (K)
Amount of substance: mole (mol)
Electric current: ampere (A)
Luminous intensity: candela (cd)
Metric System: Prefix Multipliers
Prefix multipliers are used to express multiples or fractions of base units. For example, kilo (k) means 1000 times the base unit, milli (m) means 1/1000 of the base unit.
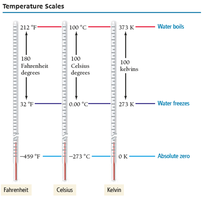
Temperature Scales: Comparison
Temperature is measured using three main scales: Fahrenheit, Celsius, and Kelvin. Each scale has different reference points for freezing and boiling of water, and only Kelvin has no negative values.
Fahrenheit: Water freezes at 32°F, boils at 212°F.
Celsius: Water freezes at 0°C, boils at 100°C.
Kelvin: Water freezes at 273 K, boils at 373 K. Absolute zero is 0 K.
Temperature Calculations
Conversions between temperature scales use specific equations:
To convert Celsius to Kelvin:
To convert Celsius to Fahrenheit:
To convert Fahrenheit to Celsius:
Significant Figures and Measurements
Significant figures reflect the precision of a measurement. Conversion factors are treated as exact values and have an infinite number of significant figures.
Exact values: Defined quantities (e.g., 1 inch = 2.54 cm, 100 pennies = $1).
Significant Figure Rules
All nonzero digits are significant. Example: 536 has three significant figures.
Zeroes between nonzero digits are significant. Example: 6703 has four significant figures.
Leading zeroes are not significant. Example: 0.0043 has two significant figures.
Trailing zeroes after a decimal point are significant. Example: 45.00 has four significant figures.
Trailing zeroes before an implied decimal point are ambiguous. Use scientific notation to clarify.
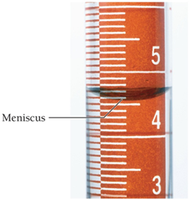
Precision of Laboratory Glassware
When measuring liquids, the last digit is estimated between the marks on the glassware. The meniscus should be read at eye level for accuracy.
Significant Figures and Scientific Notation
Scientific notation helps clarify the number of significant figures in a measurement:
Example: 3010 in scientific notation is
Example: 0.0310 in scientific notation is
Reliability of Measurements: Precision, Accuracy, and Uncertainty
Precision: Closeness of repeated measurements.
Accuracy: Closeness to the true value.
Uncertainty: The estimated range of error in the last digit.
Quantitative Measurement Errors
Systematic (Determinate) Error: Consistent deviation in one direction.
Random (Indeterminate) Error: Unpredictable variation, equally likely to be high or low.
Mathematical Operations and Significant Figures
Multiplication and Division: The answer has the same number of significant figures as the least precise value.
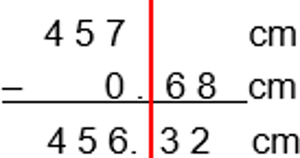
Addition and Subtraction: The answer has the same number of decimal places as the value with the fewest decimal places.
Addition and Subtraction Operations
Examples of significant figures in addition and subtraction:

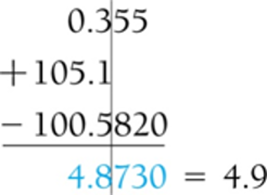

Density: An Intensive Physical Property
Density is defined as mass per unit volume and is an intensive property, meaning it does not depend on the amount of substance.
Formula:
Mass and volume are extensive properties (depend on amount).
Density of liquids and gases changes with temperature.
Introduction to Energy and Its Units
Energy is the capacity to do work, and work is the action of a force applied through a distance. Electrostatic force is a push or pull on charged objects.

Energy Overview
Energy is classified as kinetic or potential:
Kinetic energy: Energy of motion.
Potential energy: Energy due to position or composition.
Energy can be converted between forms (e.g., chemical to mechanical).

Energy Units and Conversion Factors
Calorie (cal): Heat needed to raise 1 g of water by 1°C.
Kilocalorie (kcal):
Joule (J): SI unit of energy. Often used as kilojoule (kJ).
Kilowatt-hour (kWh): Used for electrical energy.
Energy Terminology
First Law of Thermodynamics: Energy is conserved in the universe.
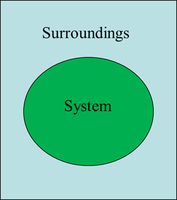
System: The part under study (e.g., a chemical reaction).
Surroundings: Everything outside the system.
Universe: System + surroundings.
Exothermic vs. Endothermic Processes
Exothermic: System loses heat; energy flows to surroundings. Heat is negative.
Endothermic: System gains heat; energy flows from surroundings. Heat is positive.
Dimensional Analysis: Strategy for Solving Problems
Dimensional analysis uses units to guide problem solving. Conversion factors are written as fractions, with the unit being converted from on the bottom and the unit being converted to on top.
How to Use Dimensional Analysis
Arrange conversion factors so the starting unit cancels.
String multiple conversion factors as needed.
Example: To convert inches to centimeters, use .
Practice Problem: Dimensional Analysis
Example: Convert 1.76 yards to centimeters. Conceptual plan:
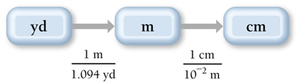
Use relationships: ,
Final answer: 161 centimeters
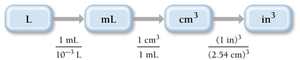
Unit Conversions Involving Units Raised to a Power
When converting units raised to a power (e.g., cubic centimeters to cubic inches), cube the conversion factors.
Using Dimensional Analysis and Equations
Example: Calculating mass from volume and density. Conceptual plan:
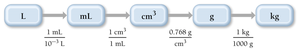
Convert volume to mass using density, then mass to kilograms.
Use relationships:
Strategy for Solving Problems
Sort information: Identify given and find quantities.
Devise a plan: Identify conversion factors and equations.
Solve: Apply mathematical rules for significant figures and cancel units.
Check: Ensure units and magnitude are reasonable.
