 Back
BackAlkanes and Cycloalkanes: Structure, Nomenclature, and Conformations
Study Guide - Smart Notes

Alkanes and Cycloalkanes
Introduction to Alkanes and Cycloalkanes
Alkanes and cycloalkanes are fundamental classes of hydrocarbons in organic chemistry. Alkanes are saturated hydrocarbons containing only single bonds, while cycloalkanes are ring-shaped saturated hydrocarbons. Understanding their structure, nomenclature, and conformational analysis is essential for further study in organic chemistry.
Nomenclature of Alkanes
Parent Names and Alkyl Substituents
The systematic naming of alkanes is governed by the IUPAC system. The parent name is determined by the longest continuous chain of carbon atoms, and substituents are named as alkyl groups derived from the parent alkane by removing one hydrogen atom.
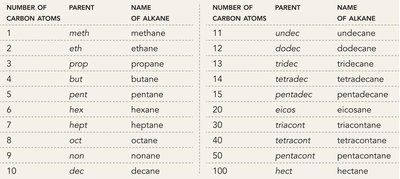
Parent Names: Methane (1C), Ethane (2C), Propane (3C), Butane (4C), Pentane (5C), Hexane (6C), Heptane (7C), Octane (8C), Nonane (9C), Decane (10C), and so on.
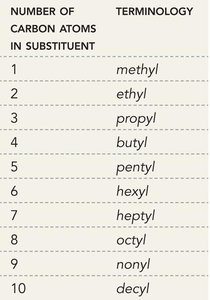
Alkyl Substituents: Methyl (1C), Ethyl (2C), Propyl (3C), Butyl (4C), etc.
Cycloalkanes
Cycloalkanes are named by adding the prefix "cyclo-" to the parent alkane name. The number of carbons in the ring determines the name: cyclopropane (3C), cyclobutane (4C), cyclopentane (5C), etc.
Cyclopropane: 3-membered ring
Cyclobutane: 4-membered ring
Cyclopentane: 5-membered ring

Rules for Selecting the Parent Chain
Choose the longest continuous chain of carbon atoms as the parent chain.
If multiple chains of equal length exist, select the one with the most substituents.
If the parent chain is cyclic, use the "cyclo-" prefix.
Naming Substituents
Substituents are named by replacing the "-ane" ending of the parent alkane with "-yl." For example, methane becomes methyl, ethane becomes ethyl, etc.
Complex Substituents
Branched alkyl groups are named by identifying the longest chain within the substituent, numbering from the point of attachment, and naming side groups as prefixes.
Numbering and Assembling the IUPAC Name
Numbering the Parent Chain
Number the parent chain to give the first substituent the lowest possible number.
If there is a tie, assign the lowest number to the next substituent in alphabetical order.
For cycloalkanes, the same rules apply, starting at a substituent and proceeding to give the lowest set of numbers.

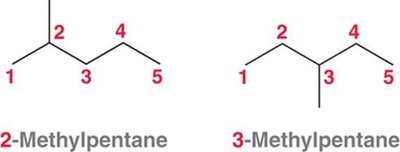
Alphabetizing Substituents
List substituents in alphabetical order, ignoring prefixes like di-, tri-, sec-, tert- (but not iso- or cyclo-).
Use hyphens to separate numbers from letters and commas to separate numbers.





Conformations of Alkanes
Bond Rotation and Representations
Single bonds in alkanes can rotate, leading to different spatial arrangements called conformations. The most common representations are wedge-and-dash, sawhorse, and Newman projections.
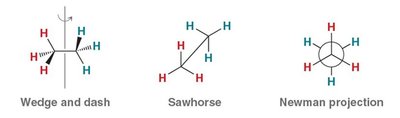
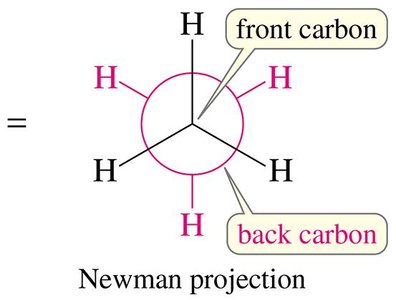
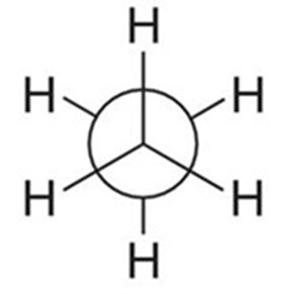
Newman Projections
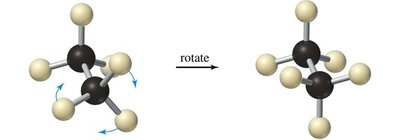
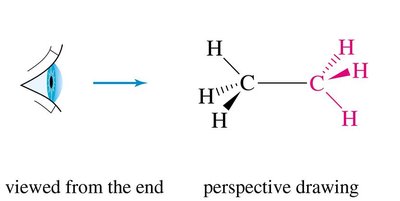
Newman projections are used to visualize the spatial relationship of bonds on adjacent carbons by looking straight down a C–C bond. The front carbon is represented as a point, and the back carbon as a circle.
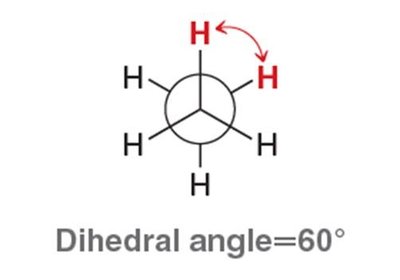
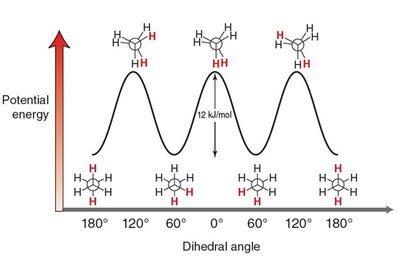
Conformational Analysis: Ethane
Ethane has two key conformations:
Staggered: Lowest energy, most stable. All C–H bonds on adjacent carbons are as far apart as possible.
Eclipsed: Highest energy, least stable. C–H bonds on adjacent carbons are aligned, leading to torsional strain.
The energy difference between staggered and eclipsed conformations is about 12 kJ/mol.
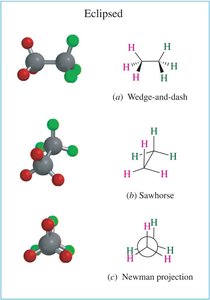
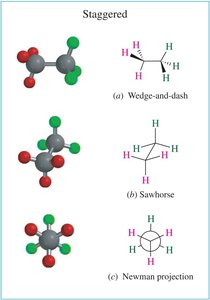
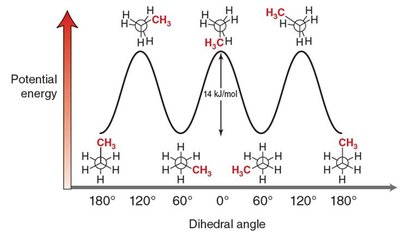
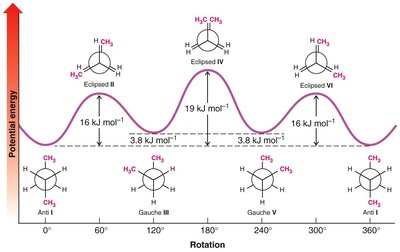
Conformational Analysis: Butane
Butane has several possible conformations due to rotation about the C2–C3 bond:
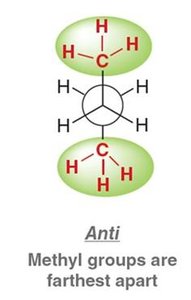
Anti (staggered): Methyl groups are 180° apart, lowest energy, most stable.
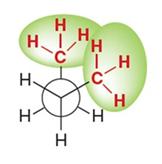
Gauche (staggered): Methyl groups are 60° apart, higher energy due to steric strain.
Eclipsed: Methyl groups are aligned, highest energy, least stable.
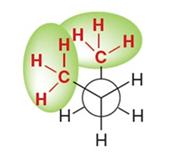
Cycloalkane Conformations and Ring Strain
Ring Strain in Cycloalkanes
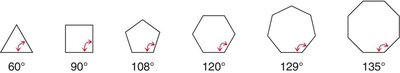
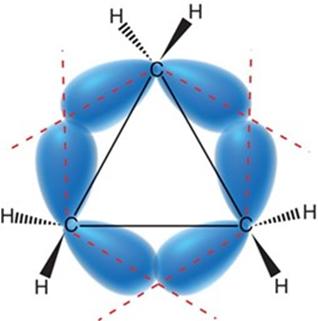
Cycloalkanes experience ring strain due to deviations from ideal bond angles (109.5° for sp3 carbons) and torsional strain from eclipsing interactions.
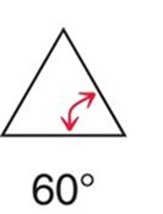
Cyclopropane: 60° bond angles, significant angle and torsional strain.
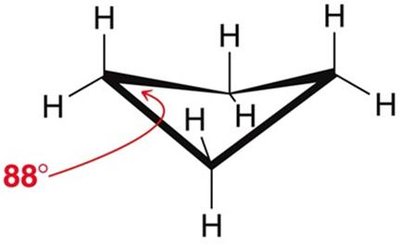
Cyclobutane: 88° bond angles, less angle strain than cyclopropane, but still significant torsional strain.
Cyclopentane: Nearly ideal bond angles, minimal angle strain, slight torsional strain.
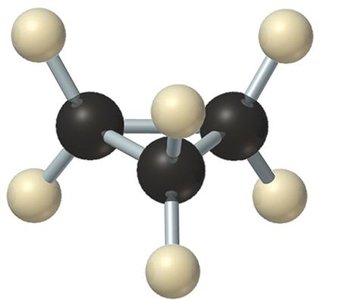
Cyclohexane Conformations
Cyclohexane adopts several conformations, with the chair conformation being the most stable due to the absence of angle and torsional strain. Each carbon in the ring has two substituents: one axial (up or down, perpendicular to the ring) and one equatorial (slightly off the ring, roughly in the plane).
Chair conformation: Most stable, all bond angles are 109.5°, all adjacent C–H bonds are staggered.
Boat and twist-boat conformations: Less stable due to steric and torsional strain.
Axial and Equatorial Positions
In the chair conformation, substituents can occupy axial or equatorial positions. Larger substituents prefer the equatorial position to minimize steric interactions (1,3-diaxial interactions).
Ring Flipping
Cyclohexane can undergo a ring flip, interconverting axial and equatorial positions for each substituent. The up/down orientation of substituents does not change during a ring flip.
Monosubstituted and Disubstituted Cyclohexanes
Monosubstituted: The conformation with the substituent in the equatorial position is more stable.
Disubstituted: Use cis/trans nomenclature. The most stable conformation places the larger group equatorial.
Summary Table: Parent Names and Alkyl Substituents
Number of Carbons | Parent Name | Name as Alkane | Name as Substituent |
|---|---|---|---|
1 | meth | methane | methyl |
2 | eth | ethane | ethyl |
3 | prop | propane | propyl |
4 | but | butane | butyl |
5 | pent | pentane | pentyl |
6 | hex | hexane | hexyl |
7 | hept | heptane | heptyl |
8 | oct | octane | octyl |
9 | non | nonane | nonyl |
10 | dec | decane | decyl |
Key Equations and Concepts
General formula for alkanes:
General formula for cycloalkanes:
Dihedral angle: The angle between two bonds on adjacent atoms, important for conformational analysis.
Practice Problems
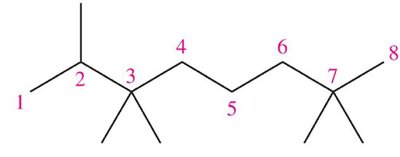
Name the following structure:
Draw the Newman projection for butane in the anti conformation.
Draw the chair conformation of methylcyclohexane and indicate the more stable conformation.
Additional info: This guide covers the essential nomenclature and conformational analysis of alkanes and cycloalkanes, foundational for understanding more complex organic molecules and reactions.
