 Back
BackAmines: Structure, Properties, Synthesis, and Reactions
Study Guide - Smart Notes

Chapter 19: Amines
Introduction to Amines
Amines are organic derivatives of ammonia (NH3) in which one or more hydrogen atoms are replaced by alkyl or aryl groups. They are a fundamental class of organic compounds with significant roles in biological systems, pharmaceuticals, and industrial chemistry.
General Structure: Amines contain a nitrogen atom bonded to one or more carbon-containing groups (alkyl or aryl).
Classification: Based on the number of organic substituents attached to nitrogen:
Primary (1°) amine: One alkyl/aryl group (RNH2)
Secondary (2°) amine: Two alkyl/aryl groups (R2NH)
Tertiary (3°) amine: Three alkyl/aryl groups (R3N)
Quaternary ammonium salt: Four alkyl/aryl groups (R4N+ X-)
Hybridization: Nitrogen in amines is sp3 hybridized, with a lone pair of electrons.
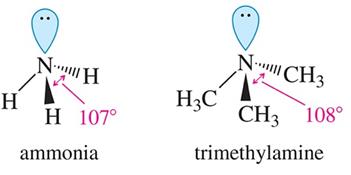
Bond Angles: The bond angle in ammonia is about 107°, and in trimethylamine, it is about 108°, both slightly less than the ideal tetrahedral angle due to the presence of the lone pair.
Physical Properties of Amines
Amines exhibit distinct physical properties due to their structure and the presence of a lone pair on nitrogen.
Polarity: Amines are polar molecules. The polarity depends on the degree of substitution and molecular size.
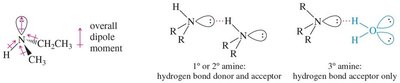
Hydrogen Bonding: Primary and secondary amines can form hydrogen bonds (N–H), but these are weaker than O–H bonds in alcohols. Tertiary amines cannot donate hydrogen bonds but can accept them.
Boiling Points: For compounds of similar molecular weight, the trend is: Alcohols > 1°/2° Amines > Ethers > 3° Amines.
Solubility: Primary and secondary amines are generally soluble in water due to hydrogen bonding. Tertiary amines are less soluble, and amines with long alkyl chains are less soluble due to increased hydrophobic character.
Basicity of Amines
Amines act as bases because the lone pair on nitrogen can accept a proton (H+). The basicity depends on the availability of this lone pair, which is influenced by electronic and steric effects.
Alkylamines: Most basic (pKa ≈ 10–11) due to electron-donating alkyl groups.
Ammonia: Reference point (pKa ≈ 9.25).
Arylamines: Least basic (pKa ≈ 3–5) due to delocalization of the lone pair into the aromatic ring.
Order of Basicity: Alkylamines > Ammonia > Arylamines.
Nomenclature of Amines
Common Names
Common names are formed by naming the alkyl groups attached to nitrogen, followed by the suffix "-amine." Prefixes di-, tri-, and tetra- are used for multiple identical groups.
Examples: ethylamine, diisopentylamine, tetrabutylammonium chloride, cyclohexyldimethylamine, benzylamine, diphenylamine.
IUPAC Nomenclature
Identify the longest carbon chain containing the -NH2 group.
Replace the "-e" ending of the parent alkane with "-amine."
Number the chain to give the amine the lowest possible number.
For rings: use "cycloalkanamine." For aromatic amines: use "aniline."
For secondary and tertiary amines, use "N-" to indicate substituents on nitrogen.
When an amine is a substituent, use the "amino-" prefix.
Spectroscopy of Amines
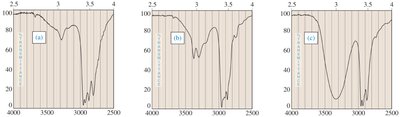
Infrared (IR) Spectroscopy
Primary and secondary amines show absorptions between 3200–3500 cm-1. These are often broad and may be confused with O–H stretches.
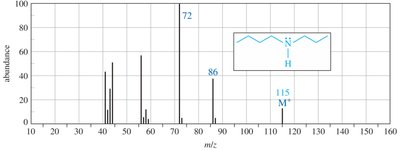
Mass Spectrometry
Amines often show molecular ions with odd mass numbers due to the presence of nitrogen. Fragment ions typically have even mass numbers.
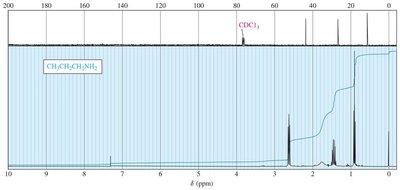
NMR Spectroscopy
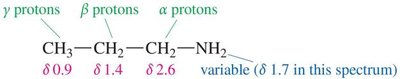
Protons on α-carbons (adjacent to nitrogen) appear in the range δ 1–4. Amine protons are exchangeable and may disappear or shift in D2O.
Common Uses of Amines
Amines are widely used in pharmaceuticals, agriculture, and industry.
Pharmaceuticals: Antihistamines, antidepressants, local anesthetics (e.g., lidocaine), analgesics, vasodilators, antibacterial agents (sulfa drugs).
Agricultural Chemicals: Herbicides, pesticides, fungicides, and insecticides.
Dyes and Pigments: Methylene blue, Congo red (azo dye).
Synthesis of Amines
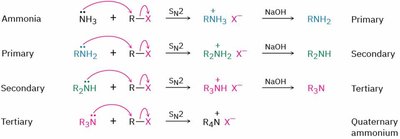
1. Alkylation with Alkyl Halides (SN2 Mechanism)
Amines are good nucleophiles and react with alkyl halides via SN2 mechanisms. Over-alkylation can occur, leading to mixtures of products.
2. Gabriel Synthesis
Gabriel synthesis is used to prepare primary amines from alkyl halides using phthalimide as a nucleophile.
3. Reduction of Nitro and Nitrile Compounds
Nitro and nitrile compounds can be reduced to amines using hydrogenation (H2/Pd, H2/Ni), metal hydrides (LiAlH4), or metals (Fe, Sn) in acid.
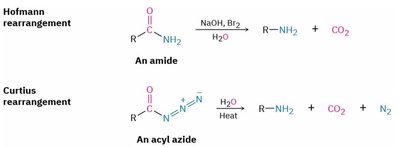
4. Rearrangements: Hofmann and Curtius
Both rearrangements convert amides or acyl azides to primary amines, with loss of CO2 (and N2 in Curtius).
Reactions of Amines
1. Reactions with Acids
Amines react with acids to form ammonium salts. The basicity is quantified by Kb and pKb values.
Factors Influencing Basicity: Electron-donating groups increase basicity; electron-withdrawing groups decrease it. Resonance and hybridization also affect basicity (sp3 > sp2).
2. Acylation
Primary and secondary amines react with acid chlorides to form amides (nucleophilic acyl substitution).
3. Sulfonylation
Primary or secondary amines react with sulfonyl chlorides to form sulfonamides, important in the synthesis of sulfa drugs.
4. Imine (Schiff Base) Formation
Primary amines react with aldehydes or ketones to form imines (Schiff bases) via nucleophilic addition and elimination of water.
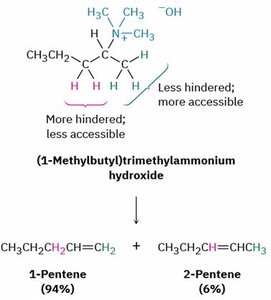
5. Hofmann Elimination
Quaternary ammonium salts undergo elimination (E2) to form alkenes. The major product is the less substituted alkene (Hofmann product), due to steric effects.
Summary Table: Synthesis and Reactions of Amines
Synthesis Method | Key Reagents/Conditions | Product |
|---|---|---|
Alkylation with Alkyl Halides | Alkyl halide, NH3 or amine, NaOH | 1°, 2°, 3° amines, quaternary ammonium salts |
Gabriel Synthesis | Phthalimide, alkyl halide, base | Primary amine |
Reduction of Nitro/Nitrile | H2/Pd, LiAlH4, Fe/H+ | Primary amine |
Hofmann/Curtius Rearrangement | Amide/acyl azide, NaOH/Br2 or heat | Primary amine |
Reaction Type | Reagents/Conditions | Product |
|---|---|---|
Acid-Base Reaction | Acid (e.g., HCl) | Ammonium salt |
Acylation | Acid chloride, base | Amide |
Sulfonylation | Sulfonyl chloride, base | Sulfonamide |
Imine Formation | Aldehyde/ketone, acid catalyst | Imine (Schiff base) |
Hofmann Elimination | Excess CH3I, Ag2O, heat | Alkene (Hofmann product) |
Practice Problems
Give IUPAC names for various amines.
Interpret IR, mass, and NMR spectra for amines.
Predict products and mechanisms for amine synthesis and reactions.
