 Back
BackAlkanes and Cycloalkanes: Structure, Nomenclature, and Conformations
Study Guide - Smart Notes

Alkanes and Cycloalkanes: Structure and Nomenclature
Introduction to Alkanes and Cycloalkanes
Alkanes and cycloalkanes are classes of hydrocarbons in which all carbon-carbon (C–C) bonds are single bonds. These compounds are fundamental in organic chemistry due to their structural simplicity and prevalence in natural products and synthetic materials.
Alkanes: Saturated hydrocarbons with the general formula .
Cycloalkanes: Saturated hydrocarbons containing rings, with the general formula .
Hydrocarbons with double bonds are called alkenes (), and those with triple bonds are alkynes ().
Naming Cycloalkanes
Cycloalkanes are named by attaching the prefix cyclo- to the name of the corresponding alkane. For monocyclic cycloalkanes (one ring), the rules are as follows:
Identify the parent ring and name it as a cycloalkane.
Number the ring to give substituents the lowest possible numbers.
For substituted cycloalkanes, the carbon bearing the highest-priority group (e.g., OH) gets the lowest number.
Example: For a cyclohexane ring with methyl and hydroxyl substituents, number the ring so that the OH group gets the lowest possible number, even if this does not give the lowest set of numbers overall.
Cycloalkylalkanes
When a cycloalkane ring is attached to an alkyl chain, the parent structure is chosen based on the number of carbons:
If the ring has more carbons than the chain, the ring is the parent.
If the chain has more carbons, the chain is the parent and the ring is a substituent (cycloalkyl group).
Ring Strain and Stability of Cycloalkanes
Ring Strain: Angle and Torsional Strain
Cycloalkanes do not all possess the same stability due to ring strain, which is the sum of angle strain and torsional strain:
Angle strain: Deviation from the ideal tetrahedral bond angle (109.5°) in sp3 hybridized carbons.
Torsional strain: Arises from eclipsed interactions between adjacent bonds due to restricted rotation in rings.
Relative Stabilities of Cycloalkanes
The stability of cycloalkanes varies with ring size. Small rings (e.g., cyclopropane, cyclobutane) and medium rings are strained, while cyclohexane and larger rings are nearly strain-free.
Cycloalkane | Internal Angle (°) | Deviation from 109.5° | Planarity |
|---|---|---|---|
Cyclopropane | ~60 | ~49.5 | Planar |
Cyclobutane | ~88 | ~21 | Slightly folded |
Cyclopentane | ~108 (if planar) | ~1.5 | Slightly bent |
Cyclohexane | 109.5 | 0 | Non-planar (chair) |
Figure: Cycloalkane strain energies are calculated by comparing the heat of combustion per CH2 group to that of acyclic alkanes.
Cyclopropane
Cyclopropane has significant angle strain due to its 60° bond angles, which deviate by 49.5° from the ideal tetrahedral angle. This results in high ring strain and reactivity.
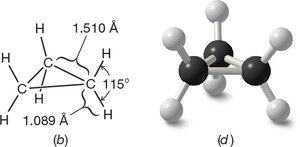
Cyclobutane
Cyclobutane has internal bond angles of about 88°, deviating by 21° from the ideal. The ring is not planar but slightly folded to reduce torsional strain, as a planar structure would have all C–H bonds eclipsed, increasing torsional strain.
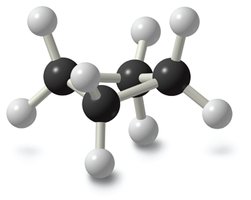
Cyclopentane
If cyclopentane were planar, its bond angles would be close to the ideal tetrahedral angle, but planarity would introduce significant torsional strain. Therefore, cyclopentane adopts a slightly bent (envelope) conformation to minimize strain.
Conformations of Cyclohexane
The Chair and Boat Conformations
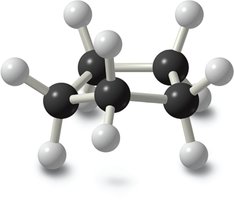
Cyclohexane is unique among cycloalkanes because it can adopt conformations that are nearly free of angle and torsional strain. The most stable conformation is the chair form, while the boat and twist boat forms are higher in energy due to eclipsed interactions and steric strain (1,4-flagpole interactions).
The chair conformation is the lowest energy form.
The boat conformation is less stable due to eclipsed bonds and steric interactions.
The twist boat conformation is intermediate in energy between the chair and boat forms.
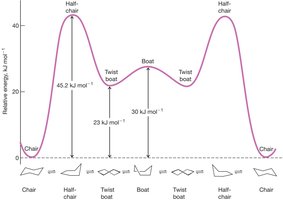
Energy diagram: The interconversion between chair, half-chair, twist boat, and boat conformations shows the relative energies and transition states.
Axial and Equatorial Positions in Cyclohexane
In the chair conformation, each carbon has two types of bonds:
Axial bonds: Oriented perpendicular (up or down) to the ring plane.
Equatorial bonds: Oriented slightly outward from the ring, roughly parallel to the ring plane.
Substituents prefer the equatorial position to minimize steric (1,3-diaxial) interactions.
Conformational Analysis of Substituted Cyclohexanes
Substituents on cyclohexane rings can occupy axial or equatorial positions. The chair conformation with a bulky group in the axial position is less stable due to 1,3-diaxial interactions. The equilibrium favors the conformation with the bulky group in the equatorial position.
The larger the substituent, the greater the preference for the equatorial position.
For disubstituted cyclohexanes, the most stable conformation is the one with the largest groups equatorial.
Cis-Trans Isomerism in Cycloalkanes
Cis-Trans Isomerism
Disubstituted cycloalkanes can exhibit cis-trans isomerism depending on whether the substituents are on the same side (cis) or opposite sides (trans) of the ring. This is especially important in cyclohexane derivatives, where the spatial arrangement affects physical and chemical properties.
Cis isomer: Both substituents are on the same side of the ring.
Trans isomer: Substituents are on opposite sides of the ring.
Special Topic: Buckminsterfullerene (C60)
Buckminsterfullerene is a spherical molecule composed of 60 carbon atoms arranged in a pattern of pentagons and hexagons, resembling a soccer ball. It is an example of a fullerene, a class of closed-cage carbon molecules with unique chemical and physical properties.

Additional info: Buckminsterfullerene is not a cycloalkane but is included here as an example of a unique carbon structure with significant relevance in materials science and nanotechnology.
