 Back
BackAcid-Base Reactions in Organic Chemistry: Principles and Applications
Study Guide - Smart Notes

Chapter 2: Acid-Base Reactions
Brønsted-Lowry Acid-Base Theory
The Brønsted-Lowry theory is foundational in organic chemistry, describing acids as proton donors and bases as proton acceptors. Acid-base reactions are proton-transfer processes, and every such reaction involves both an acid and a base. Water, an amphiprotic species, can act as either an acid or a base depending on the reactants.
Acid: Species that donates a proton (H+).
Base: Species that accepts a proton.
Conjugate Acid-Base Pair: When an acid loses a proton, it forms its conjugate base; when a base gains a proton, it forms its conjugate acid.
Strength Relationship: The stronger the acid, the weaker its conjugate base.
Measuring Acid Strength: Ka and pKa
The strength of an acid in water is quantified by its acid dissociation constant (Ka) and its logarithmic counterpart, pKa. Stronger acids have higher Ka and lower pKa values, indicating a greater tendency to ionize in water.
Ka: Acid dissociation constant, measures the extent of ionization.
pKa:
Thermodynamic Favorability: Lower pKa means more favorable ionization ().
Acid | Name | Ka | pKa |
|---|---|---|---|
HCl | hydrogen chloride | -7 | |
CH3COOH | acetic acid | 4.76 | |
H2O | water | 15.7 | |
NH3 | ammonia | 36 | |
CH3CH3 | ethane | 60 |
Predicting Acid-Base Reaction Favorability
The position of equilibrium in acid-base reactions is determined by the relative strengths of acids and bases involved. The reaction favors the side with the weaker acid and weaker base, as these species are more thermodynamically stable.
Equilibrium Position: Favors formation of weaker acid and weaker base.
Application: Use pKa values to predict equilibrium direction for reactions not involving water as the base.
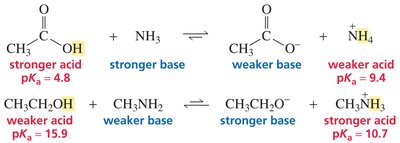
Structural Effects on Acid Strength
The strength of an acid is influenced by the stability of its conjugate base, which is affected by several structural features:
Electronegativity
Within a period, acidity increases with electronegativity. A more electronegative atom stabilizes the negative charge on the conjugate base, increasing acid strength.
Trend: C < N < O < F (most electronegative)
Acidity: CH4 < NH3 < H2O < HF (strongest acid)
Conjugate Base Stability: CH3- < NH2- < HO- < F- (most stable)
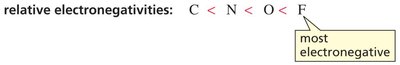
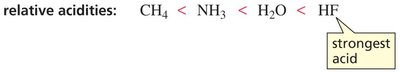
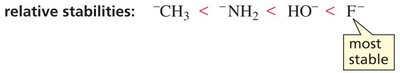
Hybridization
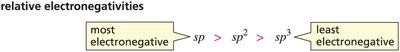
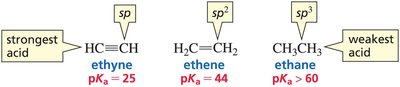
The effective electronegativity of an atom increases with the percentage of s character in its hybrid orbitals. Acidity increases as the s character increases (sp > sp2 > sp3).
sp: 50% s character (most acidic)
sp2: 33% s character
sp3: 25% s character (least acidic)
Atomic Size
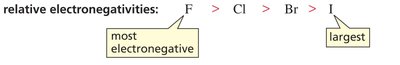
Down a group, acidity increases with atomic size. Larger atoms stabilize negative charge better by spreading it over a greater volume.
Electronegativity Trend: F > Cl > Br > I
Acidity Trend: HF < HCl < HBr < HI (HI is strongest acid)
Conjugate Base Stability: F- < Cl- < Br- < I- (I- is most stable)
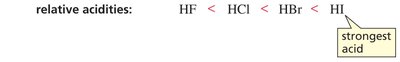
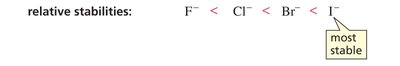
Inductive Effects
Electron-withdrawing groups (EWGs) stabilize the conjugate base by pulling electron density away from the negatively charged site, increasing acidity. The effect diminishes with distance from the acidic hydrogen.
EWGs: Groups like Cl, NO2, CF3 increase acidity.
Distance Effect: Inductive stabilization is strongest when the EWG is close to the acidic hydrogen.
Resonance
Resonance stabilization increases the stability of the conjugate base, thereby increasing acid strength. Delocalization of charge over multiple atoms leads to greater stability.
Example: Acetate ion (CH3COO-) is resonance-stabilized, making acetic acid much stronger than ethanol, which lacks resonance stabilization.
Resonance Hybrid: The actual structure is a hybrid of all possible resonance forms.
Effect of Solution pH on Protonation State
The predominant form of a compound at a given pH depends on the relationship between the pH and the pKa of the acid:
pH < pKa: Acidic form predominates (protonated).
pH > pKa: Basic form predominates (deprotonated).
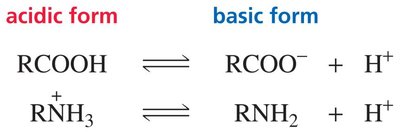
Example: At pH 1, carboxylic acids are mostly in the protonated (acidic) form; at pH 14, they are mostly in the deprotonated (basic) form. Amines are protonated at low pH and deprotonated at high pH.
Additional info: These principles are essential for understanding reactivity, mechanisms, and biological function in organic chemistry.
