 Back
BackAmines and Neurotransmitters: Structure, Function, and Biological Importance
Study Guide - Smart Notes

Amines and Heterocycles
Introduction to Amines
Amines are organic compounds derived from ammonia (NH3) in which one or more hydrogen atoms are replaced by alkyl or aryl groups. The nitrogen atom in amines can also be incorporated into ring structures, forming heterocycles. These compounds are fundamental in organic chemistry and biochemistry due to their roles in biological systems and pharmaceuticals.
Primary, secondary, and tertiary amines are classified based on the number of organic substituents attached to the nitrogen atom.
Heterocyclic amines contain the nitrogen atom as part of a ring structure, which can significantly affect their chemical properties and biological activity.
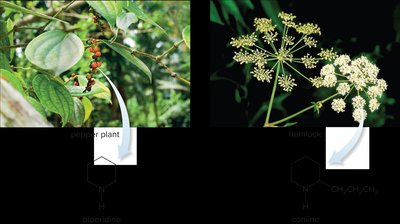
Common Amine Heterocycles
Several important heterocyclic amines are found in nature and synthetic compounds. Their structures influence their reactivity and biological function.
Piperidine: Six-membered ring with one nitrogen atom.
Pyrrolidine: Five-membered ring with one nitrogen atom.
Pyridine: Six-membered aromatic ring with one nitrogen atom.
Pyrrole: Five-membered aromatic ring with one nitrogen atom.
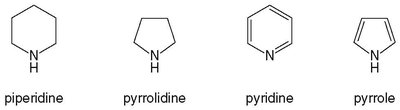
Biologically Active Amine Heterocycles
Caffeine and Nicotine
Caffeine and nicotine are naturally occurring alkaloids containing amine heterocycles. Both are widely used stimulants of the central nervous system and are found in plants.
Caffeine: A bitter-tasting amine that acts as a mild stimulant, increasing heart rate and promoting glucose production. Its effects are temporary, leading to repeated consumption and mild addiction.
Nicotine: A highly toxic amine from tobacco, acting as a stimulant in small doses. It is highly addictive and associated with significant health risks, including cancer and cardiovascular disease.
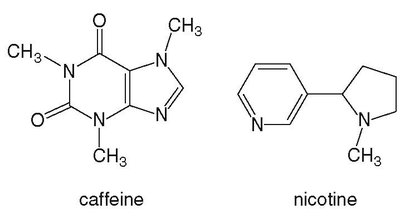
Alkaloids: Morphine, Codeine, and Heroin
Alkaloids are naturally occurring amines with pronounced physiological effects. Morphine and codeine are derived from the opium poppy and are used as pain relievers, though both are highly addictive. Heroin, synthesized from morphine, is even more potent due to its increased solubility in fat cells.
Morphine: Potent pain reliever, highly addictive.
Codeine: Less potent, used for milder pain, still addictive.
Heroin: Illegal drug, more potent than morphine due to increased lipid solubility.
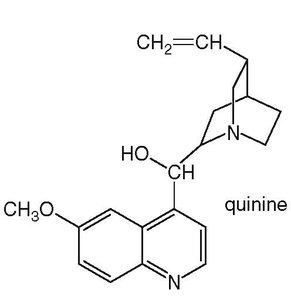
Quinine
Quinine is an alkaloid isolated from the bark of the cinchona tree. It is used as a fever reducer and to treat malaria.
Neurotransmitters: Chemical Messengers in the Nervous System
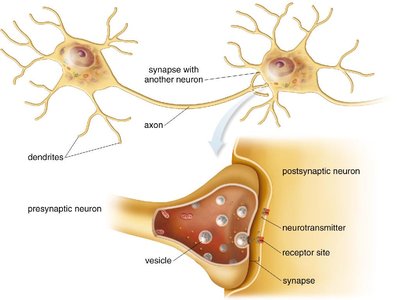
Definition and Mechanism
A neurotransmitter is a chemical messenger that transmits nerve impulses from one neuron to another across a synapse. The presynaptic neuron releases the neurotransmitter, which binds to receptors on the postsynaptic neuron, delivering the chemical message. The neurotransmitter is then degraded or reabsorbed to reset the system.
Drugs can affect neurotransmitter release, binding, or reuptake, altering nervous system function.
Norepinephrine and Dopamine
Both norepinephrine and dopamine are synthesized from the amino acid tyrosine and play crucial roles in mood, movement, and the body's response to stress.
Norepinephrine: Elevates mood; excessive levels can cause mania. Converted to adrenaline (epinephrine) during stress.
Dopamine: Regulates movement, emotions, and pleasure. Deficiency is linked to Parkinson's disease; excess is associated with schizophrenia and addiction.
L-Dopa is used to treat Parkinson's disease because it can cross the blood-brain barrier and is converted to dopamine in the brain.
Serotonin
Serotonin is synthesized from tryptophan and is important for mood, sleep, perception, and temperature regulation. Deficiency leads to depression, and many antidepressants are selective serotonin reuptake inhibitors (SSRIs), which increase serotonin levels in the synapse.
SSRIs inhibit the reuptake of serotonin by the presynaptic neuron, increasing its concentration and improving mood.
Acetylcholine
Acetylcholine is a quaternary ammonium ion that acts as a neurotransmitter between neurons and muscle cells. Nicotine can bind to acetylcholine receptors, triggering dopamine release and contributing to addiction.
Epinephrine (Adrenaline) and the Fight-or-Flight Response
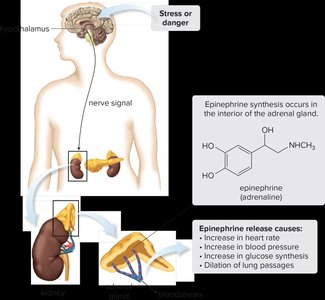
Hormonal Role and Synthesis
Epinephrine is a hormone produced by the adrenal glands from norepinephrine. It is released in response to stress or danger, initiating the 'fight-or-flight' response.
Effects include increased heart rate, blood pressure, glucose synthesis, and dilation of lung passages.
Other Biologically Active Amines
2-Phenylethylamine, Amphetamines, and Related Compounds
2-Phenylethylamine is a structural motif found in many biologically active amines. Amphetamine and methamphetamine are powerful central nervous system stimulants, used medically for ADHD but also widely abused due to their addictive properties. Pseudoephedrine, a common decongestant, can be chemically converted to methamphetamine, leading to its replacement by phenylephedrine in many products.
Summary Table: Key Amines and Their Biological Roles
Compound | Structure Type | Biological Role | Notes |
|---|---|---|---|
Caffeine | Heterocyclic amine | CNS stimulant | Mildly addictive |
Nicotine | Heterocyclic amine | CNS stimulant | Highly addictive, toxic |
Morphine | Alkaloid | Pain relief | Highly addictive |
Codeine | Alkaloid | Pain relief | Less potent, still addictive |
Quinine | Alkaloid | Antimalarial | Fever reducer |
Norepinephrine | Neurotransmitter | Mood, stress response | Precursor to epinephrine |
Dopamine | Neurotransmitter | Movement, pleasure | Deficiency: Parkinson's; Excess: Schizophrenia |
Serotonin | Neurotransmitter | Mood, sleep | Deficiency: Depression |
Acetylcholine | Neurotransmitter | Muscle activation | Target of nicotine |
Epinephrine | Hormone | Fight-or-flight | Increases heart rate, glucose |
Additional info: The structures and functions of these amines are central to understanding their pharmacological and physiological effects. Their study bridges organic chemistry and biochemistry, highlighting the importance of molecular structure in biological activity.
