 Back
BackMicrobial Metabolism: Catabolism, Anabolism, and Energy Pathways
Study Guide - Smart Notes

Microbial Metabolism: Overview
Definition and Importance
Metabolism is the sum of all chemical reactions occurring within a microbial cell. These reactions are essential for energy production, growth, and maintenance of cellular functions. Microbial metabolism is divided into two main processes: catabolism (breakdown of molecules to release energy) and anabolism (synthesis of complex molecules from simpler ones).
Dissimilative pathways: Energy-yielding breakdown of substrates.
Assimilative pathways: Incorporation of nutrients into cellular material.
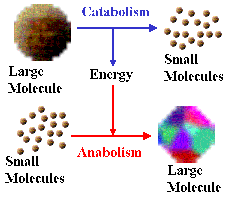
Applications of microbial metabolism:
Biogeochemical cycles (carbon, nitrogen, sulfur cycles)
Wastewater treatment and bioremediation (e.g., degradation of petroleum hydrocarbons, pesticides, plastics)
Food industry (production of cheese, alcohol, vinegar, yogurt, bread)
Human microbiome (microbial cells outnumber human cells by about 10:1)
Production of small biological molecules (vitamins, amino acids, antibiotics, vaccines, biopolymers, restriction enzymes)

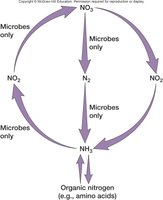
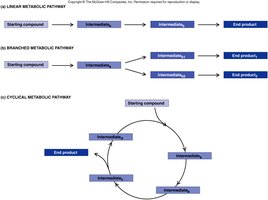
Organization of Metabolic Pathways
Metabolic Pathways and Enzyme Function
Metabolic pathways are organized sequences of enzymatic reactions, each catalyzed by a specific enzyme. Pathways can be linear, branched, or cyclic, and they transform substrates into end products through a series of intermediates.
Substrate: The starting molecule of a pathway.
Product: The final molecule produced.
Each enzyme catalyzes a specific reaction type.
Thermodynamics and Enzyme Catalysis
First Law of Thermodynamics
The first law of thermodynamics states that energy cannot be created or destroyed, only transformed from one form to another. In biological systems, chemical energy is often converted to heat, mechanical work, or stored in molecules like ATP.
Energy transformations are central to metabolism.
Some energy is always lost as heat during these transformations (second law of thermodynamics).

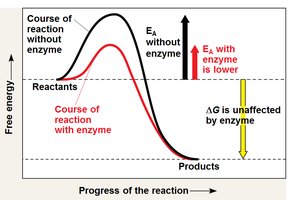
Activation Energy and Enzymes
Activation energy (Ea) is the energy required to initiate a chemical reaction. Enzymes act as biological catalysts, lowering the activation energy and increasing the rate of metabolic reactions without being consumed.
Enzymes do not alter the overall free energy change (ΔG) of a reaction.
They provide a specific environment for the reaction to occur more efficiently.
Catabolism: Energy Release and Conservation
Catabolic Pathways
Catabolism involves the breakdown of complex molecules into simpler ones, releasing energy that is conserved in the form of ATP or other energy carriers. Major catabolic processes include aerobic respiration, anaerobic respiration, and fermentation.
Aerobic respiration: Uses oxygen as the terminal electron acceptor.
Anaerobic respiration: Uses other inorganic molecules (e.g., NO3-, SO42-, CO2) as electron acceptors.
Fermentation: Organic molecules serve as both electron donors and acceptors; less ATP is produced.
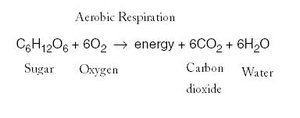
Nutritional Types of Microorganisms
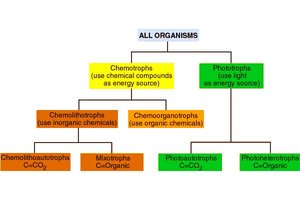
Microorganisms are classified based on their sources of carbon, energy, and electrons:
Type | Carbon Source | Energy Source | Electron Source |
|---|---|---|---|
Autotrophs | CO2 | Light or chemicals | Inorganic or organic molecules |
Heterotrophs | Organic compounds | Light or chemicals | Organic molecules |
Phototrophs | CO2 or organics | Light | Varies |
Chemotrophs | CO2 or organics | Chemicals | Varies |
Lithotrophs | CO2 or organics | Light or chemicals | Inorganic molecules |
Organotrophs | CO2 or organics | Light or chemicals | Organic molecules |

Major Catabolic Pathways
Glycolysis: The breakdown of glucose to pyruvate, generating ATP and NADH.
Krebs (TCA) cycle: Oxidizes acetyl-CoA to CO2, producing NADH, FADH2, and ATP/GTP.
Electron Transport Chain (ETC): Transfers electrons from NADH/FADH2 to terminal electron acceptors, generating a proton motive force (PMF) used to synthesize ATP.
ATP yield varies by pathway and terminal electron acceptor:
Aerobic respiration: up to 36 ATP per glucose
Anaerobic respiration: 2–32 ATP per glucose
Fermentation: 2 ATP per glucose
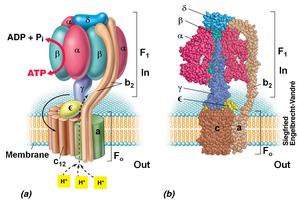
ATP Synthesis
ATP synthase is a membrane-bound enzyme complex that synthesizes ATP using the energy from the proton motive force (PMF) as protons flow back into the cell.
Approximately 3 H+ are required to generate 1 ATP molecule.
ATP turnover in bacteria is extremely high, supporting rapid growth and metabolism.
Redox Balance and Fermentation
Redox Balance
Cells maintain a balance between oxidized (NAD+) and reduced (NADH) forms of electron carriers. During fermentation, NAD+ is regenerated by transferring electrons to organic acceptors, producing end products such as ethanol or lactate.
Alcohol fermentation: Saccharomyces species convert glucose to ethanol and CO2, regenerating NAD+.
Fermentation is essential for redox balance when external electron acceptors are absent.
Phototrophy and Chemolithotrophy
Phototrophy
Phototrophic microorganisms capture light energy and convert it to chemical energy. There are two main types:
Oxygenic phototrophy: Generates oxygen (e.g., cyanobacteria, algae).
Anoxygenic phototrophy: Does not generate oxygen (e.g., purple and green bacteria).
Photosynthesis consists of light reactions (energy capture) and dark reactions (CO2 fixation).
Anabolism: Biosynthesis of Cellular Components
Principles of Anabolism
Anabolism is the synthesis of complex molecules from simpler precursors, requiring energy (usually from ATP) and reducing power (NADPH). Key principles include:
Macromolecules are synthesized from a limited set of monomers (e.g., amino acids, nucleotides).
Many enzymes function in both catabolic and anabolic pathways, but some steps are unique to each direction.
Catabolic and anabolic pathways are often physically separated and use different cofactors (NADH for catabolism, NADPH for anabolism).
Precursor Metabolites
Central metabolic pathways generate precursor metabolites that serve as starting materials for biosynthesis (e.g., intermediates from glycolysis and the TCA cycle).
Examples: Acetyl-CoA, pyruvate, ribose-5-phosphate, oxaloacetate.
These are used to synthesize amino acids, nucleotides, lipids, and other macromolecules.
CO2 Fixation Pathways
Autotrophic microorganisms fix CO2 into organic molecules using several pathways:
Calvin-Benson cycle: Main pathway in plants, algae, and cyanobacteria.
Reductive TCA cycle, hydroxypropionate bi-cycle, reductive acetyl-CoA pathway: Used by various bacteria and archaea.
The Calvin-Benson cycle consists of three phases: carboxylation, reduction, and regeneration. For each CO2 fixed, three ATP and two NADPH are consumed.
Summary Table: Sources of Carbon, Energy, and Electrons
Type | Carbon Source | Energy Source | Electron Source |
|---|---|---|---|
Autotrophs | CO2 | Light or chemicals | Inorganic or organic molecules |
Heterotrophs | Organic compounds | Light or chemicals | Organic molecules |
Phototrophs | CO2 or organics | Light | Varies |
Chemotrophs | CO2 or organics | Chemicals | Varies |
Lithotrophs | CO2 or organics | Light or chemicals | Inorganic molecules |
Organotrophs | CO2 or organics | Light or chemicals | Organic molecules |
Additional info: The above notes integrate and expand upon the provided material, ensuring a comprehensive, self-contained study guide for microbial metabolism, suitable for college-level microbiology students.
