 Back
BackInnate Immunity: Principles and Mechanisms (Chapter 11 Study Notes)
Study Guide - Smart Notes

Innate Immunity: Principles and Mechanisms
Overview of Immune Responses
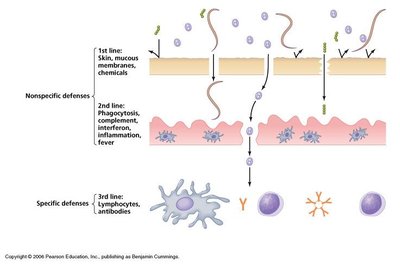
The immune system protects the body from pathogens through coordinated physiological processes. Immunity is divided into two main branches: innate immunity (inborn, nonspecific) and adaptive immunity (acquired, specific). Both branches share the abilities to recognize diverse pathogens, eliminate invaders, and distinguish self from non-self antigens.
Innate Immunity: Present in all eukaryotes, provides immediate, nonspecific defense.
Adaptive Immunity: Found only in vertebrates, matures over time, is highly specific, and exhibits memory.
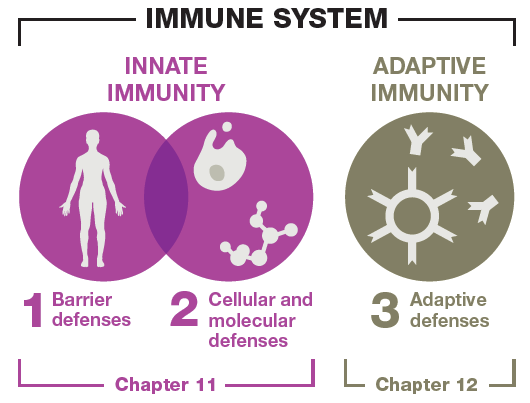
Comparison of Innate and Adaptive Immunity
Innate and adaptive immunity differ in their mechanisms, response times, and specificity. The following table summarizes their key features:
Feature | Innate Immunity | Adaptive Immunity |
|---|---|---|
Response time | Immediate | 4–7 days |
Organisms | All eukaryotes | Only vertebrates |
Distinguishes self from foreign | Yes | Yes |
Kills invaders | Yes | Yes |
Effective against diverse threats | Yes | Yes |
Tailors response to specific antigen | No | Yes |
Memory | No | Yes |
Normal Microbiota and Immune System Development
Role of Normal Microbiota
Normal microbiota are symbiotic microbes that inhabit the body and play a crucial role in shaping immune responses. They help train the immune system to distinguish between harmful and harmless agents, and their disruption can lead to immune confusion, allergies, or autoimmunity.
Hygiene Hypothesis: Suggests that reduced microbial exposure may negatively affect immune development.
Germ-free animal studies show underdeveloped immune systems without microbiota.
First-Line Defenses: Preventing Pathogen Entry
Types of First-Line Defenses
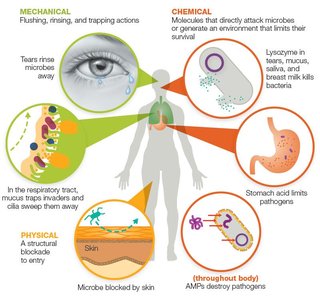
First-line defenses are barriers that prevent pathogen entry and are categorized as mechanical, chemical, or physical barriers.
Mechanical Barriers: Rinse, flush, or trap pathogens (e.g., tears, urine, saliva, mucus, mucociliary escalator, earwax).
Chemical Barriers: Directly attack invaders or create hostile environments (e.g., lysozyme, stomach acid, skin acidity, fatty acids, antimicrobial peptides).
Physical Barriers: Structures that physically block entry (e.g., skin, epithelial tissue).
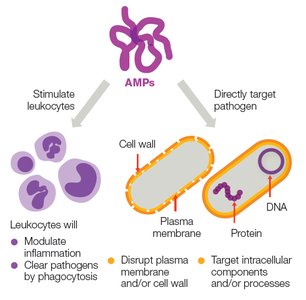
Antimicrobial Peptides (AMPs)
AMPs are proteins that destroy a wide range of pathogens. They can stimulate leukocytes, modulate inflammation, and directly disrupt microbial membranes or intracellular processes.
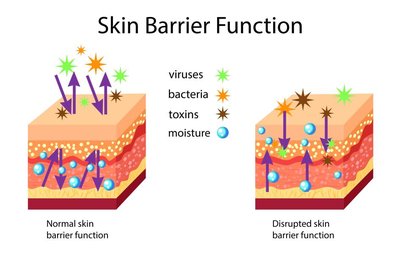
Skin as a Physical Barrier
The skin is a critical physical barrier composed of tightly packed, dead epithelial cells enriched with proteins and lipids, forming a water-resistant layer that blocks pathogen entry.
Second-Line Defenses: Cellular and Molecular Responses
Leukocytes and Molecular Factors
When first-line defenses are breached, second-line defenses involving leukocytes (white blood cells) and molecular factors are activated to eliminate invaders.
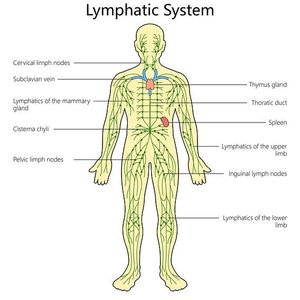
Lymphatic System
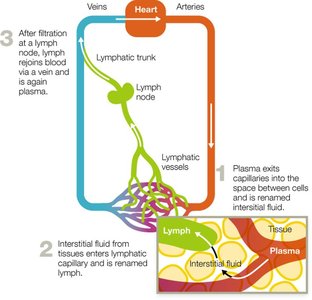
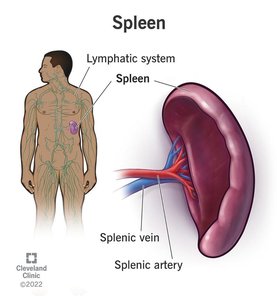
The lymphatic system collects, circulates, and filters body fluids, playing a vital role in immune surveillance and response. Lymph nodes filter lymph, while the spleen filters blood and removes damaged erythrocytes.
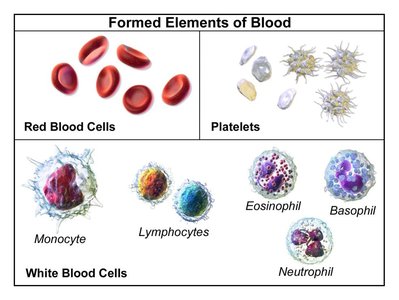
Formed Elements of Blood
Blood contains erythrocytes (red blood cells), leukocytes (immune cells), and platelets (involved in clotting and confining pathogens).
Primary and Secondary Lymphoid Tissues
Primary lymphoid tissues: Sites of immune cell production and maturation (bone marrow, thymus).
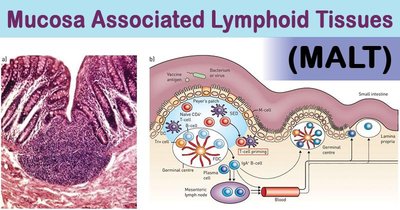
Secondary lymphoid tissues: Sites where immune responses are initiated (lymph nodes, spleen, MALT).
Leukocytes: Types and Functions
Classification of Leukocytes
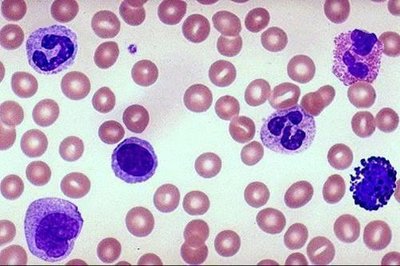
Leukocytes are classified as granulocytes (with cytoplasmic granules) or agranulocytes (without granules). Major types include neutrophils, eosinophils, basophils, mast cells, monocytes, macrophages, dendritic cells, and lymphocytes.
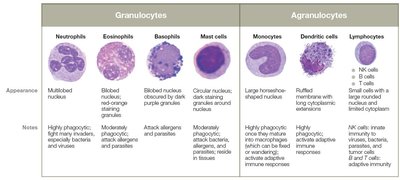
Granulocytes
Neutrophils: Most abundant, first responders, phagocytize pathogens, release AMPs.
Eosinophils: Combat parasites, involved in allergies, moderate phagocytic activity.
Basophils: Release histamine, involved in allergic responses, rare in circulation.
Mast Cells: Reside in tissues, release histamine, fight parasites, activate adaptive immunity.
Agranulocytes
Monocytes: Largest WBCs, migrate into tissues to become macrophages.
Macrophages: Highly phagocytic, reside in tissues or wander, destroy pathogens.
Dendritic Cells: Highly phagocytic, present antigens to T cells, prevent autoimmunity.
Lymphocytes: Include NK cells (innate), B cells, and T cells (adaptive).
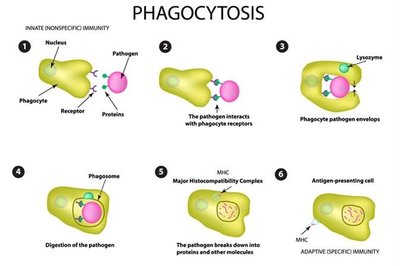
Molecular Second-Line Defenses
Cytokines
Cytokines are signaling proteins that coordinate immune responses, including inflammation, fever, leukocyte recruitment, and tissue repair. Major types include chemokines, interleukins, interferons, and tumor necrosis factors.
Cytokine Type | Examples | Functions |
|---|---|---|
Chemokines | Monocyte chemoattractant protein-1 | Recruit WBCs, wound healing, blood vessel formation |
Interleukins (IL-1, IL-2) | IL-1, IL-2 | Regulate inflammation, fever, T cell development |
Interferons | IFN-α, IFN-β, IFN-γ | Antiviral responses, activate macrophages |
Tumor necrosis factors | TNF-α | Induce inflammation, kill tumor cells, fever |
Iron-Binding Proteins
Iron-binding proteins (hemoglobin, ferritin, lactoferrin, transferrin) limit free iron to restrict bacterial growth. Some pathogens have evolved mechanisms to acquire iron, such as producing siderophores or lysing red blood cells.
Complement System
The complement system consists of over 30 proteins that enhance immune responses through a cascade mechanism. Activation occurs via three pathways: classical (antibody-triggered), alternative (direct pathogen interaction), and lectin (mannose-binding lectin). Outcomes include opsonization, membrane attack complex (cytolysis), and inflammation.
Inflammation and Fever
Inflammation
Inflammation is a key innate response to tissue damage or infection, with three main goals: recruit immune defenses, limit pathogen spread, and promote tissue repair. The process occurs in three phases: vascular changes, leukocyte recruitment, and resolution.
Vascular changes: Vasodilation, increased permeability, exudate formation (mediated by histamine, kinins, eicosanoids).
Leukocyte recruitment: Margination and diapedesis allow leukocytes to exit vessels and reach infection sites.
Resolution: Inflammation is toned down, tissue repair begins, and pus may form.
Fever
Fever (pyrexia) is a systemic response to infection, induced by pyrogens (e.g., bacterial toxins, cytokines) that signal the hypothalamus to raise body temperature. Fever enhances immune efficiency but can be dangerous if too high.
Low-grade fever: Protective, enhances immune responses.
High fever (>40.5°C): Medical emergency, can cause protein denaturation and death.
Fever Type | Description |
|---|---|
Fever of undetermined origin (FUO) | ≥38.3°C for >3 weeks, no clear cause |
Intermittent | Elevates, returns to normal daily |
Pel–Ebstein | 3–10 days fever, then similar nonfever period |
Relapsing | Recurrent episodes, days/weeks apart |
Remittent | Elevated, fluctuates, never normal |
Sustained | Consistently elevated, minimal fluctuation |
Tertian | Fever on 1st and 3rd days (malaria) |
Quartan | Fever on 1st and 4th days (malaria) |
Clinical Application: Case Study
Case Summary
A 5-year-old boy with recurrent infections was diagnosed with bacterial meningitis caused by Streptococcus pneumoniae despite vaccination. He was found to have a deficiency in mannose-binding lectin (MBL), affecting his lectin pathway of complement activation.
Key findings: Low MBL levels, recurrent pyrogenic infections, normal adaptive response to vaccines expected.
Clinical implications: Early infection detection and prompt treatment are critical; MBL deficiency impairs innate immunity but not adaptive vaccine response.
Key Concepts Illustrated by the Case
Differential WBC count: Likely shows increased neutrophils due to bacterial infection.
Fever mechanism: Pyrogens induce cytokine release, signaling the hypothalamus to raise temperature; antipyretics inhibit prostaglandin synthesis.
First-line defenses breached: Pathogen overcame mechanical, chemical, and physical barriers; second-line defenses (phagocytes, complement) were activated.
Hemochromatosis check: To rule out iron overload, which increases infection risk.
