 Back
BackAlkanes and Cycloalkanes: Structure, Nomenclature, and Conformations
Study Guide - Smart Notes

Alkanes and Cycloalkanes
Introduction to Alkanes
Alkanes are saturated hydrocarbons composed solely of hydrogen and carbon atoms. They are characterized by single bonds between carbon atoms and have the general formula CnH2n+2. Alkanes can exist as straight-chain, branched, or cyclic compounds.
Saturated hydrocarbons: Only single bonds between carbons.
Unsaturated hydrocarbons: Contain double or triple bonds (not covered in this chapter).
IUPAC Nomenclature of Alkanes
The International Union of Pure and Applied Chemistry (IUPAC) system provides a systematic method for naming alkanes. The name consists of the parent chain, substituents, and their locations.
Parent chain: The longest continuous chain of carbon atoms.
Substituents: Groups attached to the parent chain, named as alkyl groups (ending in -yl).
Location: Number the parent chain to give substituents the lowest possible locants.
Steps for Naming Alkanes
Identify the longest carbon chain (parent chain).
Name and identify all substituents.
Number the parent chain to assign the lowest locants to substituents.
List substituents in alphabetical order, using prefixes (di-, tri-, etc.) for multiples.
Special Cases in Nomenclature
Cyclic compounds: Add the prefix "cyclo" to the parent name.
Complex substituents: Number the longest chain within the substituent, starting from the attachment point.
Bicyclic compounds: Use the prefix "bicyclo" and count carbons in each bridge between bridgehead carbons.
Constitutional Isomers
Constitutional isomers are compounds with the same molecular formula but different connectivity of atoms. The number of possible isomers increases with the number of carbon atoms.
Recognition: Isomers have different IUPAC names or cannot be superimposed by rotation.
Relative Stability of Isomeric Alkanes
The stability of isomeric alkanes can be compared by measuring their heats of combustion. Lower heat of combustion per CH2 group indicates greater stability.
Sources and Uses of Alkanes
Alkanes are major components of petroleum and natural gas. They are separated by distillation and used as fuels and chemical feedstocks. Processes such as cracking and reforming increase the yield of gasoline from petroleum.
Conformational Analysis of Alkanes
Alkanes can adopt multiple 3-D shapes (conformations) due to rotation around single bonds. Newman projections are used to visualize these conformations.
Staggered conformation: Lower energy, more stable.
Eclipsed conformation: Higher energy, less stable due to torsional strain.
Dihedral angle: The angle between atoms on adjacent carbons; 60° in staggered ethane.
Energetics of Conformations
Ethane: Staggered is 12 kJ/mol more stable than eclipsed.
Propane: Barrier to rotation is 14 kJ/mol; CH3-H eclipsing interactions contribute more strain.
Butane: Gauche interactions (methyl groups 60° apart) cause steric strain; anti conformation is most stable.
Cycloalkanes: Structure and Strain
Cycloalkanes are ring-shaped alkanes. Their stability depends on ring size and the presence of angle and torsional strain.
Ideal bond angle: 109.5° for sp3 hybridized carbon.
Angle strain: Deviation from ideal bond angles.
Torsional strain: Eclipsing interactions in the ring.
Stability of Cycloalkanes
Cyclohexane: Most stable, adopts a chair conformation with no ring strain.
Cyclobutane: Significant angle strain (88° bond angles), puckered to reduce torsional strain.
Cyclopentane: Minimal angle strain, envelope conformation to reduce torsional strain.
Conformations of Cyclohexane
Cyclohexane can adopt several conformations, but the chair conformation is the most stable due to minimal angle and torsional strain. Each carbon has two substituents: one axial (up/down) and one equatorial (off the ring, parallel to chair lines).
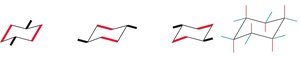
Monosubstituted Cyclohexane
When cyclohexane has one substituent, two chair conformations are possible due to ring flipping. The conformation with the substituent in the equatorial position is more stable, as axial substituents cause steric (1,3-diaxial) interactions.
Disubstituted Cyclohexane
With two substituents, the relative stability of chair conformations depends on the positions (axial/equatorial) of each group. Solid and dashed wedges indicate the orientation of substituents on the ring.
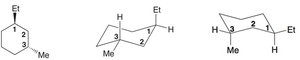
cis-trans Stereoisomerism in Cycloalkanes
Disubstituted cycloalkanes can exist as cis (same side) or trans (opposite sides) stereoisomers. These isomers have different physical and chemical properties and are best represented using chair conformations.
Polycyclic Systems
Polycyclic compounds contain multiple fused rings. Decalin is an example of two fused cyclohexane rings. Many natural products, such as camphor and camphene, have polycyclic structures.
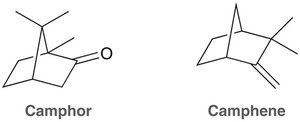
Camphor: A fragrant natural product with a ketone functional group.
Camphene: A bicyclic hydrocarbon found in essential oils.
Summary of Key Concepts
Alkanes and cycloalkanes are fundamental classes of organic compounds.
IUPAC nomenclature provides a systematic way to name these compounds.
Conformational analysis explains the stability of different shapes due to strain.
Cyclohexane's chair conformation is a model for understanding ring stability and substituent effects.
Polycyclic systems are important in natural products and materials chemistry.
Additional info: Tables referenced in the original material (e.g., parent names, alkyl groups, energy costs) are not included here but should be reviewed in the textbook for comprehensive study.
