 Back
BackAldehydes and Ketones: Nucleophilic Addition and Properties
Study Guide - Smart Notes

Aldehydes and Ketones: Structure and Nomenclature
Carbonyl Group Structure
The carbonyl group (C=O) is a central feature of both aldehydes and ketones. It consists of one σ-bond formed by the overlap of sp2-hybridized orbitals and one π-bond formed by the overlap of parallel 2p-orbitals. The oxygen atom has two lone pairs in sp2-hybridized orbitals, while the carbon atom forms bonds with other atoms.
σ-bond: Overlap of sp2 orbitals
π-bond: Overlap of 2p orbitals
Lone pairs: Oxygen has two lone pairs
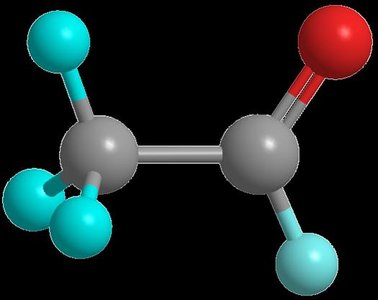
Aldehyde Structure and Nomenclature
An aldehyde is characterized by a carbonyl group bonded to at least one hydrogen atom. The simplest aldehyde, methanal (formaldehyde), has the carbonyl group attached to two hydrogens. Other aldehydes have the carbonyl group attached to one hydrogen and one carbon atom.
Formaldehyde: Methanal, C=O bonded to two H atoms
Acetaldehyde: Ethanal, C=O bonded to one H and one methyl group
Naming: IUPAC names use the suffix -al (e.g., propanal, butanal)
Aromatic aldehydes: Benzaldehyde is the accepted name for benzenecarbaldehyde
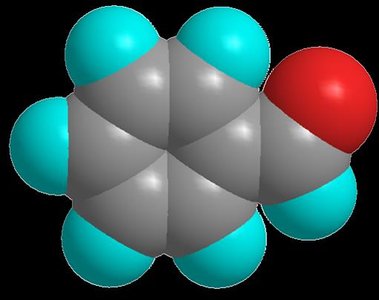
Ketone Structure and Nomenclature
A ketone has a carbonyl group bonded to two carbon atoms. The simplest ketone is acetone (propanone), with the carbonyl group attached to two methyl groups. Ketones are named using the suffix -one (e.g., propanone, butanone).
Acetone: Propanone, C=O bonded to two methyl groups
Acetophenone: 1-Phenylethanone, C=O bonded to a phenyl and methyl group
Benzophenone: Diphenylmethanone, C=O bonded to two phenyl groups
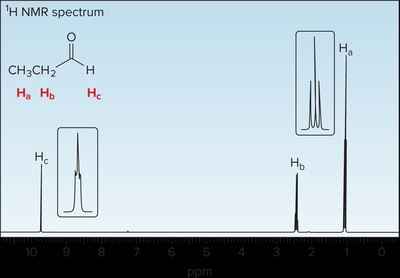
Spectroscopic Properties of Aldehydes and Ketones
Infrared (IR) Spectroscopy
Aldehydes and ketones exhibit characteristic IR absorptions:
Aldehyde C-H stretch: 2700–2830 cm-1 (two peaks)
Carbonyl (C=O) stretch: ~1700 cm-1 (frequency increases with decreasing ring size)
Conjugated C=O: 1680 cm-1
NMR Spectroscopy
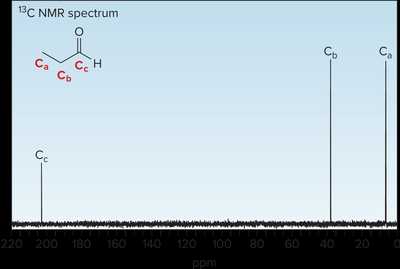
1H NMR: Aldehyde protons appear at 9–10 ppm, methyl/methylene protons at 2–2.5 ppm. 13C NMR: Carbonyl carbons appear at 190–215 ppm.
Biologically and Industrially Important Aldehydes and Ketones
Geranial (Citral)
Geranial is a component of essential oils, such as lemongrass oil (65–85%). It is a highly conjugated aldehyde used in fragrances and flavorings.
Cortisone
Cortisone is an adrenocorticoid hormone (glucocorticoid) that regulates carbohydrate metabolism and reduces inflammation.

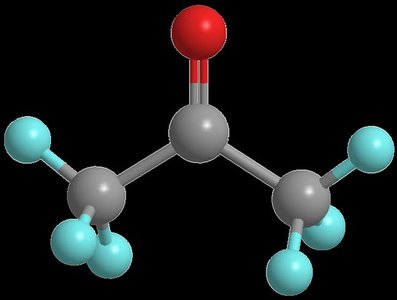
Preparation of Aldehydes and Ketones
Preparation from Carboxylic Acids
Aldehydes can be prepared by partial reduction of carboxylic acids using reagents such as LiAlH4 followed by water, or by oxidation of primary alcohols using PCC in CH2Cl2.
Reduction:
Oxidation:
Preparation of Ketones
Ketones can be synthesized by oxidation of secondary alcohols or by addition of Grignard reagents to aldehydes followed by dehydration.
Oxidation:
Grignard addition:
Reactivity of Aldehydes and Ketones: Nucleophilic Addition
General Mechanism
The most common reaction of aldehydes and ketones is nucleophilic addition to the carbonyl carbon. The nucleophile attacks the electrophilic carbon, breaking the π-bond and forming a tetrahedral intermediate.
Electrophilic carbon: Partial positive charge (δ+)
Nucleophile: Adds to carbonyl carbon
Tetrahedral intermediate: sp3-hybridized carbon
Acid-Catalyzed Nucleophilic Addition
Protonation of the carbonyl oxygen increases the electrophilicity of the carbonyl carbon, making it more reactive toward nucleophiles. Weak nucleophiles require protonation before attack.
Oxonium ion: Formed by protonation
Carbocation: Stabilized intermediate
Nucleophilic Addition of Cyanide: Cyanohydrin Formation
Hydrogen cyanide (HCN) adds to the carbonyl group of aldehydes and ketones to form cyanohydrins. The reaction is reversible under basic conditions, and cyanohydrins can be hydrolyzed to carboxylic acids.
Mechanism: Cyanide ion adds to carbonyl carbon, forming a tetrahedral intermediate, which is then protonated.
Hydrolysis: Cyanohydrins can be converted to carboxylic acids.
The Wittig Reaction
The Wittig reaction converts a carbonyl group (C=O) to an alkene (C=C) by forming two new C–C bonds. The Wittig reagent (ylide) is prepared by SN2 reaction of triphenylphosphine and an alkyl halide, followed by deprotonation with a strong base.
Ylide: A species with two charged atoms bonded together
Mechanism: Nucleophilic attack forms oxaphosphetane, elimination of Ph3P=O forms alkene
Isomer ratio: Both E and Z isomers can form
Addition of Amines: Imine and Enamine Formation
Primary Amines: Imine Formation
Primary amines react with aldehydes and ketones to form imines (Schiff bases) via nucleophilic addition followed by elimination of water. The reaction is fastest under mildly acidic conditions.
Carbinolamine intermediate: Formed by nucleophilic attack
Imine: Formed after loss of water
Application: Chemistry of vision (rhodopsin formation)
Secondary Amines: Enamine Formation
Secondary amines react with aldehydes and ketones to give enamines. Hydrolysis of enamines regenerates the original carbonyl compound.
Enamine: Formed by nucleophilic addition and elimination
Hydrolysis: Returns to aldehyde or ketone
Addition of Water and Alcohols: Hydration and Acetal Formation
Hydration
Water adds to the carbonyl group to form hydrates. The equilibrium amount of hydrate decreases as the number of alkyl groups on the carbonyl carbon increases.
Chloral hydrate: Used as a sedative
Acetal Formation
Alcohols add to aldehydes and ketones to form acetals (from aldehydes) or ketals (from ketones). Two equivalents of alcohol are required, and the reaction is acid-catalyzed.
Hemiacetal: Intermediate formed after addition of one alcohol
Acetal: Formed after addition of second alcohol
Cyclic acetals: Formed from diols
Acetals as protecting groups: Used to protect carbonyls during synthesis
Hydrolysis of Acetals
Acetals can be hydrolyzed back to aldehydes or ketones by treatment with aqueous acid. The process is reversible and involves conversion of acetal to hemiacetal, then to carbonyl compound.
Cyclic Hemiacetals
Cyclic hemiacetals, also called lactols, are stable five- and six-membered ring compounds formed by intramolecular cyclization of hydroxy aldehydes.
Formation: Nucleophilic addition of hydroxy group to carbonyl group
Stability: Five- and six-membered rings are particularly stable
