 Back
BackAlkanes and Cycloalkanes: Structure, Nomenclature, Conformations, and Properties
Study Guide - Smart Notes

Alkanes and Cycloalkanes
Introduction to Hydrocarbons
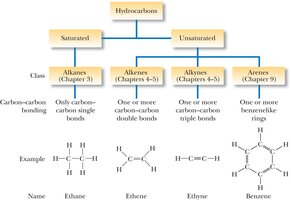
Hydrocarbons are organic compounds composed exclusively of carbon and hydrogen. They are classified based on the types of bonds between carbon atoms and the presence or absence of rings.
Saturated hydrocarbons: Only single bonds between carbons (alkanes and cycloalkanes).
Unsaturated hydrocarbons: Contain double or triple bonds (alkenes, alkynes) or aromatic rings (arenes).
Structure and Geometry of Alkanes
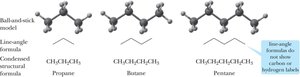
Alkanes are saturated hydrocarbons with only single bonds and a general formula of $C_nH_{2n+2}$. Each carbon atom in an alkane is sp3 hybridized, resulting in a tetrahedral geometry with bond angles of approximately 109.5°.
Aliphatic hydrocarbon: Another term for alkanes.
Line-angle formula: A simplified way to represent alkane structures, where each vertex represents a carbon atom.
Names, Formulas, and Isomerism
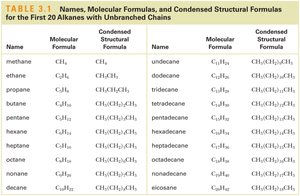
Alkanes are named according to the number of carbon atoms and follow the general formula $C_nH_{2n+2}$. Isomerism arises when compounds have the same molecular formula but different connectivity.
Constitutional isomers: Compounds with the same molecular formula but different atom connectivity.
Name | Molecular Formula | Condensed Structural Formula |
|---|---|---|
methane | CH4 | CH4 |
ethane | C2H6 | CH3CH3 |
propane | C3H8 | CH3CH2CH3 |
butane | C4H10 | CH3(CH2)2CH3 |
pentane | C5H12 | CH3(CH2)3CH3 |
Constitutional Isomerism
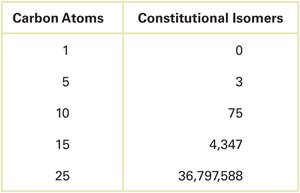
The number of possible constitutional isomers increases dramatically with the number of carbon atoms.
Carbon Atoms | Constitutional Isomers |
|---|---|
1 | 0 |
5 | 3 |
10 | 75 |
15 | 4,347 |
25 | 36,797,588 |
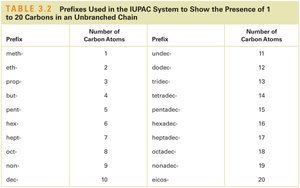
IUPAC Nomenclature of Alkanes
The International Union of Pure and Applied Chemistry (IUPAC) system provides rules for naming alkanes and their derivatives.
The suffix -ane specifies an alkane.
The prefix indicates the number of carbon atoms in the parent chain.
Prefix | Number of Carbon Atoms |
|---|---|
meth- | 1 |
eth- | 2 |
prop- | 3 |
but- | 4 |
pent- | 5 |
Parent Chain and Substituents
The parent name is based on the longest continuous carbon chain. Groups attached to this chain are called substituents, and those derived from alkanes by removing a hydrogen are called alkyl groups (symbol R-).
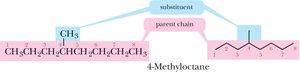
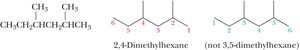
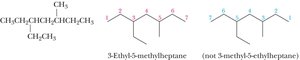
Naming Branched Alkanes
Number the parent chain from the end nearest a substituent.
If two or more identical substituents are present, use prefixes (di-, tri-, tetra-, etc.).
List different substituents alphabetically (ignoring di-, tri-, etc., but including iso-).
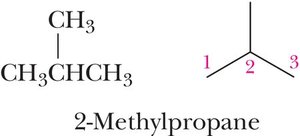
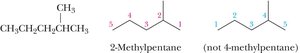
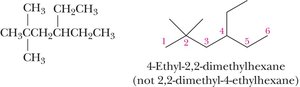
Common Alkyl Groups
Name | Condensed Structural Formula |
|---|---|
methyl | –CH3 |
ethyl | –CH2CH3 |
propyl | –CH2CH2CH3 |
isopropyl | –CH(CH3)2 |

Classification of Carbons
Primary (1°): Carbon bonded to one other carbon.
Secondary (2°): Carbon bonded to two other carbons.
Tertiary (3°): Carbon bonded to three other carbons.
Quaternary (4°): Carbon bonded to four other carbons.

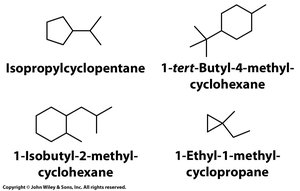
Cycloalkanes: Structure and Nomenclature
Cycloalkanes are saturated hydrocarbons containing rings. Their general formula is $C_nH_{2n}$. The most common ring sizes are five- and six-membered rings.
Name by prefixing the corresponding alkane with cyclo-.
Number substituents to give the lowest set of numbers, and list them alphabetically.
IUPAC General System: Prefix-Infix-Suffix
The IUPAC system uses a three-part name: prefix (number of carbons), infix (type of C–C bonds), and suffix (class of compound).
Infix | Nature of Carbon–Carbon Bonds |
|---|---|
-an- | All single bonds |
-en- | One or more double bonds |
-yn- | One or more triple bonds |

Suffix | Class of Compound |
|---|---|
-e | Hydrocarbon |
-ol | Alcohol |
-al | Aldehyde |
-one | Ketone |
-oic acid | Carboxylic acid |

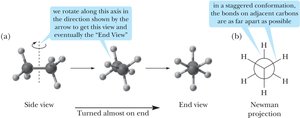
Conformations of Alkanes and Cycloalkanes
Conformations are different spatial arrangements of atoms resulting from rotation about single bonds. The most important conformations are staggered (lowest energy) and eclipsed (highest energy).
Torsional strain: Strain due to eclipsed interactions.
Angle strain: Strain due to deviation from ideal bond angles.

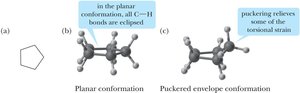
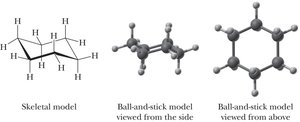
Cyclopentane and Cyclohexane Conformations
Cyclopentane relieves torsional strain by adopting an "envelope" conformation, while cyclohexane adopts a "chair" conformation, which is nearly strain-free.
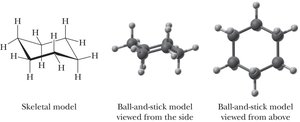
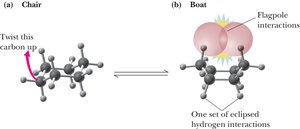
Chair and Boat Conformations of Cyclohexane
The chair conformation is the most stable, with all bonds staggered and bond angles near 109.5°. The boat conformation is less stable due to torsional and steric strain.
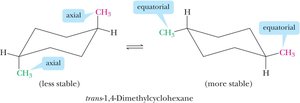
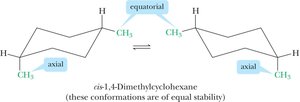
Axial and Equatorial Bonds
In the chair conformation, each carbon has one axial and one equatorial hydrogen. Substituents prefer the equatorial position to minimize steric interactions.

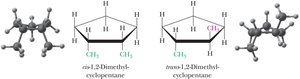
Cis-Trans Isomerism in Cycloalkanes
Cis-trans isomers have the same molecular formula and connectivity but differ in the spatial arrangement of substituents around a ring. These isomers cannot be interconverted by rotation about single bonds.

Physical Properties of Alkanes
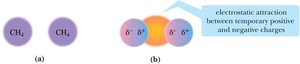
Alkanes are nonpolar and interact via weak dispersion forces. Their physical properties depend on molecular weight and branching.
Low-molecular-weight alkanes (C1–C4) are gases at room temperature.
Medium-weight alkanes (C5–C17) are liquids.
High-molecular-weight alkanes (C18+) are solids.
Density is typically around 0.7 g/mL; alkanes float on water.

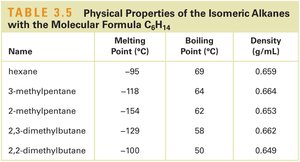
Name | Melting Point (°C) | Boiling Point (°C) | Density (g/mL) |
|---|---|---|---|
hexane | -95 | 69 | 0.659 |
3-methylpentane | -118 | 64 | 0.664 |
2-methylpentane | -154 | 62 | 0.653 |
2,3-dimethylbutane | -129 | 58 | 0.662 |
2,2-dimethylbutane | -100 | 50 | 0.649 |
Reactions of Alkanes: Combustion
Alkanes are primarily used as fuels due to their exothermic combustion reactions. The heat of combustion is the energy released when one mole of alkane is oxidized to CO2 and H2O.
Example: $CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O$
Example: $C_3H_8 + 5O_2 \rightarrow 3CO_2 + 4H_2O$

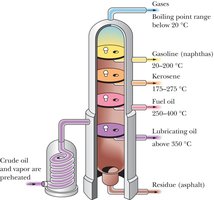
Sources of Alkanes
Alkanes are found in natural gas, petroleum, and coal. Petroleum is separated into useful fractions by fractional distillation based on boiling points.
Natural gas: Mainly methane and ethane.
Petroleum: Source of gasoline, kerosene, fuel oil, lubricating oils, and asphalt.
Synthesis Gas and Methanol Economy
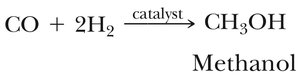
Synthesis gas is a mixture of CO and H2 used to produce methanol and acetic acid. Methanol is a potential alternative fuel and chemical feedstock.
Reaction: $CO + 2H_2 \xrightarrow{catalyst} CH_3OH$
Additional info: This guide covers the structure, nomenclature, conformational analysis, physical properties, and sources of alkanes and cycloalkanes, as well as their industrial significance and isomerism. It is suitable for exam preparation and foundational understanding in organic chemistry.
