 Back
BackMicrobial Metabolism: Structure, Function, and Pathways
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Metabolism
Microbial metabolism encompasses all chemical reactions and physical processes occurring within a microbial cell. These reactions are essential for energy production, synthesis of cellular components, and overall cell survival. Metabolism is divided into two main processes: anabolism (building up) and catabolism (breaking down).
Anabolism: Synthesis of complex molecules from simpler ones, requiring energy input.
Catabolism: Breakdown of complex molecules into simpler ones, releasing energy.
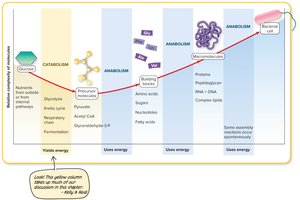
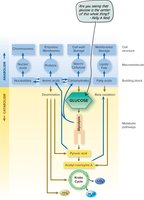
Additional info: The diagram illustrates the flow from nutrients through catabolic pathways to precursor molecules, which are then used in anabolic pathways to build macromolecules and eventually the bacterial cell.
Enzymes: Catalysts of Life
Structure and Function of Enzymes
Enzymes are biological catalysts that accelerate chemical reactions without being consumed or permanently altered. They provide a specific site (active site) for substrate binding, facilitating the transformation into products.
Simple enzymes: Consist of protein only.
Conjugated enzymes (holoenzymes): Contain a protein part (apoenzyme) and a nonprotein cofactor (either a coenzyme or a metallic ion).
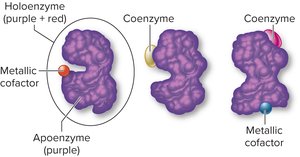
Additional info: Cofactors are essential for enzyme activity, often participating directly in the catalytic process.
Enzyme-Substrate Interactions
Enzymes bind substrates at their active sites, forming an enzyme-substrate complex. This interaction is highly specific, often described as a "lock-and-key" fit. The enzyme facilitates the reaction and releases the product, remaining unchanged and ready to catalyze subsequent reactions.
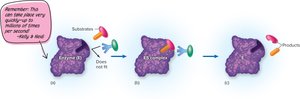
Cofactors and Coenzymes
Cofactors are nonprotein components required for enzyme activity. They can be:
Metal ions: Such as iron, magnesium, or zinc, which help stabilize enzyme structure or participate in catalysis.
Coenzymes: Organic molecules (often derived from vitamins) that transfer chemical groups between molecules.
Types of Enzymes: Constitutive vs. Regulated
Enzymes can be classified based on their regulation:
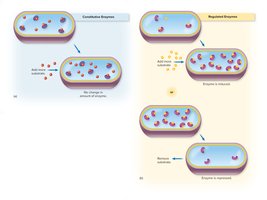
Constitutive enzymes: Produced continuously by the cell, regardless of substrate presence.
Regulated enzymes: Synthesized only in response to specific substrates or environmental conditions.
Control of Enzyme Activity
Cells regulate enzyme activity through several mechanisms:
Competitive inhibition: A molecule similar to the substrate competes for binding at the active site, blocking the actual substrate.
Noncompetitive inhibition: A regulatory molecule binds to a site other than the active site, altering enzyme conformation and reducing activity.
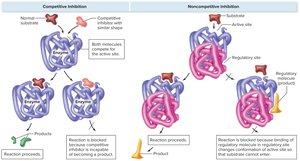
Control of Enzyme Synthesis
Enzyme synthesis can be regulated at the genetic level:
Enzyme repression: Excess end-product binds to DNA, inhibiting further enzyme production.
Enzyme induction: Enzymes are produced only when their substrate is present.
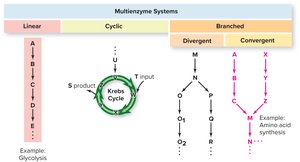
Patterns of Metabolic Pathways
Types of Pathways
Metabolic pathways can be organized in several patterns:
Linear: Sequential reactions (e.g., glycolysis).
Cyclic: The starting molecule is regenerated (e.g., Krebs cycle).
Branched: Pathways diverge or converge, allowing flexibility in metabolism (e.g., amino acid synthesis).
Energy and Redox Reactions in Cells
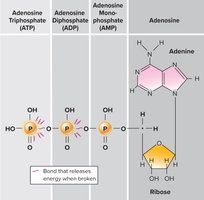
ATP: The Energy Currency
Cells store and transfer energy using adenosine triphosphate (ATP). ATP consists of adenine, ribose, and three phosphate groups. The high-energy bonds between phosphate groups release energy when broken, fueling cellular processes.
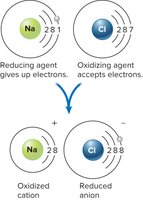
Oxidation-Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons between molecules, essential for energy extraction from nutrients. Oxidation is the loss of electrons, while reduction is the gain of electrons. These reactions are catalyzed by oxidoreductases and involve electron carriers such as NAD+ and FAD.
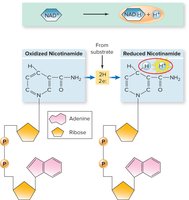
Catabolic Pathways and ATP Yield
Overview of Catabolic Pathways
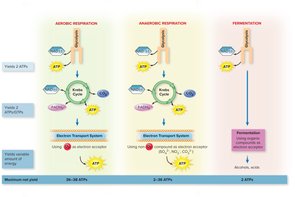
Microbes utilize three main catabolic pathways to extract energy from glucose:
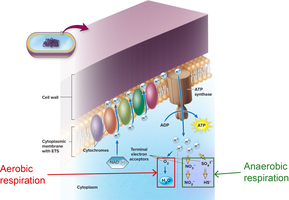
Aerobic respiration: Glycolysis, Krebs cycle, and electron transport chain; oxygen is the final electron acceptor; yields 36–38 ATP per glucose.
Anaerobic respiration: Similar to aerobic but uses non-oxygen electron acceptors (e.g., nitrate, sulfate); yields 2–36 ATP per glucose.
Fermentation: Incomplete oxidation of glucose; organic molecules are final electron acceptors; yields 2 ATP per glucose.
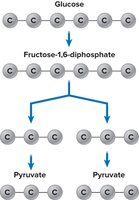
Glycolysis
Glycolysis is the first step in all three pathways, converting glucose (6C) into two molecules of pyruvate (3C), generating ATP and NADH.
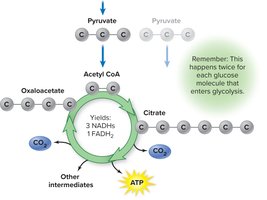
Krebs Cycle (Citric Acid Cycle)
The Krebs cycle processes acetyl CoA, producing NADH, FADH2, ATP, and CO2. It is a cyclic pathway that also provides intermediates for biosynthesis.
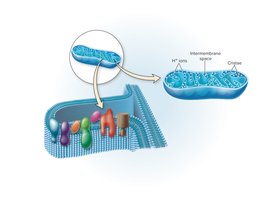
Electron Transport Chain (ETC)
The ETC is a series of redox carriers embedded in the membrane. Electrons from NADH and FADH2 are transferred through the chain, driving proton pumps that generate a proton motive force used by ATP synthase to produce ATP. Oxygen (aerobic) or other inorganic molecules (anaerobic) serve as terminal electron acceptors.
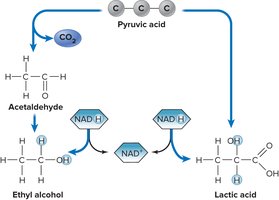
Fermentation
Fermentation allows cells to generate ATP in the absence of oxygen by using organic molecules as electron acceptors. Common products include ethanol, lactic acid, and other organic acids.
Anabolism and Amphibolism
Amphibolic Pathways
Amphibolism refers to the integration of catabolic and anabolic pathways, allowing cells to efficiently use intermediates for both energy production and biosynthesis. For example, intermediates from glycolysis and the Krebs cycle can be diverted to synthesize amino acids, nucleotides, and lipids.
Anabolic Pathways
Anabolic reactions use energy and intermediates from catabolism to build macromolecules:
Carbohydrate biosynthesis: Formation of polysaccharides, cell wall components, and storage molecules.
Protein synthesis: Assembly of amino acids into proteins, enzymes, and structural components.
Nucleic acid synthesis: Production of DNA and RNA for genetic continuity and protein synthesis.
Lipid synthesis: Formation of fatty acids and complex lipids for membranes and storage.
Additional info: The cell's genetic makeup determines its biosynthetic capabilities, influencing its nutritional requirements and ecological niche.
