 Back
BackMicrobial Metabolism: Structure, Function, and Diversity
Study Guide - Smart Notes

Microbial Metabolism
Overview of Metabolism
Metabolism encompasses all biochemical reactions necessary for life, divided into two main categories: catabolism (energy-releasing breakdown) and anabolism (energy-requiring biosynthesis). These processes are fundamental to microbial growth, energy conservation, and cellular function.
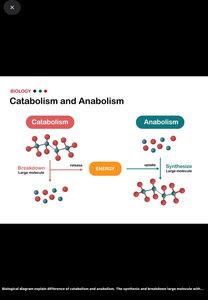
Catabolism: Breakdown of large molecules into smaller units, releasing energy.
Anabolism: Synthesis of large molecules from smaller precursors, requiring energy input.
Energy Conservation and ATP
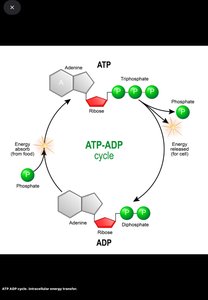
Cells conserve energy by converting it into ATP (adenosine triphosphate), which powers cellular work. The principle of energy conservation states that energy is neither created nor destroyed, but transformed and stored for cellular processes.
ATP-ADP Cycle: ATP is hydrolyzed to ADP, releasing energy for cellular activities. Energy from catabolic reactions is used to regenerate ATP from ADP and phosphate.
Electron Flow and Energy Conservation
Metabolism relies on electron transfer. Electron donors lose electrons, while electron acceptors gain electrons. The energy released during these transfers is conserved as ATP.
Key Principle: Electron flow is central to energy conservation in cells.
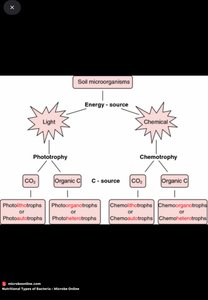
Energy Classes of Microorganisms
Microbes are classified based on their energy, electron, and carbon sources. This classification highlights their metabolic diversity and ecological roles.
By Energy Source:
Chemoorganotrophs: Use organic chemicals for energy.
Chemolithotrophs: Use inorganic chemicals (e.g., H2, H2S, NH4+).
Phototrophs: Use light as an energy source.
By Carbon Source:
Heterotrophs: Obtain carbon from organic compounds.
Autotrophs: Obtain carbon from CO2.
Enzymes and Catalysis
Enzymes are biological catalysts, mostly proteins, that lower the activation energy required for reactions. They are highly specific, not consumed in reactions, and do not alter equilibrium—only the reaction rate.
Activation Energy: Minimum energy needed to start a reaction.
Enzyme Structure: Substrate binds at the active site; catalysis depends on correct substrate positioning.
Helpers:
Prosthetic groups: Tightly bound (e.g., heme).
Coenzymes: Loosely bound, often vitamin derivatives.
Energy-Rich Storage Compounds
Microbes store energy in various compounds for long-term use.
Prokaryotes: Glycogen, PHB/PHA (lipid polymers), elemental sulfur.
Eukaryotes: Starch, lipids (fats).
Glycolysis, Fermentation, and Respiration
Microbes utilize different pathways to extract energy from glucose, depending on oxygen availability.
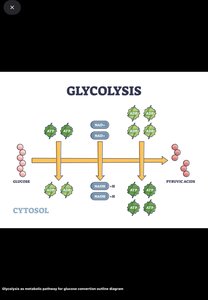
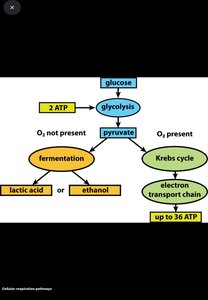
Glycolysis (EMP Pathway): Breaks glucose into pyruvate, producing 2 ATP via substrate-level phosphorylation.
Fermentation: Anaerobic; organic compound is both electron donor and acceptor; no electron transport chain.
Respiration: Uses electron transport chain; electron acceptor can be O2 (aerobic) or other compounds (anaerobic); produces more ATP than fermentation.
Electron Transport and Carriers
The Electron Transport System (ETS) is located in the cytoplasmic membrane and consists of a series of oxidation-reduction reactions that conserve energy by building a proton gradient.
Electron Carriers: NADH dehydrogenase, flavoproteins, iron–sulfur proteins, cytochromes, quinones.
Proton Motive Force (PMF)
Electron flow separates protons from electrons, creating a pH and electrical gradient across the membrane. PMF drives ATP synthase, making most cellular ATP.
Key Point: Most ATP is made via PMF, not glycolysis.

Alternative Energy Strategies
Microbes employ diverse strategies to generate energy, reflecting their adaptability and ecological roles.
Anaerobic Respiration: Uses electron acceptors other than O2; less energy than aerobic respiration; still uses ETS and PMF.
Chemolithotrophy: Inorganic electron donors; typically aerobic; often autotrophic; important in nutrient cycling.
Phototrophy: Light energy converted to ATP (photophosphorylation); photoautotrophs use CO2 as carbon source; photoheterotrophs use organic carbon.
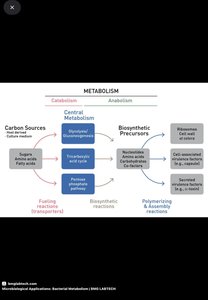
Sugars and Polysaccharide Biosynthesis
Microbes synthesize sugars and polysaccharides for cell wall construction and energy storage.
Activated Glucose Molecules: UDPG for cell wall sugars (NAG, NAM); ADPG for glycogen synthesis.
Gluconeogenesis: Synthesis of glucose from non-carbohydrates, starting with phosphoenolpyruvate.
Pentose Phosphate Pathway: Produces pentose sugars for nucleic acids; major source of NADPH for biosynthesis.
Amino Acid and Nucleotide Biosynthesis
Microbes build amino acids and nucleotides using carbon skeletons from glycolysis or the TCA cycle, incorporating nitrogen as ammonia.
Key Enzymes: Glutamine synthetase, glutamate dehydrogenase, transaminases.
Fatty Acid and Lipid Biosynthesis
Fatty acids are synthesized two carbons at a time, using acyl carrier protein (ACP). Adaptations in fatty acid composition occur in response to temperature.
Cold: Shorter, unsaturated fatty acids.
Heat: Longer, saturated fatty acids.
Domain Differences: Bacteria and Eukarya use fatty acids + glycerol; Archaea use phytanyl side chains.
Big Picture Summary
Metabolism: Integrates energy, electrons, and enzymes.
ATP: Universal energy currency.
PMF: Major driver of ATP synthesis.
Metabolic Diversity: Microbes exhibit vast metabolic capabilities.
Ecological Connection: Metabolism links energy flow, biosynthesis, and ecology.
Key Concepts and Definitions
Microbial Nutritional Types
Chemoorganotroph: Energy from organic compounds.
Chemolithotroph: Energy from inorganic compounds.
Chemotroph: Energy from chemicals.
Phototroph: Energy from light.
Troph: Means "to feed" or "to obtain nourishment".
Autotroph vs Heterotroph
Autotroph: Uses CO2 as carbon source.
Heterotroph: Uses organic compounds as carbon source.
Enzyme Structure and Function
Composition: Mostly proteins.
Temperature Sensitivity: High temperatures can denature proteins, causing loss of enzyme function.
Active Site: Substrate binds here.
Activation Energy: Minimum energy needed to start a chemical reaction.
Catabolism vs Anabolism
Catabolism: Breaks molecules down, releases energy.
Anabolism: Builds molecules, requires energy.
Respiration vs Fermentation
Respiration: Uses electron transport chain and external electron acceptor.
Fermentation: No ETC; organic molecule is both donor and acceptor.
End Products of Fermentation: Organic acids (lactic acid), alcohols (ethanol), gases (CO2, H2).
Yeast Used in Fermentation: Saccharomyces cerevisiae.
Glycolysis
Process: Glucose → pyruvate; produces 2 ATP and NADH; occurs in cytoplasm.
Glucose Transport: Via transport proteins in cytoplasmic membrane (facilitated diffusion or active transport).

ATP Production
Most ATP: Produced in the Electron Transport Chain (ETC), more than glycolysis or Krebs cycle.
Electron Acceptors
Aerobic Respiration: Final electron acceptor is O2, forms water.
Anaerobic Respiration: Final electron acceptors include nitrate (NO3-), sulfate (SO42-), carbon dioxide (CO2), ferric iron (Fe3+).
Autotrophs: Carbon and Energy Sources
Carbon Source: CO2.
Energy Source: Light (photoautotrophs) or inorganic chemicals (chemoautotrophs).
Bacterial Nutrient Acquisition
Sources: Soil, water, hosts, organic matter, inorganic chemicals.
Nitrogen Fixation
Process: Conversion of atmospheric nitrogen (N2) into ammonia (NH3).
Carried out by: Free-living bacteria (e.g., Azotobacter), symbiotic bacteria (e.g., Rhizobium), some cyanobacteria.
Summary Table: Microbial Metabolic Types
Type | Energy Source | Electron Source | Carbon Source |
|---|---|---|---|
Chemoorganotroph | Organic chemicals | Organic chemicals | Organic compounds |
Chemolithotroph | Inorganic chemicals | Inorganic chemicals | CO2 or organic compounds |
Phototroph | Light | Varies | CO2 (photoautotroph) or organic compounds (photoheterotroph) |
Summary Table: Respiration vs Fermentation
Process | Electron Transport Chain | Electron Acceptor | ATP Yield |
|---|---|---|---|
Respiration (Aerobic) | Yes | O2 | High |
Respiration (Anaerobic) | Yes | NO3-, SO42-, CO2, Fe3+ | Moderate |
Fermentation | No | Organic compound | Low |
Key Equations
ATP Hydrolysis:
Glycolysis (overall):
Fermentation (lactic acid):
Fermentation (ethanol):
Nitrogen Fixation:
Additional info:
Metabolic pathways are interconnected, allowing microbes to adapt to diverse environments.
Microbial metabolism is central to biogeochemical cycles and ecosystem functioning.
