 Back
BackMicrobial Metabolism: Foundations and Pathways
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Metabolism
Metabolism encompasses all controlled biochemical reactions occurring within a microbe, serving the ultimate function of reproducing the organism. These reactions are responsible for both the breakdown and synthesis of nutrients, providing energy and essential substances for life.
Metabolism: The sum of all chemical reactions in an organism.
Catabolism: Breakdown of larger molecules into smaller products, releasing energy (exergonic).
Anabolism: Synthesis of larger molecules from smaller products, requiring energy (endergonic).
Energy is stored and transferred via ATP (adenosine triphosphate).
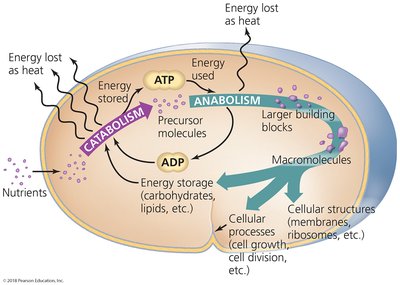
Eight Elementary Statements Guiding Metabolic Processes
Cells acquire nutrients.
Metabolism requires energy from light or catabolism of nutrients.
Energy is stored in ATP.
Catabolism forms precursor metabolites.
Precursor metabolites, ATP, and enzymes drive anabolic reactions.
Enzymes and ATP form macromolecules.
Cells grow by assembling macromolecules.
Cells reproduce after doubling in size.
Basic Chemical Reactions Underlying Metabolism
Oxidation and Reduction Reactions
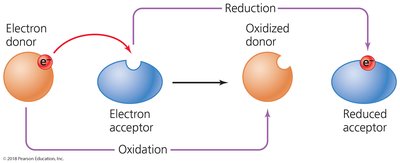
Oxidation-reduction (redox) reactions involve the transfer of electrons from an electron donor to an electron acceptor. These reactions are fundamental to energy production in cells and always occur simultaneously.
Oxidation: Loss of electrons.
Reduction: Gain of electrons.
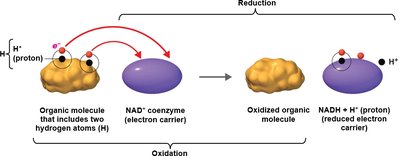
Cells use electron carriers such as NAD+, NADP+, and FAD.
ATP Production and Energy Storage
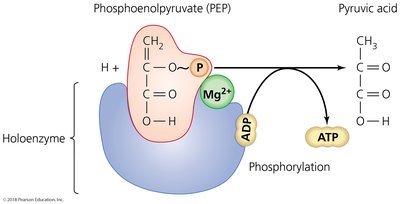
Organisms release energy from nutrients and store it in high-energy phosphate bonds of ATP. Phosphorylation is the process of adding inorganic phosphate to a substrate, and cells phosphorylate ADP to ATP in three ways:
Substrate-level phosphorylation
Oxidative phosphorylation
Photophosphorylation
Anabolic pathways utilize ATP by breaking a phosphate bond.
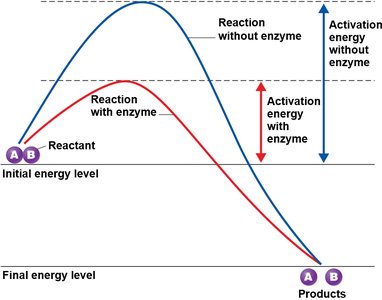
Collision Theory and Enzyme Function
The collision theory states that chemical reactions occur when atoms, ions, and molecules collide with sufficient energy (activation energy). Enzymes, as biological catalysts, lower the activation energy required for reactions, increasing reaction rates without being consumed.
Activation energy: Minimum energy required for a reaction.
Enzymes: Proteins that act on specific substrates.
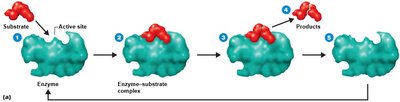
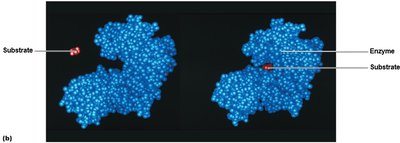
Mechanism of Enzymatic Action
Enzymes bind substrates at their active sites, forming an enzyme-substrate complex. The substrate is transformed into products, which are released, leaving the enzyme unchanged and ready for another reaction. This is often described as the "lock and key" model.
Enzyme specificity is determined by the shape of the active site.
Turnover number: Number of substrate molecules converted per second (typically 1 to 10,000).
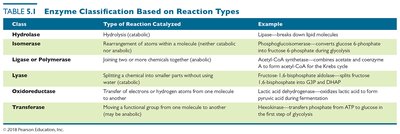
Enzyme Classification and Naming
Enzymes are named based on the reactions they catalyze, usually ending in "-ase." There are six major classes:
Class | Type of Reaction Catalyzed | Example |
|---|---|---|
Hydrolase | Hydrolysis (catabolic) | Lipase—breaks down lipid molecules |
Isomerase | Rearrangement of atoms within a molecule | Phosphoglucoisomerase—converts glucose 6-phosphate to fructose 6-phosphate |
Ligase/Polymerase | Joining two or more chemicals together (anabolic) | Acetyl-CoA synthetase—combines acetate and CoA to form acetyl-CoA |
Lyase | Splitting a chemical into smaller parts without using water | Fructose 1,6-bisphosphate aldolase—splits fructose 1,6-bisphosphate into G3P and DHAP |
Oxidoreductase | Transfer of electrons or hydrogen atoms from one molecule to another | Lactic acid dehydrogenase—oxidizes lactic acid to form pyruvic acid |
Transferase | Moving a functional group from one molecule to another | Hexokinase—transfers phosphate from ATP to glucose in the first step of glycolysis |
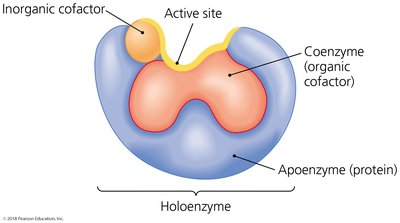
Enzyme Structure and Cofactors
Many enzymes are complete proteins, but some require nonprotein cofactors (inorganic ions or coenzymes) to be active. The combination of an apoenzyme (inactive protein) and its cofactor forms a holoenzyme. Some enzymes are RNA molecules called ribozymes.
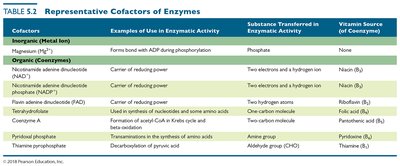
Cofactor | Examples of Use | Substance Transferred | Vitamin Source |
|---|---|---|---|
Magnesium (Mg2+) | Forms bond with ADP during phosphorylation | Phosphate | None |
NAD+ | Center of reducing power | Two electrons and a hydrogen ion | Niacin (B3) |
NADP+ | Center of reducing power | Two hydrogen atoms | Riboflavin (B2) |
FAD | Used in synthesis of nucleotides and some amino acids | One carbon molecule | Pantothenic acid (B5) |
Pyridoxal phosphate | Transamination in the synthesis of amino acids | Amino group | Pyridoxine (B6) |
Thiamine pyrophosphate | Decarboxylation of pyruvic acid | Aldehyde group (CHO) | Thiamine (B1) |
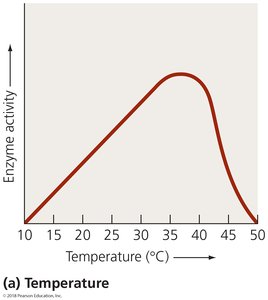
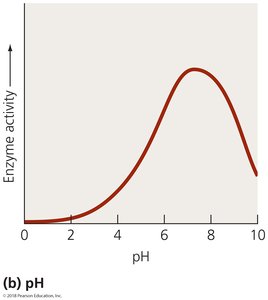
Factors Affecting Enzyme Activity
Enzyme activity is influenced by several factors:
Temperature: Each enzyme has an optimal temperature for activity.
pH: Enzymes function best at specific pH levels.
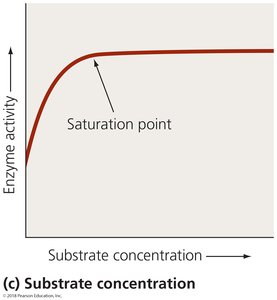
Substrate concentration: Activity increases with substrate concentration up to a saturation point.
Presence of inhibitors: Can block enzyme activity.
Control of Enzymatic Activity
Enzyme activity can be regulated by activators and inhibitors:
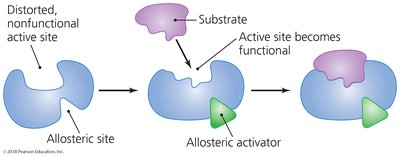
Allosteric activation: Cofactor binds to a site other than the active site, activating the enzyme.
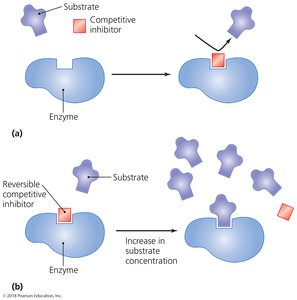
Competitive inhibition: Inhibitor competes with substrate for the active site.
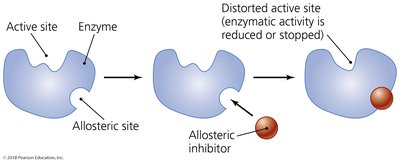
Noncompetitive inhibition: Inhibitor binds to an allosteric site, altering the enzyme's shape and function.
Feedback inhibition: End product of a pathway inhibits an earlier enzyme, regulating the pathway.
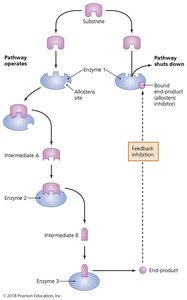
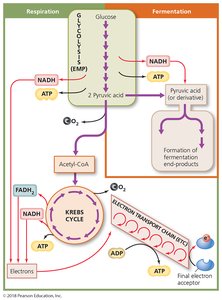
Carbohydrate Catabolism
Overview of Glucose Catabolism
Most organisms oxidize carbohydrates, primarily glucose, as their main energy source. Glucose catabolism occurs via two main processes: cellular respiration and fermentation.
Glycolysis
Glycolysis is the process of splitting a six-carbon glucose into two three-carbon molecules (pyruvic acid), occurring in the cytoplasm. It involves substrate-level phosphorylation and yields a net gain of two ATP, two NADH, and two pyruvic acid molecules.
Three stages: Energy-investment, lysis, and energy-conserving.
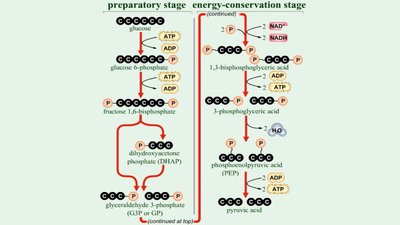
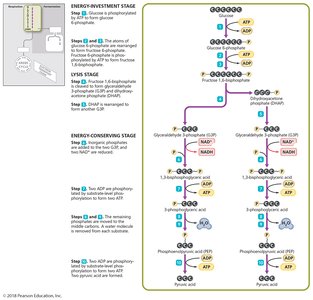
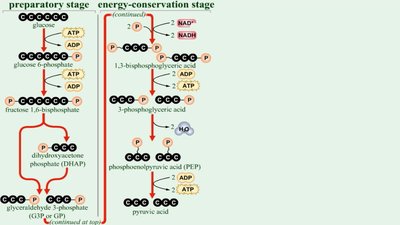
Substrate-Level Phosphorylation
Direct transfer of phosphate between two substrates during glycolysis and other metabolic pathways.
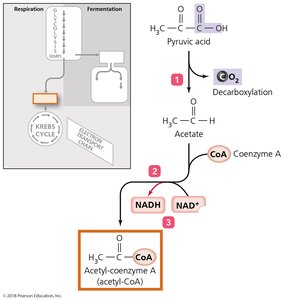
Cellular Respiration
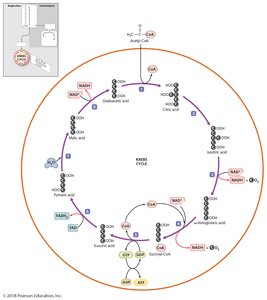
Cellular respiration completely oxidizes pyruvic acid to produce ATP through a series of redox reactions. It consists of three stages:
Synthesis of acetyl-CoA
Krebs cycle
Electron transport chain (ETC)
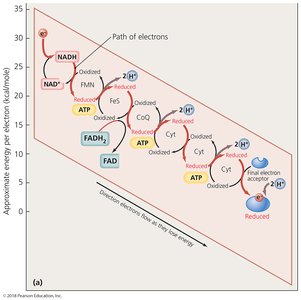
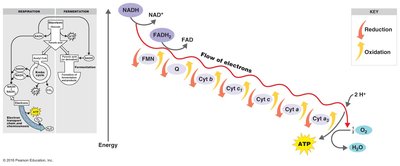
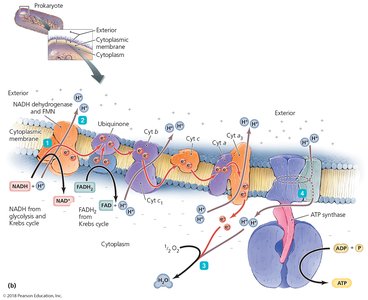
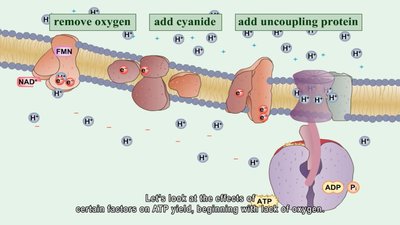
Electron Transport Chain and Chemiosmosis
The ETC is a series of carrier molecules that pass electrons to a final electron acceptor, generating a proton gradient used to produce ATP via chemiosmosis. In aerobic respiration, oxygen is the final electron acceptor; in anaerobic respiration, it is another molecule.
Four categories of carrier molecules: Flavoproteins, ubiquinones, metal-containing proteins, cytochromes.
Chemiosmosis
Cells use the energy released in ETC redox reactions to create a proton gradient. Protons flow through ATP synthase, phosphorylating ADP to ATP. This process is called oxidative phosphorylation.
Metabolic Diversity: Alternative Pathways
Entner-Doudoroff (ED) pathway: Alternative to glycolysis in some prokaryotes, producing one ATP, NADH, and NADPH.
Pentose phosphate pathway: Produces precursor metabolites and NADPH, used for biosynthesis of nucleotides, steroids, and fatty acids.
Fermentation
Fermentation provides cells with an alternative source of NAD+ when cellular respiration cannot fully oxidize glucose. It involves partial oxidation of sugar, using an organic molecule as the final electron acceptor.
Lactic acid fermentation: Produces lactic acid (homolactic or heterolactic).
Alcohol fermentation: Produces ethanol and CO2.
Industrial Uses of Fermentation
Fermentation End-Product(s) | Industrial Use | Starting Material | Microorganism |
|---|---|---|---|
Ethanol | Beer, wine | Starch, sugar | Saccharomyces cerevisiae |
Acetic Acid | Vinegar | Ethanol | Acetobacter |
Lactic Acid | Cheese, yogurt | Milk | Lactobacillus, Streptococcus |
Propionic Acid and CO2 | Swiss cheese | Lactic acid | Propionibacterium freudenreichii |
Acetone and Butanol | Pharmaceutical, industrial | Molasses | Clostridium acetobutylicum |
Citric Acid | Flavoring | Molasses | Aspergillus |
Methane | Fuel | Acetic acid | Methanosarcina |
Sorbose | Vitamin C | Sorbitol | Gluconobacter |
Other Catabolic and Anabolic Pathways
Lipid and Protein Catabolism
Lipids and proteins can be catabolized to provide energy and precursor metabolites for glycolysis and the Krebs cycle. Catabolism of amino acids produces ammonia and other nitrogenous wastes.
Anabolic Pathways
Anabolic reactions synthesize complex molecules from simpler ones, requiring energy and precursor metabolites. Many anabolic pathways are the reverse of catabolic pathways and are termed amphibolic if they can proceed in either direction.
Integration and Regulation of Metabolic Function
Regulatory Mechanisms
Cells regulate metabolism by controlling gene expression (amount and timing of enzyme production) and metabolic expression (activity of enzymes once produced). Feedback inhibition is essential for controlling anabolic pathways, preventing overproduction of metabolites.
