 Back
BackLiquids, Solids, and Intermolecular Forces: Chapter 11 Study Guide
Study Guide - Smart Notes

Liquids, Solids, and Intermolecular Forces
States of Matter
The physical state of matter is determined by the arrangement and movement of its constituent particles. The four primary states are solid, liquid, gas, and plasma.
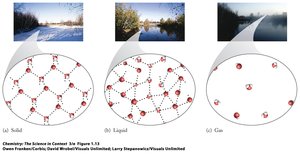
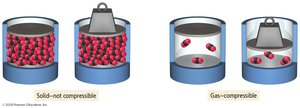
Solid: Fixed volume and rigid shape; particles are closely packed in fixed positions and vibrate but do not move past each other.
Liquid: Fixed volume but no defined shape; particles are closely packed but can move past each other, allowing liquids to flow and take the shape of their container.
Gas: No fixed volume or shape; particles are far apart and move freely, making gases compressible.
Plasma: A gas of charged particles, often found in high-energy environments such as lightning.

Intermolecular Forces
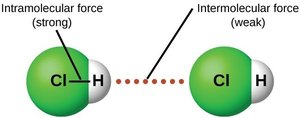
Intermolecular forces are the attractive forces between molecules and atoms, responsible for the existence of condensed phases (liquids and solids). They are weaker than intramolecular (bonding) forces but crucial for determining physical properties.
Types of Intermolecular Forces:
Ion–Ion: Attraction between charged ions.
Ion–Dipole: Attraction between an ion and a polar molecule.
Dipole–Dipole: Attraction between polar molecules.
Hydrogen Bond: Special dipole–dipole interaction involving H bonded to O, N, or F.
Dispersion (London) Forces: Temporary attractions due to instantaneous dipoles.
Induced Dipole: Attraction between a polar molecule and a nonpolar molecule.
Electrostatic Nature: All intermolecular forces are electrostatic and attractive, governed by Coulomb’s Law:
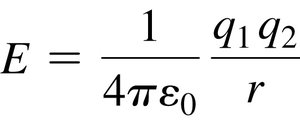
Polarity and Molecular Structure
Polarity arises from differences in electronegativity and molecular geometry. Polar molecules have uneven charge distribution, leading to dipole moments.
Electronegativity: The ability of an atom to attract electrons in a bond. Increases across a period and decreases down a group.
Bond Dipole Moment: (measured in Debyes, D)
Polarity of Molecules: Requires polar bonds and an unsymmetrical shape.
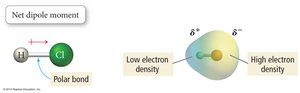
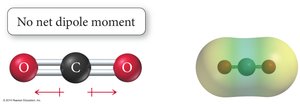
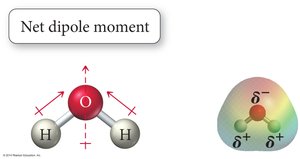
Manifestations of Intermolecular Forces
Intermolecular forces influence boiling point, melting point, vapor pressure, solubility, surface tension, viscosity, and capillary action.
Boiling Point: Stronger intermolecular forces result in higher boiling points.
Solubility: "Like dissolves like"—polar substances dissolve in polar solvents, nonpolar in nonpolar.
Surface Tension: The energy required to increase the surface area of a liquid. Stronger forces yield higher surface tension.
Viscosity: Resistance to flow; increases with stronger intermolecular forces.
Capillary Action: The ability of a liquid to flow up a thin tube, due to cohesive and adhesive forces.


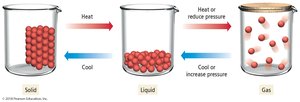
Phase Transitions
Phase changes occur when matter transitions between solid, liquid, and gas states, induced by changes in temperature or pressure.
Vaporization: Endothermic process where liquid becomes gas.
Condensation: Exothermic process where gas becomes liquid.
Melting (Fusion): Endothermic process where solid becomes liquid.
Freezing: Exothermic process where liquid becomes solid.
Sublimation: Solid to gas.
Deposition: Gas to solid.
Energetics of Phase Changes
Energy is required to overcome intermolecular forces during phase changes. The heat of vaporization () and heat of fusion () quantify the energy needed for vaporization and melting, respectively.
Heat of Vaporization: is always positive (endothermic).
Heat of Fusion: is always positive (endothermic).
Relationship:
Dynamic Equilibrium and Vapor Pressure
In a closed container, vaporization and condensation reach a dynamic equilibrium. The vapor pressure is the pressure exerted by the vapor in equilibrium with its liquid.
Vapor Pressure: Increases with temperature and decreases with stronger intermolecular forces.
Boiling Point: The temperature at which vapor pressure equals external pressure.
Clausius–Clapeyron Equation
The Clausius–Clapeyron equation relates vapor pressure and temperature, allowing calculation of heat of vaporization:
Two-Point Form:
Phase Diagrams
Phase diagrams show the state of a substance at various temperatures and pressures. Key features include regions for each state, lines for phase transitions, the triple point, and the critical point.
Triple Point: All three states coexist.
Critical Point: Above this, a supercritical fluid exists.
Special Properties of Water
Water exhibits unique properties due to hydrogen bonding:
High boiling point for its molar mass.
Excellent solvent for ionic and polar substances.
High specific heat moderates climate.
Expansion upon freezing makes ice less dense than liquid water.
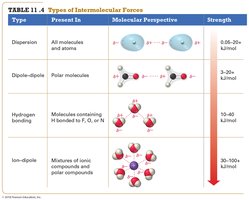
Summary Table: Types of Intermolecular Forces
Type | Present In | Molecular Perspective | Strength (kJ/mol) |
|---|---|---|---|
Dispersion | All molecules and atoms | Temporary dipoles | 0.05–20 |
Dipole–Dipole | Polar molecules | Permanent dipoles | 3–20 |
Hydrogen Bonding | Molecules with H bonded to F, O, or N | Strong dipole–dipole | 10–40 |
Ion–Dipole | Mixtures of ions and polar compounds | Ion and dipole interaction | 30–100+ |
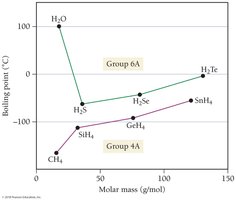
Boiling Points of Group 4A and 6A Compounds
Hydrogen bonding in water, HF, and NH3 leads to higher boiling points than expected. For nonpolar molecules, boiling points increase with molar mass due to stronger dispersion forces.
Surface Tension and Capillary Action
Surface tension is the tendency of liquids to minimize surface area, resulting from cohesive forces. Capillary action is the rise of liquid in a thin tube due to adhesive and cohesive forces.


Practice and Application
Predict boiling points based on intermolecular forces.
Use the Clausius–Clapeyron equation to calculate heat of vaporization.
Interpret phase diagrams to determine state changes.
Key Takeaways
Intermolecular forces determine many physical properties of substances.
Phase changes require energy to overcome these forces.
Water’s unique properties are due to hydrogen bonding.
