 Back
BackAmino Acids, Peptides, and Proteins: Structure, Properties, and Synthesis
Study Guide - Smart Notes

Chapter 24: Amino Acids, Peptides, and Proteins
Introduction to Amino Acids
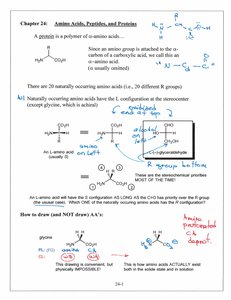
Amino acids are the building blocks of proteins, each containing an amino group, a carboxylic acid group, a hydrogen atom, and a unique side chain (R group) attached to a central (α) carbon. Proteins are polymers of α-amino acids, and their sequence and structure determine biological function.
General Structure:
Chirality: Most naturally occurring amino acids are L-enantiomers (except glycine, which is achiral).
Number of Amino Acids: There are 20 standard amino acids found in proteins.
Classification of Amino Acids
Amino acids are classified based on the properties of their side chains (R groups):
Nonpolar (N): Alkyl or aryl side chains (e.g., Gly, Ala, Val, Leu, Ile, Phe, Pro)
Polar Neutral (PN): Side chains with alcohol, amide, or thiol groups (e.g., Ser, Thr, Asn, Gln, Tyr, Met, Cys)
Acidic (A): Side chains with carboxylic acid groups (e.g., Asp, Glu)
Basic (B): Side chains with amine groups (e.g., Lys, Arg, His)

Acid-Base Chemistry of Amino Acids
Amino acids exist as zwitterions (internal salts) at physiological pH, with both a positively charged ammonium group and a negatively charged carboxylate group. The acid-base properties are characterized by their pKa values:
pKa (COOH): Typically around 2
pKa (NH3+): Typically around 9-10
Isoelectric Point (pI): The pH at which the amino acid has no net charge, calculated as for amino acids without ionizable side chains.

Separation of Amino Acids Using Acid-Base Chemistry
Amino acids can be separated based on their charge at different pH values using techniques such as electrophoresis. The migration depends on the net charge, which is determined by the pH relative to the pI of the amino acid.
Electrophoresis: At pH < pI, amino acids are positively charged and migrate toward the cathode; at pH > pI, they are negatively charged and migrate toward the anode.
Titration Curves: Show the stepwise ionization of amino and carboxyl groups.

Laboratory Synthesis of Amino Acids
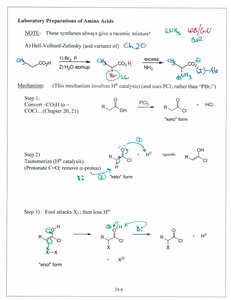
Hell-Volhard-Zelinsky (HVZ) Synthesis
The HVZ reaction is used to synthesize α-amino acids from carboxylic acids via halogenation, followed by nucleophilic substitution with ammonia. This method produces racemic mixtures.
Step 1: Halogenation of carboxylic acid to form α-halo acid.
Step 2: Nucleophilic substitution with ammonia to yield the amino acid.
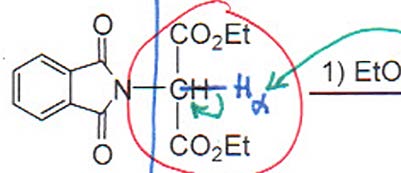
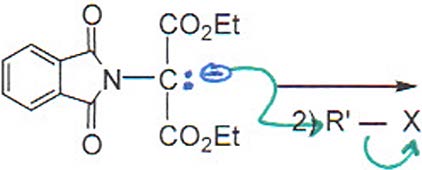
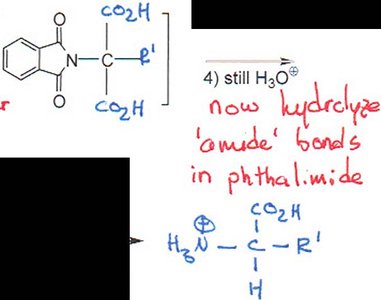
Gabriel Synthesis
The Gabriel synthesis allows for the preparation of primary amines (including amino acids) using phthalimide as a protected amine source. The process involves alkylation, hydrolysis, and decarboxylation steps.
Step 1: Alkylation of phthalimide with an α-halo ester.
Step 2: Hydrolysis to release the free amine and carboxylic acid.
Step 3: Decarboxylation to yield the amino acid.
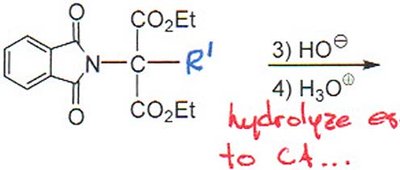
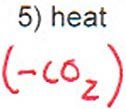
Summary Table: Amino Acid Properties
Name | Symbol | Side Chain Type | pI | Essential? |
|---|---|---|---|---|
Glycine | Gly (G) | Nonpolar | 6.0 | No |
Alanine | Ala (A) | Nonpolar | 6.0 | No |
Serine | Ser (S) | Polar Neutral | 5.7 | No |
Aspartic Acid | Asp (D) | Acidic | 2.8 | No |
Lysine | Lys (K) | Basic | 9.8 | Yes |
Key Equations
pKa definition:
Isoelectric point (pI):
Summary
Amino acids are classified by their side chains and exist as zwitterions at physiological pH.
They can be synthesized in the laboratory by several methods, including the HVZ and Gabriel syntheses.
Their acid-base properties are crucial for separation and identification techniques such as electrophoresis.
