 Back
BackAcid Halides, Anhydrides, and Esters: Preparation and Reactions
Study Guide - Smart Notes

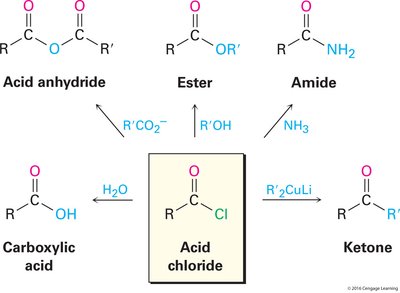
Acid Halides and Their Chemistry
Preparation of Acid Halides
Acid halides, particularly acid chlorides, are important intermediates in organic synthesis. They are typically prepared from carboxylic acids using reagents such as thionyl chloride (SOCl2) or phosphorus tribromide (PBr3).
Acid Chlorides: Prepared by reacting carboxylic acids with SOCl2 in dichloromethane or PBr3 in ether.
Mechanism: The carboxylic acid reacts with SOCl2 to form the acid chloride, releasing SO2 and HCl as byproducts.
Reactivity and General Reactions of Acid Halides
Acid chlorides are the most reactive carboxylic acid derivatives, making them versatile for various transformations.
Hydrolysis: Acid chlorides react readily with water to yield carboxylic acids. This is often an unwanted side reaction.
Anhydride Formation: Reaction with carboxylate ions produces anhydrides, both symmetrical and unsymmetrical.
Ester Formation: Acid chlorides react with alcohols in the presence of a base (e.g., pyridine) to form esters. Steric hindrance affects the reaction rate, with primary alcohols being most reactive.
Amide Formation: Reaction with ammonia or primary/secondary amines yields amides. Two equivalents of amine are required, or the Schotten-Baumann method can be used with NaOH.
Reduction: LiAlH4 reduces acid chlorides to primary alcohols. Less reactive agents (e.g., LiAlH(OtBu)3) can stop at the aldehyde stage (Rosenmund reduction).
Organometallic Reactions: Grignard reagents (2 equiv) convert acid chlorides to tertiary alcohols, while organocuprates (R'2CuLi) yield ketones.
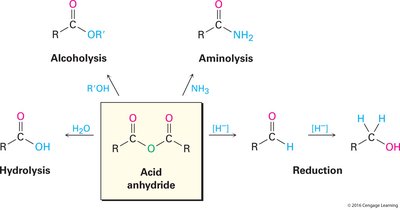
Anhydrides: Preparation and Reactions
Preparation of Anhydrides
Anhydrides are less commonly prepared directly from carboxylic acids due to harsh conditions required.
Cyclic Anhydrides: Five- and six-membered cyclic anhydrides can be synthesized from diacids by heating.
Acid Chloride Method: More commonly, anhydrides are prepared by reacting acid chlorides with carboxylate ions, allowing for both symmetrical and unsymmetrical anhydrides.
Reactions of Anhydrides
Anhydrides undergo reactions similar to acid chlorides, including hydrolysis, alcoholysis, aminolysis, and reduction.
Hydrolysis: Reaction with water yields carboxylic acids.
Alcoholysis: Reaction with alcohols forms esters.
Aminolysis: Reaction with ammonia or amines produces amides.
Reduction: Reduction with hydride reagents yields alcohols.
Efficiency: Only half of the anhydride is used in these reactions; the other half acts as the leaving group. Acid chlorides are generally more efficient for these transformations, except for simple anhydrides.
Esters: Preparation and Reactions
Preparation of Esters
Esters can be synthesized from carboxylic acids under both basic and acidic conditions.
Basic Conditions (SN2): Carboxylic acids are deprotonated with NaH, then reacted with alkyl halides to form esters.
Acidic Conditions (Fischer Esterification): Carboxylic acids react with excess alcohol and acid catalyst (HCl or H2SO4) to form esters. This method is limited to simple alcohols.
Reactions of Esters
Esters are less reactive than acid chlorides or anhydrides but still undergo nucleophilic acyl substitution.
Hydrolysis: Can occur under basic (saponification) or acidic conditions. Basic hydrolysis is more common and requires an acidic workup.
Amide Formation: Esters react with ammonia or amines to form amides, though acid chlorides are preferred for higher yields.
Reduction: Esters are reduced by LiAlH4 (not NaBH4) to primary alcohols. DIBAH can stop the reduction at the aldehyde stage.
Grignard Addition: Esters and lactones react with two equivalents of Grignard or organolithium reagents to give tertiary alcohols. The reaction cannot be stopped at the ketone stage.
Summary Table: Reactivity and Transformations of Carboxylic Acid Derivatives
Derivative | Preparation | Main Reactions |
|---|---|---|
Acid Chloride | SOCl2 or PBr3 from carboxylic acid | Hydrolysis, Anhydride formation, Esterification, Amide formation, Reduction, Grignard addition |
Anhydride | Acid chloride + carboxylate ion; heating diacids | Hydrolysis, Alcoholysis, Aminolysis, Reduction |
Ester | Fischer esterification; SN2 with alkyl halide | Hydrolysis, Amide formation, Reduction, Grignard addition |
Key Concepts and Definitions
Acid Chloride: A carboxylic acid derivative with a chloride substituent, highly reactive in nucleophilic acyl substitution.
Anhydride: A compound formed from two carboxylic acids with loss of water, used in ester and amide synthesis.
Ester: A carboxylic acid derivative formed by replacing the hydroxyl group with an alkoxy group.
Amide: A carboxylic acid derivative formed by replacing the hydroxyl group with an amino group.
Nucleophilic Acyl Substitution: The fundamental mechanism for reactions of carboxylic acid derivatives, involving attack by a nucleophile and substitution of the leaving group.
Important Equations
Acid Chloride Formation:
Fischer Esterification:
Amide Formation from Acid Chloride:
Reduction of Acid Chloride:
Grignard Addition to Acid Chloride:
Additional info:
Selective esterification is possible due to steric effects; primary alcohols react fastest.
Acid chlorides are generally preferred for amide and ester synthesis due to higher reactivity and efficiency.
Basic hydrolysis of esters (saponification) is widely used in soap production.
