 Back
BackAlkanes and Cycloalkanes: Structure, Properties, and Nomenclature
Study Guide - Smart Notes

Alkanes
Introduction to Alkanes
Alkanes are saturated hydrocarbons containing only carbon–carbon (C–C) and carbon–hydrogen (C–H) single bonds. They are fundamental compounds in organic chemistry, forming the basis for more complex molecules.
Acyclic Alkanes: Linear or branched chains with no rings. General formula: .
Cycloalkanes: Carbon atoms joined in rings. General formula: .
All alkane names end with the suffix "-ane".
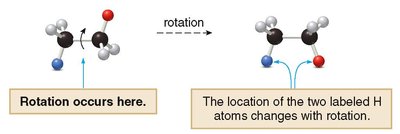
Bond Rotation and Skeletal Structures
Alkanes allow free rotation around C–C single bonds, which affects their three-dimensional structure and conformations.
Rotation: Occurs around single bonds, changing the spatial arrangement of atoms.
Double and triple bonds do not allow rotation due to the presence of pi bonds.
Physical Properties of Alkanes
Alkanes are characterized by their nonpolar nature and weak intermolecular forces, leading to distinctive physical properties.
Low melting and boiling points: Due to weak London dispersion forces (LDF).
State at room temperature: Small alkanes (1–4 carbons) are gases; medium alkanes (5–17 carbons) are liquids; large alkanes (18+ carbons) are waxy solids.
Solubility: Insoluble in water, less dense than water.




Boiling Point Trends
As the number of carbons increases, the boiling point rises due to increased surface area and stronger London dispersion forces. 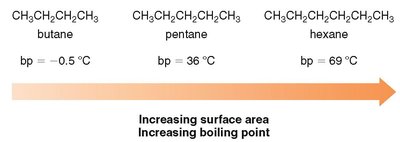
Common Alkanes and Their Structures
Methane (CH4): Simplest alkane, tetrahedral geometry.
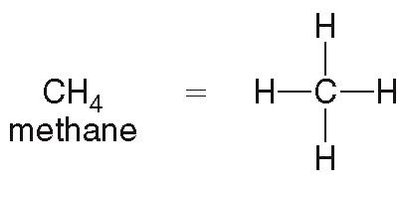

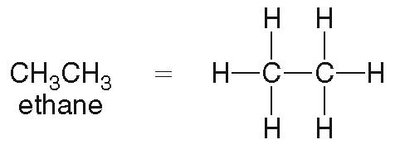
Ethane (C2H6): Two-carbon alkane, tetrahedral geometry at each carbon.
Propane (C3H8): Three-carbon alkane, can be drawn as a straight or bent chain.
Butane (C4H10): Four-carbon alkane.
Classifying Carbon Atoms
Carbon atoms in alkanes are classified based on the number of other carbons they are bonded to:
Primary (1°): Bonded to one other carbon.
Secondary (2°): Bonded to two other carbons.
Tertiary (3°): Bonded to three other carbons.
Quaternary (4°): Bonded to four other carbons.
Alkane Nomenclature
An Introduction to Nomenclature
The IUPAC system provides a systematic method for naming organic compounds, ensuring each has a unique and unambiguous name.
Systematic name: IUPAC name (e.g., 2-methylpropane).
Generic name: Official name (e.g., ibuprofen).
Trade name: Brand name (e.g., Advil).
Structure of Alkane Names
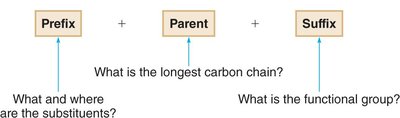
Alkane names with substituents have three parts:
Prefix: Identity, location, and number of substituents.
Parent: Number of carbons in the longest chain.
Suffix: Functional group present (for alkanes, "-ane").
Rules for Alkane Nomenclature
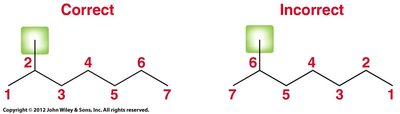
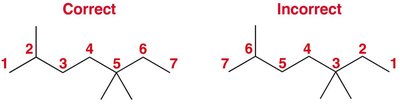
Find the longest continuous carbon chain: This is the parent. If there is a tie, choose the chain with more substituents.
Name the substituents: Use alkyl group names (drop "-ane", add "-yl").
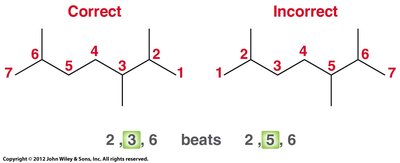
Number the parent chain: Assign numbers to give substituents the lowest possible numbers. If a tie, prioritize the next substituent.
Assemble the name: Prefix (alphabetical order) + parent + suffix. Use di-, tri-, tetra- for multiple identical substituents; these prefixes are not considered in alphabetizing.
Alkyl Groups and Special Substituents
Common Alkyl Groups
Alkyl groups are derived from alkanes by removing a hydrogen atom. Examples include methyl, ethyl, propyl, isopropyl, butyl, sec-butyl, tert-butyl, and isobutyl.
Nomenclature Involving Halogen Substituents
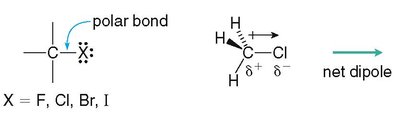
Halogens (F, Cl, Br, I) are treated as substituents in alkane nomenclature.
Use prefixes: fluoro-, chloro-, bromo-, iodo-.
Find the longest chain containing all halogens.
Follow standard numbering and alphabetizing rules.
Alkyl Halides
Physical Properties
Alkyl halides contain a polar C–X bond, resulting in a net dipole.
Higher melting and boiling points than alkanes, but lower than alcohols.
Boiling and melting points increase with the size of the alkyl group and halogen.
All alkyl halides are insoluble in water.
Cycloalkanes
Structure and Nomenclature
Cycloalkanes are ring-shaped alkanes.
Parent is the ring if two or more substituents are attached.
Number substituents to give the lowest possible numbers.
Alphabetize substituents in the name.
Isomerism in Alkanes
Constitutional Isomers
Isomers are compounds with the same molecular formula but different connectivity.
Example: Butane and isobutane (both C4H10).
Combustion Reactions of Alkanes
Combustion Reaction
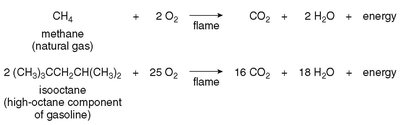
Alkanes undergo combustion in the presence of oxygen to produce carbon dioxide and water.
Complete combustion:
Example: Combustion of methane:
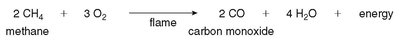
Incomplete Combustion
If oxygen is insufficient, incomplete combustion occurs, producing carbon monoxide (CO), a toxic gas.
Example:
Summary Table: Physical Properties of Alkanes and Alkyl Halides
Compound | Boiling Point (°C) | State at Room Temp |
|---|---|---|
Methane | -161 | Gas |
Ethane | -89 | Gas |
Propane | -42 | Gas |
Butane | -0.5 | Gas |
Pentane | 36 | Liquid |
Hexane | 69 | Liquid |
Octane | 126 | Liquid |
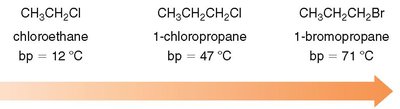
Chloroethane | 12 | Liquid |
1-Chloropropane | 47 | Liquid |
1-Bromopropane | 71 | Liquid |
Key Takeaways
Alkanes are saturated hydrocarbons with only single bonds.
Physical properties depend on chain length and molecular structure.
Nomenclature follows IUPAC rules for clarity and precision.
Alkyl halides and cycloalkanes are important derivatives with distinct properties.
Combustion is a key reaction for alkanes, producing energy and CO2.
