Back
BackAlkynes: Structure, Properties, and Reactions
Study Guide - Smart Notes

Alkynes: Structure and Nomenclature
Definition and General Properties
Alkynes, also known as acetylenes, are hydrocarbons containing at least one carbon-carbon triple bond. This triple bond consists of one sigma (σ) bond and two pi (π) bonds, resulting in a linear geometry around the triple-bonded carbons. The general formula for alkynes is CnH2n-2.
Terminal alkyne: The triple bond is at the end of the carbon chain.
Internal alkyne: The triple bond is located within the carbon chain.
Naming: Alkynes are named using the same IUPAC rules as alkenes, but the suffix changes to -yne. When both alkene and alkyne groups are present, alkenes are given priority in numbering if there is a tie. As substituents, the ending changes from -e to -yl (e.g., propargyl group). Simple alkynes are often named as derivatives of acetylene.
Examples: 6-bromo-2-methyl-3-heptyne, 2-propynylcyclohexane, 2-methyl-1-penten-3-yne
Bonding and Structure of Alkynes
Bonding Characteristics
The triple bond in alkynes consists of one sigma bond and two mutually perpendicular pi bonds. This arrangement leads to a linear geometry and a high electron density along the bond axis.
Bond length: The C–C bond in alkynes is shorter than in alkanes or alkenes due to increased s-character and stronger bonding.
Electron density: The electron density is concentrated in a cylindrical region around the bond axis.
Bond Energies
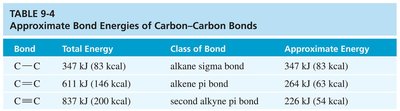
The strength of carbon-carbon bonds increases with bond order. The triple bond in alkynes is the strongest, followed by the double bond in alkenes and the single bond in alkanes.
Bond | Total Energy | Class of Bond | Approximate Energy |
|---|---|---|---|
C–C | 347 kJ (83 kcal) | alkane sigma bond | 347 kJ (83 kcal) |
C=C | 611 kJ (146 kcal) | alkene pi bond | 264 kJ (63 kcal) |
C≡C | 837 kJ (200 kcal) | second alkyne pi bond | 226 kJ (54 kcal) |
Acidity and Hybridization Effects
Acidity of Alkynes
The acidity of a hydrogen atom bonded to a carbon depends on the hybridization of the carbon atom. The greater the s-character, the more stable the conjugate base, and the more acidic the hydrogen.
Order of acidity: sp (alkyne) > sp2 (alkene) > sp3 (alkane)
pKa values: Terminal alkynes (pKa ≈ 25) are much more acidic than alkenes (pKa ≈ 44) or alkanes (pKa ≈ 50).
Deprotonation: Strong bases such as amide ion (NH2–) can deprotonate terminal alkynes, but alkoxides (RO–) cannot.
Reactivity of Alkynes
Nucleophilicity and Electrophilicity
Alkynes can act as both nucleophiles and electrophiles, depending on the reaction conditions. The pi electrons are nucleophilic, while the acidic proton in terminal alkynes can be removed to generate a nucleophilic acetylide ion.
Acetylide ions: Generated by deprotonation of terminal alkynes; strong nucleophiles in C–C bond-forming reactions.
Synthesis of Alkynes
From Acetylides
Alkynes can be synthesized by alkylation of acetylide ions. This reaction works best with methyl or primary alkyl halides; secondary, tertiary, or bulky alkyl halides tend to undergo elimination (E2) instead.
Alkylation:
Reaction with carbonyls: Acetylide ions react with carbonyl compounds (formaldehyde, aldehydes, ketones) to form alcohols after protonation.

By Elimination
Alkynes can also be synthesized by double elimination of dihalides (geminal or vicinal). This requires a strong base (e.g., KOH at 200°C or NaNH2 at 150°C) and often gives poor yields due to harsh conditions.
KOH: Favors formation of internal alkynes.
NaNH2: Favors formation of terminal alkynes.
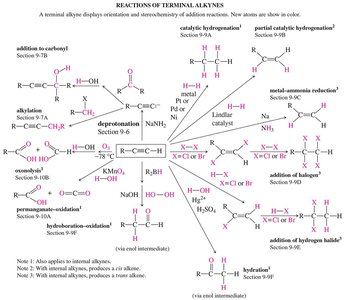
Addition Reactions of Alkynes
Hydrogenation
Alkynes undergo catalytic hydrogenation to form alkanes, passing through an alkene intermediate. Partial hydrogenation can be achieved using Lindlar's catalyst (Pd/BaSO4 with quinoline) or nickel boride (Ni2B), resulting in a cis-alkene product due to syn addition.
Metal-Ammonia Reduction
Reduction of alkynes with sodium in liquid ammonia produces trans-alkenes via anti addition. The sodium donates electrons to the alkyne, generating a solvated electron (deep blue color).

Addition of Halogens and Hydrogen Halides
Alkynes react with halogens (X2) and hydrogen halides (HX) in a stepwise manner. The stoichiometry determines whether the reaction stops at the alkene or proceeds to the alkane. The presence of peroxides can alter the regioselectivity of HX addition (anti-Markovnikov addition).
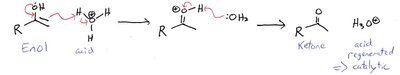
Hydration and Tautomerization
Alkynes undergo hydration in the presence of acid and mercuric ion catalysts to yield enols, which rapidly tautomerize to ketones (or aldehydes for terminal alkynes). Hydroboration-oxidation provides anti-Markovnikov addition, yielding aldehydes from terminal alkynes.
Oxidation of Alkynes
Permanganate and Ozonolysis
Alkynes can be oxidized by potassium permanganate (KMnO4) or ozone (O3). Under strong conditions, oxidative cleavage yields carboxylic acids (from terminal alkynes, formate is also produced).
Summary of Alkyne Reactions
The following diagram summarizes the major reactions of terminal alkynes, including addition, oxidation, and C–C bond-forming reactions.