 Back
Backch 9 Alkynes: Structure, Properties, and Reactions
Study Guide - Smart Notes

Alkynes: Structure, Properties, and Reactions
Introduction to Alkynes
Alkynes are hydrocarbons characterized by the presence of a carbon–carbon triple bond. They are an important class of unsaturated compounds in organic chemistry, with unique physical and chemical properties that distinguish them from alkanes and alkenes.
General Formula:
Degree of Unsaturation: Each triple bond introduces two elements of unsaturation.
Reactivity: Alkynes undergo addition and oxidation reactions similar to alkenes, but also have reactions specific to the triple bond.
Nomenclature of Alkynes
The IUPAC naming of alkynes follows systematic rules to ensure clarity and consistency.
Identify the longest carbon chain containing the triple bond.
Change the '-ane' ending of the corresponding alkane to '-yne.'
Number the chain from the end nearest the triple bond.
Assign numbers to substituents to indicate their positions.
Functional groups (except ethers and halides) have higher priority than alkynes.
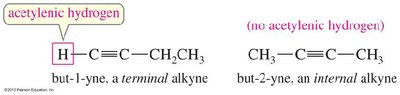
Example: But-1-yne (terminal alkyne) vs. But-2-yne (internal alkyne).
Physical Properties of Alkynes
Alkynes exhibit physical properties similar to those of alkanes and alkenes, with some distinctions due to the triple bond.
Polarity: Nonpolar, insoluble in water, but soluble in organic solvents.
Boiling Points: Comparable to alkanes of similar molecular weight.
Density: Less dense than water.
Physical State: Alkynes with up to four carbons are gases at room temperature.
Structure and Bonding in Alkynes
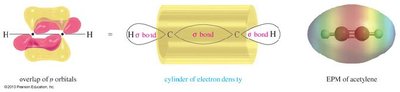
The triple bond in alkynes consists of one sigma () and two pi () bonds, resulting from the overlap of sp-hybridized orbitals and unhybridized p orbitals.
Hybridization: Each carbon in the triple bond is sp-hybridized, leading to a linear geometry (180° bond angle).
Bonding: The sigma bond forms from sp–sp overlap, while the two pi bonds arise from the side-by-side overlap of unhybridized p orbitals.
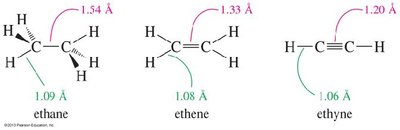
Bond Lengths: Triple bonds are shorter than double or single bonds due to increased s character and stronger orbital overlap.
Classification of Alkynes
Alkynes are classified as terminal or internal based on the position of the triple bond.
Terminal Alkynes: The triple bond is at the end of the carbon chain (contains an acetylenic hydrogen).
Internal Alkynes: The triple bond is within the carbon chain (no acetylenic hydrogen).

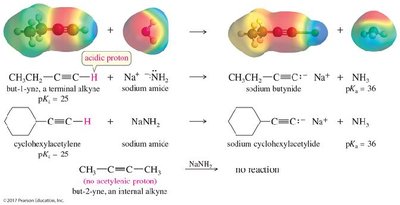
Acidity of Alkynes
Terminal alkynes are significantly more acidic than alkanes and alkenes due to the high s character of the sp-hybridized carbon.
Acidity Order: Alkynes > Alkenes > Alkanes
Deprotonation: Terminal alkynes can be deprotonated by strong bases such as sodium amide (), forming acetylide ions.
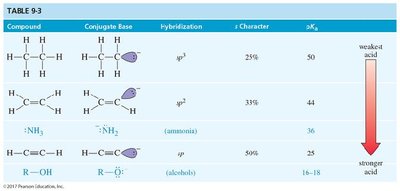
pKa Values: Terminal alkynes (pKa ≈ 25), ammonia (pKa ≈ 36), alkanes (pKa ≈ 50).
Compound | Conjugate Base | Hybridization | % s Character | pKa |
|---|---|---|---|---|
Alkane | Alkyl anion | sp3 | 25% | 50 |
Ammonia | Amide | sp3 | 33% | 36 |
Alkyne | Acetylide | sp | 50% | 25 |
Alcohol | Alkoxide | sp3 | 25% | 16–18 |
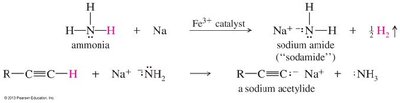
Formation and Reactions of Acetylide Ions
Terminal alkynes react with strong bases to form acetylide ions, which are strong nucleophiles and bases.
Base Used: Sodium amide () is commonly used for deprotonation.
Reactivity: Acetylide ions participate in nucleophilic substitution (SN2) and addition reactions.
Synthesis of Alkynes
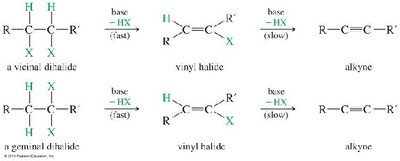
Alkynes can be synthesized by two main methods: alkylation of acetylide ions and elimination reactions of dihalides.
Alkylation: Acetylide ions react with primary alkyl halides via SN2 to extend the carbon chain.
Elimination: Dehydrohalogenation of vicinal or geminal dihalides (using strong base) yields alkynes.

Hydrogenation and Reduction of Alkynes
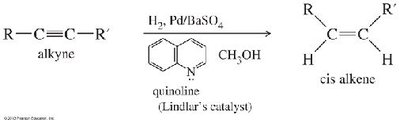
Alkynes can be reduced to alkenes or alkanes by catalytic hydrogenation. The stereochemistry of the product depends on the catalyst and conditions used.
Complete Hydrogenation: Using Pd, Pt, or Ni catalysts, alkynes are fully reduced to alkanes.
Partial Hydrogenation (cis): Lindlar's catalyst (Pd/BaSO4 with quinoline) yields cis-alkenes via syn addition.
Partial Hydrogenation (trans): Sodium in liquid ammonia (Na/NH3) gives trans-alkenes via anti addition.
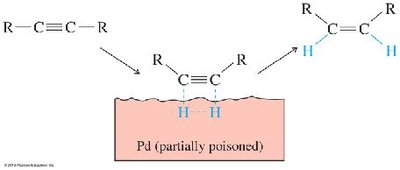
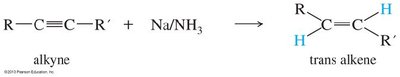
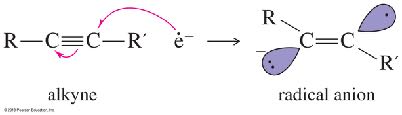
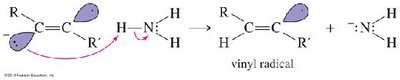
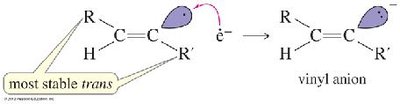
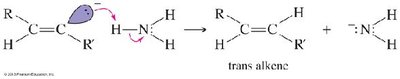
Addition Reactions of Alkynes
Alkynes undergo electrophilic addition reactions with halogens and hydrogen halides, often following Markovnikov or anti-Markovnikov orientation.
Halogenation: Cl2 and Br2 add to alkynes to form dihalides or tetrahalides (mixture of cis and trans isomers).
Hydrohalogenation: HX adds to alkynes to form vinyl halides (one equivalent) or geminal dihalides (two equivalents). Markovnikov addition predominates unless peroxides are present (anti-Markovnikov for HBr).
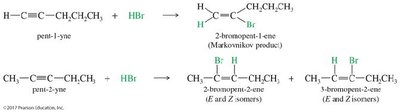
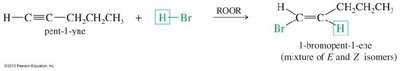
Hydration of Alkynes
Alkynes react with water in the presence of acid and mercuric ion (Hg2+) or via hydroboration–oxidation to yield carbonyl compounds through enol intermediates.
Mercuric Ion Catalysis: Markovnikov addition yields ketones via enol intermediates (keto–enol tautomerism).
Hydroboration–Oxidation: Anti-Markovnikov addition yields aldehydes (for terminal alkynes) via enol intermediates.
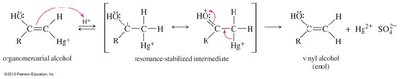
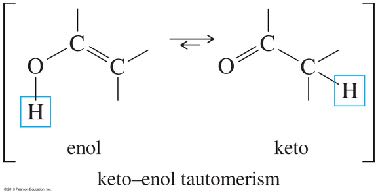
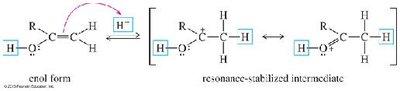
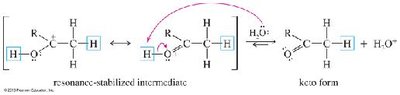
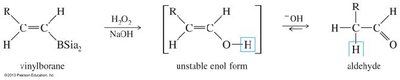
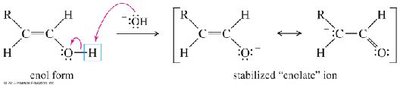
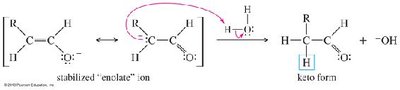
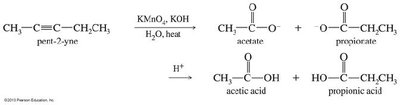
Oxidative Cleavage of Alkynes
Alkynes can be cleaved oxidatively by potassium permanganate (KMnO4) or ozone (O3), yielding carboxylic acids or diketones depending on the conditions.
Neutral KMnO4: Produces diketones.
Basic KMnO4 or Ozonolysis: Cleaves the triple bond to yield carboxylic acids.
Application: Used to determine the position of the triple bond in unknown alkynes.
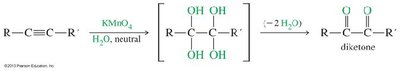
Summary Table: Key Reactions and Properties of Alkynes
Topic | Key Points |
|---|---|
Structure | sp-hybridized, linear, triple bond (1 σ, 2 π) |
Acidity | Terminal alkynes (pKa ≈ 25), deprotonated by strong bases |
Synthesis | Alkylation of acetylide ions, elimination of dihalides |
Hydrogenation | Lindlar's catalyst (cis), Na/NH3 (trans) |
Addition | Halogens, HX (Markovnikov/anti-Markovnikov) |
Hydration | HgSO4/H2SO4 (ketone), hydroboration (aldehyde) |
Oxidation | KMnO4, O3 (diketones, carboxylic acids) |
Additional info: The notes above expand on the brief points in the original slides, providing definitions, mechanisms, and context for each reaction and property. All images included are directly relevant to the adjacent explanations and reinforce the concepts discussed.
