 Back
BackAromatic Compounds: Structure, Nomenclature, Stability, and Reactions
Study Guide - Smart Notes

Aromatic Compounds
Introduction to Aromatic Compounds
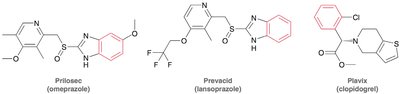
Aromatic compounds, also known as arenes, are a fundamental class of organic molecules characterized by the presence of one or more benzene rings. These compounds are widely found in nature and are important in pharmaceuticals, materials, and chemical synthesis. - Benzene is the prototypical aromatic compound, consisting of a six-membered ring with alternating double bonds. - Benzene derivatives include compounds such as toluene and xylene, which are commonly used in industry. - Aromatic rings are a common structural feature in many drugs, such as Prilosec (omeprazole), Prevacid (lansoprazole), and Plavix (clopidogrel). 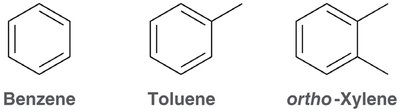
Nomenclature of Benzene Derivatives
The nomenclature of benzene derivatives is based on the position and identity of substituents attached to the benzene ring. - Dimethyl benzene derivatives are commonly known as xylene. - The terms ortho (1,2-), meta (1,3-), and para (1,4-) are used to specify the relative positions of substituents on the ring. 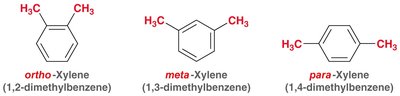 - Substituents are named and numbered to give the lowest possible locants, and listed alphabetically (ignoring prefixes except 'iso'). - Examples include ortho-nitroanisole (2-nitroanisole), meta-bromotoluene (3-bromotoluene), and para-chlorobenzaldehyde (4-chlorobenzaldehyde).
- Substituents are named and numbered to give the lowest possible locants, and listed alphabetically (ignoring prefixes except 'iso'). - Examples include ortho-nitroanisole (2-nitroanisole), meta-bromotoluene (3-bromotoluene), and para-chlorobenzaldehyde (4-chlorobenzaldehyde). 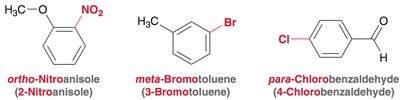 - When a substituent is part of the parent name (e.g., phenol), it is assigned locant number 1.
- When a substituent is part of the parent name (e.g., phenol), it is assigned locant number 1. 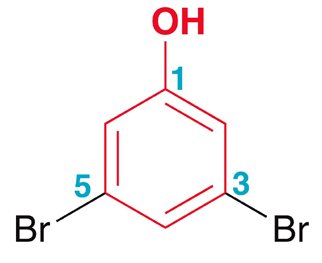
Resonance and Representation of Benzene
Benzene is best represented as a resonance hybrid, with two equivalent structures. Sometimes, a circle is drawn inside the hexagon to indicate delocalized electrons. - Resonance contributors do not exist in equilibrium; they are theoretical representations. 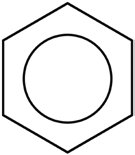
Structure and Stability of Benzene
Stability of Aromatic Rings
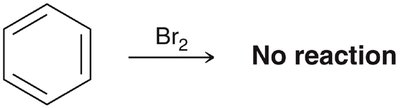
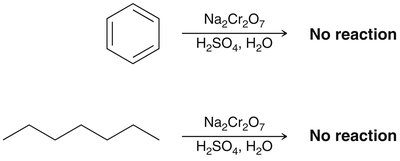
Aromatic rings are remarkably stable compared to alkenes, which readily undergo addition reactions. Benzene does not react with bromine under conditions where alkenes do. 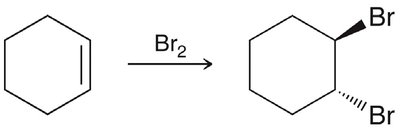
Heats of Hydrogenation
The stability of benzene can be quantified using heats of hydrogenation. Benzene's observed heat of hydrogenation is much less than expected, indicating extra stability due to aromaticity. 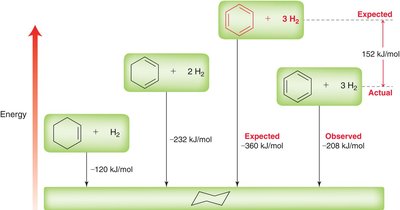
Molecular Orbital (MO) Theory and Aromaticity
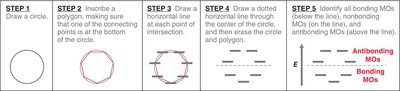
MO theory explains the stability of benzene by the delocalization of six π electrons in three bonding molecular orbitals. - The six atomic p-orbitals of benzene overlap to form six molecular orbitals (MOs). - The locations of nodes in the MOs determine their shapes. 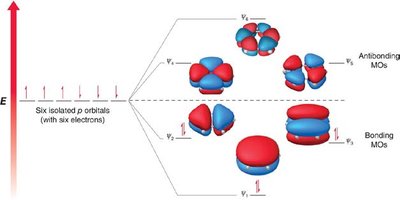
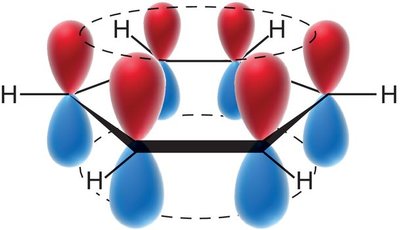
Criteria for Aromaticity and Hückel's Rule
Not all fully conjugated rings are aromatic. Aromatic compounds must meet two criteria: 1. A fully conjugated ring with overlapping p-orbitals 2. Hückel’s rule: an odd number of electron pairs or total π electrons, where - Compounds that do not meet Hückel’s rule are not aromatic. 
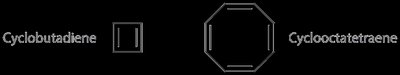
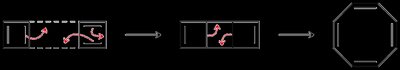
Antiaromatic and Nonaromatic Compounds
- Antiaromatic compounds have a fully conjugated ring and π electrons (even number of electron pairs). - Nonaromatic compounds fail to meet the criterion of a fully conjugated ring. - Cyclooctatetraene can adopt a tub-shaped conformation to avoid antiaromaticity, becoming nonaromatic. 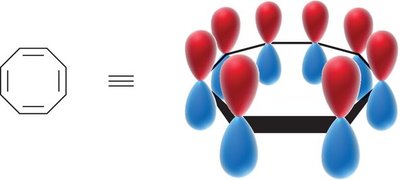
Frost Circles and MO Energy Prediction
Frost circles are a graphical tool to predict the relative energies of molecular orbitals in cyclic conjugated systems. - The number of bonding orbitals is always odd for aromatic compounds. 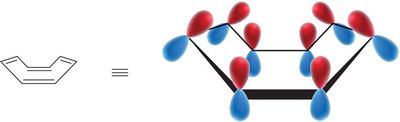
Other Aromatic Compounds
Annulenes
Annulenes are fully conjugated rings that can be aromatic, antiaromatic, or nonaromatic depending on their electron count and structure. ![Structures of [6]Annulene, [10]Annulene, and [14]Annulene](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/93dabf8c_image_29.png)
Aromatic Ions: Carbanions and Carbocations
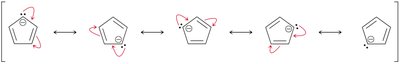
Aromatic rings can contain carbanions or carbocations. - The cyclopentadienyl anion is aromatic due to the delocalization of a lone pair over five carbon atoms, resulting in six π electrons. - The acidity of cyclopentadiene is attributed to the aromatic stability of its conjugate base. 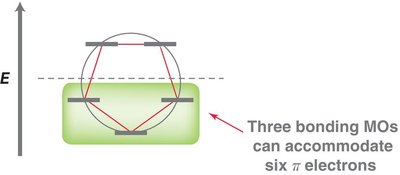
 - The tropylium cation (seven-membered ring with six π electrons) is an aromatic cation.
- The tropylium cation (seven-membered ring with six π electrons) is an aromatic cation. 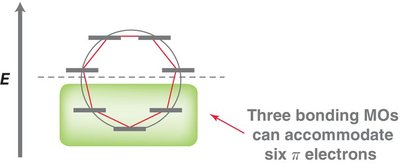
Aromatic Heterocycles
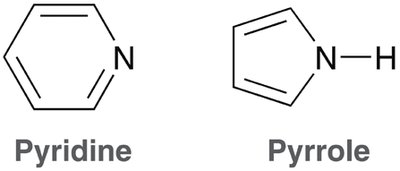
Heteroatoms (atoms other than C or H) can be part of aromatic rings. - If the heteroatom’s lone pair is necessary for aromaticity, it is included in the Hückel count of π electrons. - Examples include pyridine and pyrrole. 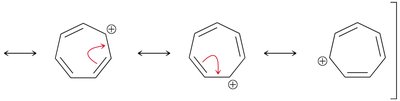
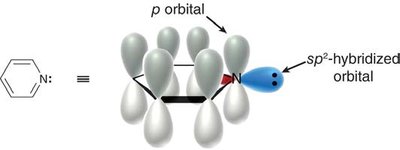 - The basicity of the lone pair depends on whether it is involved in aromaticity.
- The basicity of the lone pair depends on whether it is involved in aromaticity.  - Electrostatic potential maps show differences in electron density.
- Electrostatic potential maps show differences in electron density. 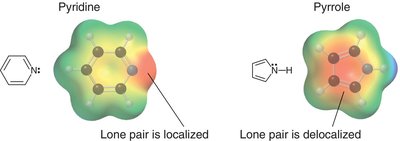
Polycyclic Aromatic Compounds
Many polycyclic compounds, such as naphthalene, anthracene, and phenanthrene, are aromatic. 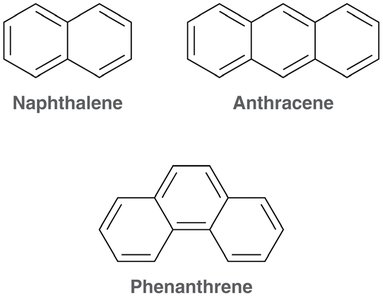
Reactions at the Benzylic Position
Benzylic Position and Oxidation
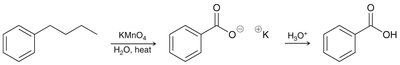
The benzylic position is the carbon directly attached to a benzene ring. - Aromatic rings and alkyl groups are not easily oxidized, but benzylic positions are readily oxidized by chromic acid or permanganate if at least one proton is present. 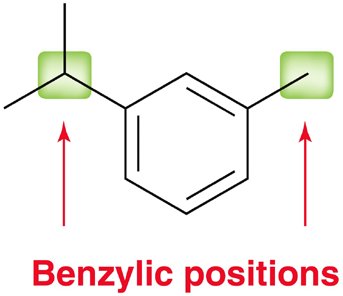
Reduction of Benzene
Hydrogenation and Birch Reduction
Benzene can be reduced to cyclohexane under forceful conditions (Ni, 100 atm, 150°C), but alkenes can be selectively hydrogenated in the presence of a benzene ring.  - Benzene can also be reduced via the Birch reduction, which does not fully saturate the ring and results in regioselectivity depending on the presence of electron-donating or electron-withdrawing groups.
- Benzene can also be reduced via the Birch reduction, which does not fully saturate the ring and results in regioselectivity depending on the presence of electron-donating or electron-withdrawing groups.  Additional info: The notes above expand on the original content by providing definitions, examples, and academic context for aromaticity, resonance, nomenclature, and reactions. All equations are formatted in LaTeX as required.
Additional info: The notes above expand on the original content by providing definitions, examples, and academic context for aromaticity, resonance, nomenclature, and reactions. All equations are formatted in LaTeX as required.
