 Back
BackAmines: Structure, Properties, and Reactions (Organic Chemistry II, Chapter 19)
Study Guide - Smart Notes

Amines: Structure, Properties, and Reactions
Introduction to Amines
Amines are organic compounds derived from ammonia (NH3) by replacement of one or more hydrogen atoms with alkyl or aryl groups. They are fundamental in biological systems and synthetic organic chemistry, serving as building blocks for drugs, natural products, and polymers.
Biologically Active Amines
Many amines, especially alkaloids, are biologically active and play crucial roles in plant defense and human pharmacology. Alkaloids are often synthesized by plants to deter herbivores and insects, and many drugs of addiction are classified as alkaloids.
Examples: Cocaine, nicotine, mescaline, morphine
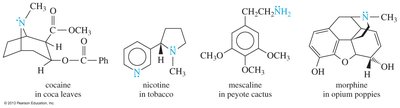
Biological Activity of Amines
Amines are found in neurotransmitters, hormones, vitamins, and amino acids. Their presence is essential for various physiological functions.
Examples: Dopamine (neurotransmitter), epinephrine (hormone), histamine (vasodilator), pyridoxine (vitamin B6), l-tryptophan (amino acid)
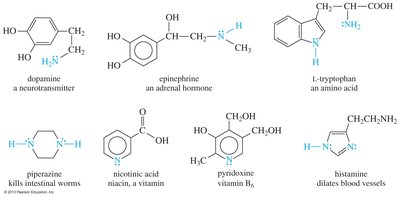
Classification and Structure of Amines
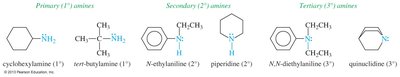
Classes of Amines
Amines are classified based on the number of alkyl or aryl groups attached to the nitrogen atom:
Primary (1°) amine: One alkyl group (RNH2)
Secondary (2°) amine: Two alkyl groups (R2NH)
Tertiary (3°) amine: Three alkyl groups (R3N)
Quaternary (4°) ammonium salt: Four alkyl groups, nitrogen bears a positive charge (R4N+)
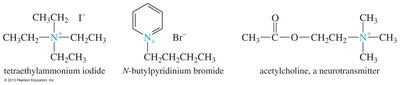
Quaternary Ammonium Salts
Quaternary ammonium salts are ionic compounds with four alkyl groups attached to nitrogen, which carries a positive charge. These salts are important in biochemistry and as phase-transfer catalysts.
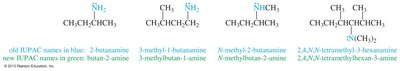
Nomenclature of Amines
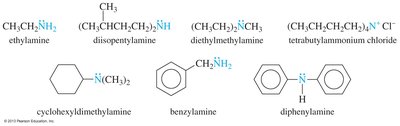
Amines are named using common and IUPAC conventions:
Common names: Names of alkyl groups followed by "-amine"
As substituents: Named as substituents when a higher-priority functional group is present
IUPAC names: Based on the longest carbon chain, replacing the "-e" of alkane with "-amine"
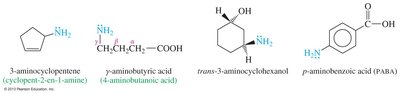
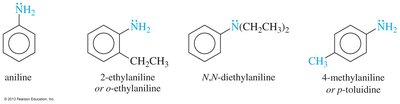
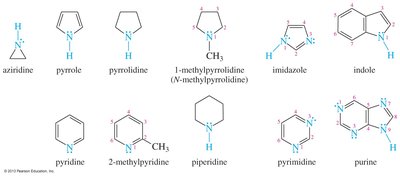
Aromatic and Heterocyclic Amines
Aromatic amines have the amino group attached to a benzene ring, with aniline as the parent compound. Heterocyclic amines contain nitrogen within a ring structure, and the nitrogen is assigned position number 1 in nomenclature.
Structure and Chirality of Amines
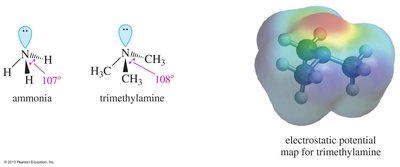
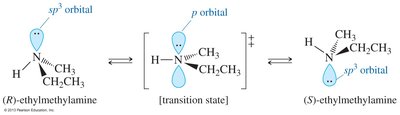
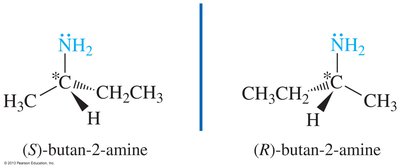
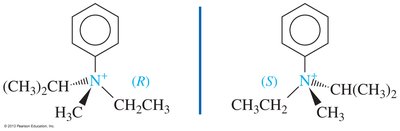
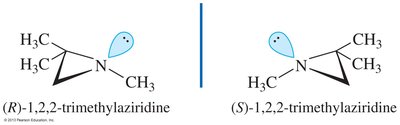
Nitrogen in amines is typically sp3 hybridized with a lone pair, resulting in bond angles slightly less than 109.5°. Chirality in amines can arise from either chiral carbon atoms or, in rare cases, from the nitrogen atom itself.
Chiral amines: Enantiomers cannot be isolated due to rapid inversion around nitrogen (sp2 transition state)
Chiral quaternary ammonium salts: No lone pair, so inversion is not possible; enantiomers can be resolved if all four substituents are different
Cyclic amines: Small rings restrict inversion, allowing resolution of enantiomers
Physical Properties of Amines
Boiling Points and Hydrogen Bonding
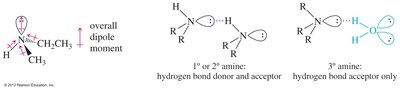
Amines exhibit lower boiling points than alcohols due to weaker N—H hydrogen bonds compared to O—H bonds. Tertiary amines cannot form hydrogen bonds, resulting in even lower boiling points.
Comparison: Primary & secondary amines can hydrogen bond; tertiary amines cannot
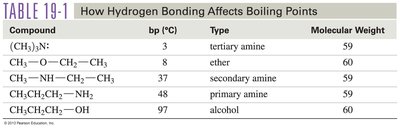
Compound | Boiling Point (°C) | Type | Molecular Weight |
|---|---|---|---|
(CH3)3N | 3 | tertiary amine | 59 |
CH3—O—CH2—CH3 | 8 | ether | 60 |
CH3—NH—CH2—CH3 | 37 | secondary amine | 59 |
CH3CH2CH2—NH2 | 48 | primary amine | 59 |
CH3CH2CH2—OH | 97 | alcohol | 60 |
Solubility and Odor
Small amines (fewer than six carbon atoms) are soluble in water due to their ability to accept hydrogen bonds. Branching increases solubility. Most amines have a characteristic odor resembling rotting fish.
Basicity and Reactivity of Amines
Basicity of Amines
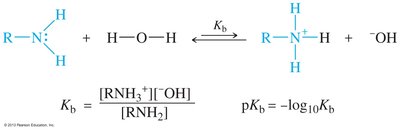
Amines are basic due to the lone pair of electrons on nitrogen, which can accept a proton. Alkyl amines are generally stronger bases than ammonia, but increasing the number of alkyl groups decreases solvation, so secondary and tertiary amines are similar to primary amines in basicity.
Base-dissociation constant (Kb): Quantifies the basicity of amines
pKb:
Alkyl Group Stabilization
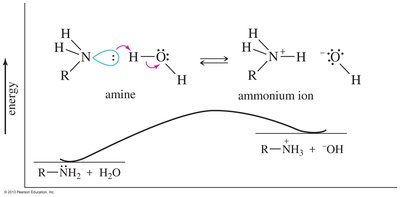
Alkyl groups stabilize the ammonium ion, making the amine a stronger base than ammonia.
Resonance and Hybridization Effects
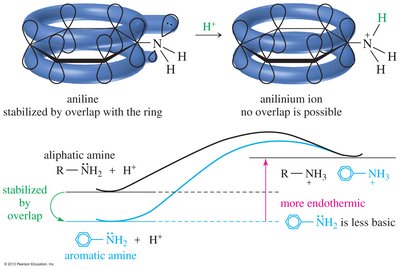
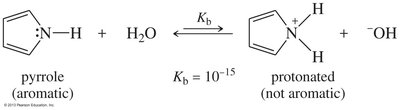
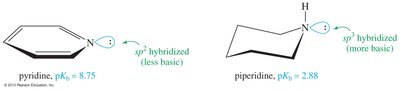
Delocalization of the electron pair (resonance) weakens the basicity of amines. Aromatic amines are less basic than aliphatic amines due to resonance and hybridization effects.
Pyrrole: Protonation destroys aromaticity, making it a very weak base
Pyridine: Less basic than aliphatic amines, but more basic than pyrrole

Purification and Spectroscopy of Amines
Purification of Amines
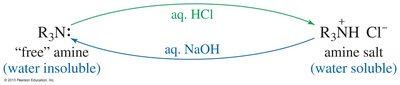
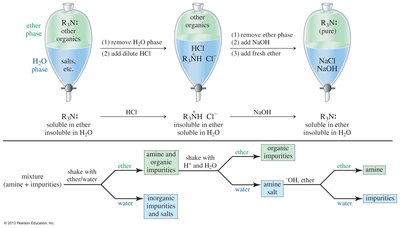
Amines can be purified by conversion to ammonium salts, which are ionic solids with high melting points and are water-soluble. The process removes the characteristic fishy odor.
IR and NMR Spectroscopy
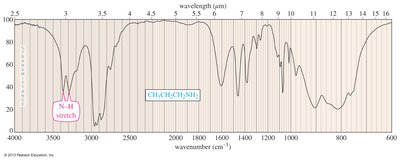
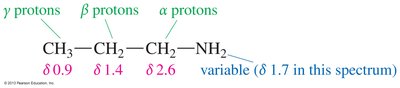
IR spectroscopy identifies amines by N—H stretching between 3200 and 3500 cm-1. Primary amines show two peaks, secondary amines one. NMR spectroscopy reveals that protons on α-carbon atoms are less deshielded than those in alcohols.
Reactions of Amines
Reactions with Ketones and Aldehydes
Amines react with ketones and aldehydes to form imines (Schiff bases) via acid-catalyzed addition and dehydration. Optimum pH for imine formation is around 4.5.
Mechanism: Acid-catalyzed addition of amine to carbonyl, followed by dehydration
*Additional info: Mechanism details are covered in Chapter 18 and 19 mechanisms.*
Aromatic Electrophilic Substitution
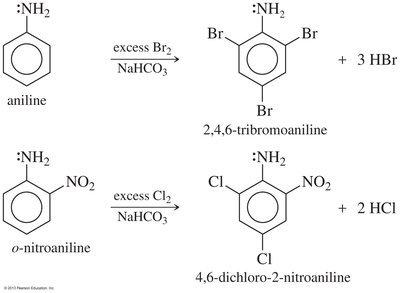
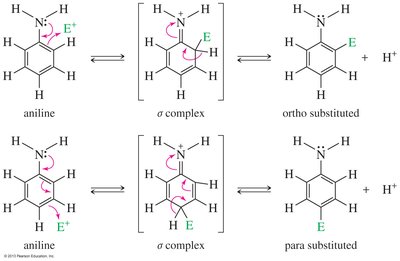
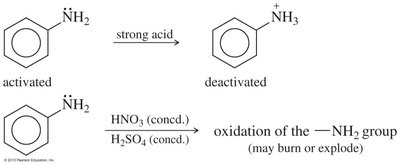
Arylamines (e.g., aniline) are strong activators and direct substitution to ortho and para positions. Multiple substitutions can occur, and protonation of the amine under acidic conditions converts it to a strong deactivator (—NH3+).
Aromatic Substitution of Pyridine
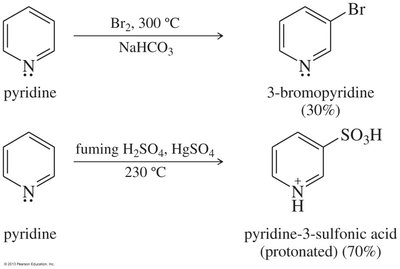
Pyridine is strongly deactivated toward electrophilic attack due to the electronegative nitrogen atom. Substitution occurs at the 3-position (meta), and electrons on nitrogen may react with the electrophile.
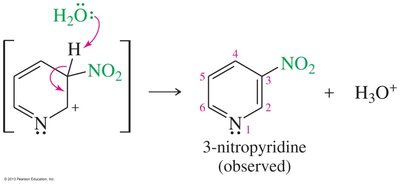
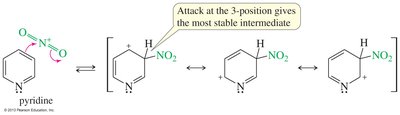
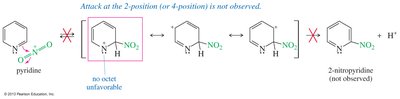
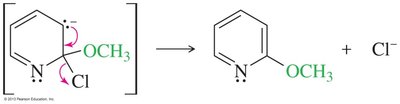
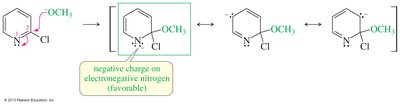
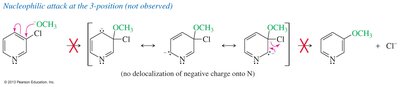
Nucleophilic Substitution of Pyridine
Pyridine is activated toward nucleophilic aromatic substitution, with nucleophiles replacing good leaving groups in the 2- or 4-positions. Attack at the 3-position does not place the negative charge on nitrogen, so substitution is not observed there.
Alkylation and Acylation of Amines
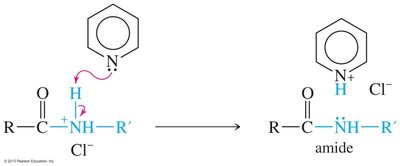
Amines can be alkylated by alkyl halides via SN2 mechanism, but multiple alkylations can lead to complex mixtures. Exhaustive alkylation forms tetraalkylammonium salts. Acylation of primary and secondary amines with acid halides forms amides via nucleophilic acyl substitution.
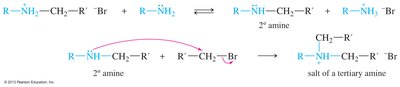
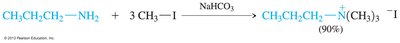
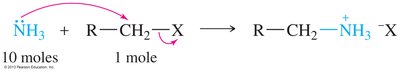
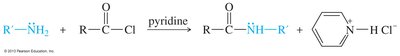
Synthetic Methods for Amines
Acylation–Reduction
Acylation of an amine with an acid chloride forms an amide, which can be reduced by LiAlH4 to yield the corresponding amine. This method is used to synthesize primary, secondary, and tertiary amines.
*Additional info: Acylation–reduction avoids overacylation and allows selective synthesis of amines.*
Formation of Sulfonamides
Primary or secondary amines react with sulfonyl chloride to produce sulfonamides, which are important antibacterial agents (e.g., sulfanilamide).
*Additional info: Sulfanilamide inhibits bacterial growth by mimicking p-aminobenzoic acid, essential for folic acid synthesis.*
Hofmann and Cope Eliminations
Exhaustive methylation of amines forms quaternary ammonium salts, which undergo Hofmann elimination to yield the least substituted alkene. Tertiary amines can be oxidized to amine oxides, which undergo Cope elimination via a syn E2 mechanism.
*Additional info: Hofmann elimination is regioselective for the least substituted alkene; Cope elimination does not require a strong base.*
Diazotization and Arenediazonium Salts
Primary amines react with nitrous acid to form diazonium salts, which are unstable and decompose to carbocations and nitrogen. Arenediazonium salts allow conversion of aromatic amines to various functional groups via Sandmeyer and diazo coupling reactions.
*Additional info: Diazotization is a key method for aromatic substitution and functional group interconversion.*
Reductive Amination
Reductive amination is a general method for synthesizing primary, secondary, and tertiary amines by condensation of amines with ketones or aldehydes, followed by reduction of the imine or iminium intermediate.
*Additional info: Reductive amination is versatile and widely used in organic synthesis.*
Gabriel Synthesis and Other Reductions
Gabriel synthesis is limited to primary amines, using phthalimide as a nucleophile. Primary amines can also be synthesized by reduction of azides, nitriles, and nitro compounds.
*Additional info: These methods are important for preparing primary amines from alkyl halides or alcohols.* -------------------------------------
