 Back
BackLec 03:Biochemistry Foundations for Microbiology: Atoms, Bonds, and Biomolecules
Study Guide - Smart Notes

Chemistry of Living Things
Importance of Biochemistry in Microbiology
Biochemistry is fundamental to understanding microbial life, as it explains how cells obtain energy, grow, utilize nutrients, and interpret genetic instructions. This knowledge is crucial for understanding microbial diseases, improving medical treatments, and advancing biotechnology.
Cellular Energy: Biochemical reactions provide energy for microbial growth and metabolism.
Nutrient Utilization: Microbes use biochemical pathways to assimilate nutrients and synthesize cellular components.
Genetic Regulation: Biochemistry explains how DNA and RNA direct protein synthesis and cellular functions.
Atoms and Their Structure
Atomic Structure and Subatomic Particles
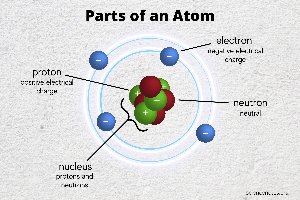
Atoms are the smallest units of matter, forming the basis of all molecules in living organisms. Each atom consists of a nucleus (containing protons and neutrons) and an electron cloud.
Protons: Positively charged particles in the nucleus; determine the atomic number and element identity.
Neutrons: Neutral particles in the nucleus; contribute to atomic mass and stability.
Electrons: Negatively charged particles orbiting the nucleus; involved in chemical bonding and reactions.
Atomic Properties: Mass, Charge, Ions, and Isotopes
Mass and Volume: Most atomic mass is concentrated in the nucleus; electrons occupy most of the volume.
Charge: Atoms are electrically neutral when protons equal electrons.
Ions: Atoms that gain or lose electrons become charged (cations or anions).
Isotopes: Atoms with varying numbers of neutrons; important in medicine and research.
Applications of Isotopes in Medicine
Isotopes are used in medical imaging, targeted therapies, and research.
PET/SPECT Imaging: Radioisotopes track biological processes and detect diseases.
Targeted Treatment: Radioisotopes deliver radiation to specific cells (e.g., thyroid cancer).
Theranostics: Combines diagnosis and treatment using the same molecule.
Sterilization: Isotopes sterilize medical equipment and trace drug movement.
Chemical Bonds
Types of Chemical Bonds
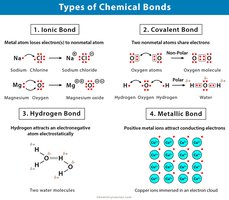
Chemical bonds are forces that hold atoms together, forming molecules essential for life.
Covalent Bonds: Atoms share electrons; strong and common in biological molecules.
Ionic Bonds: Electrons are transferred; resulting ions attract each other.
Metallic Bonds: Electrons are shared among many atoms; characteristic of metals.
Hydrogen Bonds: Weak attractions between hydrogen and electronegative atoms (O, N).
Van der Waals Forces: Weak, temporary attractions due to electron movement.
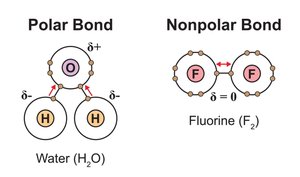
Covalent Bonds: Polar vs. Nonpolar
Nonpolar Covalent: Electrons are shared equally (e.g., O2).
Polar Covalent: Electrons are shared unequally, creating partial charges (e.g., H2O).
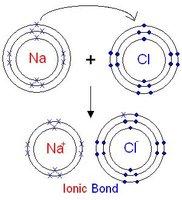
Ionic Bonds
Ionic bonds form when one atom donates an electron to another, resulting in oppositely charged ions that attract each other.
Example: Sodium chloride (NaCl) forms from Na+ and Cl-.
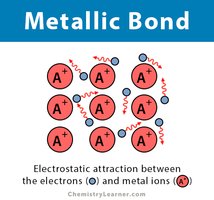
Metallic Bonds
Metallic bonds involve a "sea of electrons" shared among metal atoms, allowing conductivity and malleability.
Example: Copper and other metals.
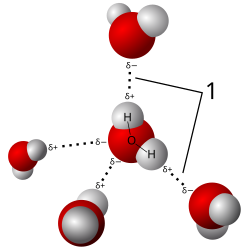
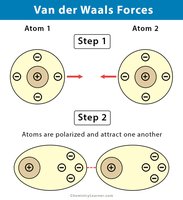
Secondary Bonds: Hydrogen and Van der Waals
Hydrogen Bonds: Weak attractions between hydrogen and electronegative atoms; important in DNA and protein structure.
Van der Waals Forces: Temporary attractions due to electron movement; stabilize large biomolecules.
Biological Importance of Chemical Bonds
Drug Design: Manipulating bond interactions for precise drug targeting.
Materials Science: Understanding bond strengths for developing new materials.
Biological Function: Constant breaking and forming of bonds enables energy storage and release (e.g., ATP hydrolysis).
Organic Macromolecules (Biomolecules)
Overview of Biomolecules
Organic macromolecules are large, carbon-based molecules essential for life. They are formed by joining monomers into polymers via dehydration synthesis.
Carbohydrates
Lipids
Proteins
Nucleic Acids
Carbohydrates
Carbohydrates provide energy and structural support. They typically have a 1:2:1 ratio of carbon, hydrogen, and oxygen.
Monosaccharides: Simple sugars (e.g., glucose, fructose, galactose).
Functions: Immediate energy, short-term storage, structural support.

Lipids
Lipids are hydrophobic molecules that store energy and form cell membranes.
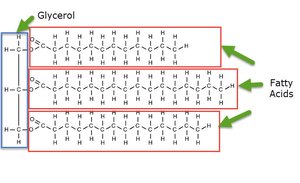
Fats: Solid at room temperature; composed of glycerol and fatty acids.
Oils: Liquid at room temperature.
Fatty Acids: Can be saturated (no double bonds) or unsaturated (one or more double bonds).
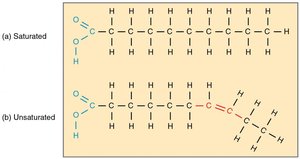
Saturated vs. Unsaturated Fatty Acids
Saturated: No double bonds; straight chains; solid at room temperature; often considered less healthy.
Unsaturated: One or more double bonds; bent chains; liquid at room temperature; considered healthier.


Compound Lipids and Sterols
Compound lipids contain additional chemical groups, such as phospholipids (phosphate group), glycolipids (hexose units), sulpholipids (sulfated hexose), and lipoproteins (protein subunits). Sterols, like cholesterol, are characterized by four fused rings and are important for hormone production, bile synthesis, and membrane structure. 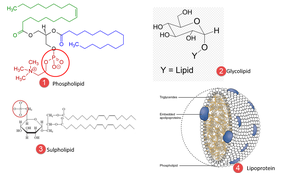
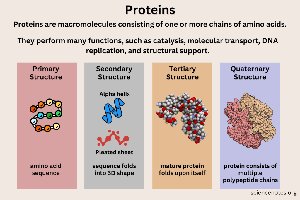
Proteins
Proteins are polymers of amino acids and are the most functionally diverse biomolecules. Their structure determines their function, including catalysis, transport, signaling, movement, and defense.
Enzymes: Catalyze biochemical reactions.
Structural: Provide cellular framework.
Transport: Move molecules across membranes.
Defense: Antibodies protect against pathogens.
Hormonal: Chemical messengers.
Movement: Muscle contraction.
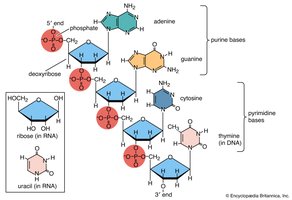
Nucleic Acids
Nucleic acids (DNA and RNA) store and transmit genetic information and direct protein synthesis.
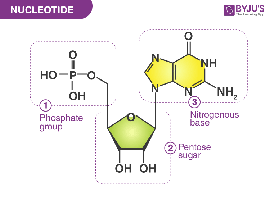
Nucleotides: Building blocks with phosphate, sugar, and base.
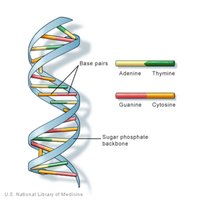
DNA: Double-stranded helix; stores hereditary information.
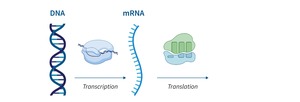
RNA: Single-stranded; involved in protein synthesis (mRNA, tRNA, rRNA).
Summary Table: Biomolecule Classes
Class | Monomer | Main Functions | Examples |
|---|---|---|---|
Carbohydrates | Monosaccharides | Energy, structure | Glucose, cellulose |
Lipids | Fatty acids, glycerol | Energy storage, membranes | Triglycerides, phospholipids |
Proteins | Amino acids | Catalysis, structure, transport | Enzymes, collagen |
Nucleic Acids | Nucleotides | Genetic information | DNA, RNA |
Key Equations and Concepts
Atomic Mass:
Dehydration Synthesis:
ATP Hydrolysis:
Conclusion
Understanding the chemistry of living things is foundational for microbiology, as it explains the molecular basis of microbial structure, function, and diversity. Mastery of atomic structure, chemical bonds, and biomolecules enables deeper insight into microbial metabolism, genetics, and cellular processes. Additional info: Expanded explanations and context were added to ensure completeness and academic quality.
