 Back
BackAtomic Structure and Bonding: Foundations for Organic Chemistry
Study Guide - Smart Notes

Atomic Structure
Composition of the Atom
The atom is the fundamental unit of matter, consisting of a dense, positively charged nucleus surrounded by negatively charged electrons. The nucleus contains protons (positively charged) and neutrons (electrically neutral), while electrons occupy regions of space at various energy levels around the nucleus.
Protons: Define the atomic number and identity of the element.
Neutrons: Contribute to the mass number but do not affect the chemical properties.
Electrons: Occupy orbitals and are responsible for chemical bonding and reactivity.
Properties of Subatomic Particles
Subatomic particles differ in mass and charge, which are summarized in the table below:
Particle | Symbol | Charge | Mass (g) | Relative Mass |
|---|---|---|---|---|
Electron | e- | -1 | 9.109 × 10-28 | 1/1836 |
Proton | p | +1 | 1.673 × 10-24 | 1 |
Neutron | n | 0 | 1.675 × 10-24 | 1 |
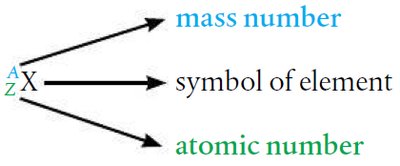
Atomic Number and Mass Number
The atomic number (Z) is the number of protons in the nucleus and determines the element. The mass number (A) is the sum of protons and neutrons. The number of neutrons is calculated as:
Isotopes
Isotopes are atoms of the same element (same atomic number) with different numbers of neutrons, resulting in different mass numbers. For example, carbon has three naturally occurring isotopes:
12C: 6 protons, 6 neutrons (most abundant)
13C: 6 protons, 7 neutrons
14C: 6 protons, 8 neutrons (radioactive, used in carbon dating)
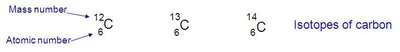
Relative Atomic and Molecular Mass
The relative atomic mass (Ar) of an element is the average mass of its atoms compared to 1/12th the mass of a carbon-12 atom. The relative molecular mass (Mr) is the average mass of a molecule compared to 1/12th the mass of a carbon-12 atom.
Atomic Orbitals and Quantum Numbers
Quantum Mechanical Model
Electrons in atoms are described by quantum mechanics, where their behavior is represented by wave functions (ψ). The square of the wave function, , gives the probability of finding an electron at a particular point, defining an atomic orbital.
Quantum Numbers
Atomic orbitals are described by four quantum numbers:
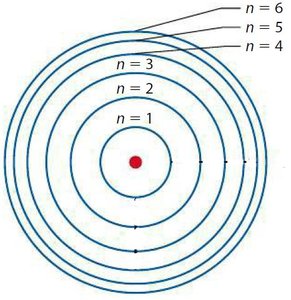
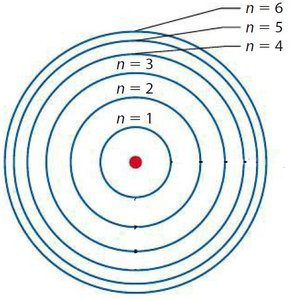
Principal quantum number (n): Indicates the energy level or shell (n = 1, 2, 3, ...).
Orbital angular momentum quantum number (l): Defines the subshell and shape of the orbital (l = 0, 1, ..., n-1; s, p, d, f).
Magnetic quantum number (ml): Specifies the orientation of the orbital in space (ml = -l to +l).
Spin quantum number (ms): Describes the spin of the electron (+1/2 or -1/2).
Shapes of Atomic Orbitals
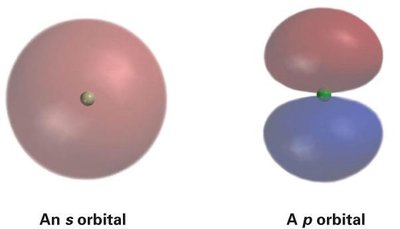
The most important orbitals in organic chemistry are s and p orbitals:
s orbitals: Spherical in shape, centered on the nucleus.
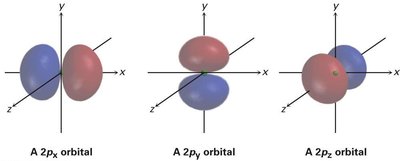
p orbitals: Dumbbell-shaped, with three orientations (px, py, pz), each with a node at the nucleus.
Electron Shells and Subshells
Orbitals are grouped into shells of increasing energy and size:
First shell: 1s (2 electrons)
Second shell: 2s, 2p (8 electrons)
Third shell: 3s, 3p, 3d (18 electrons)

Electron Configuration
Ground State Electron Configuration
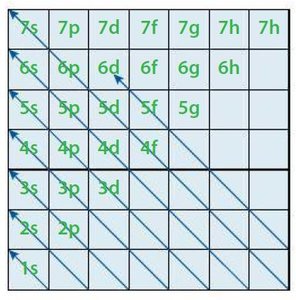
The ground state electron configuration is the lowest-energy arrangement of electrons in an atom. Three rules govern electron configuration:
Aufbau Principle: Electrons fill orbitals of lowest energy first (1s → 2s → 2p → 3s → 3p → 4s → 3d).
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers; each orbital holds a maximum of two electrons with opposite spins.
Hund's Rule: Electrons occupy degenerate orbitals singly before pairing, maximizing the number of unpaired electrons with parallel spins.
Chemical Bonding
Why Atoms Bond
Atoms bond to achieve greater stability, resulting in compounds that are lower in energy than the separate atoms. The formation of molecules is central to chemistry and underpins the design of new materials and drugs.
The Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve eight electrons in their outer shell, similar to the noble gases. This rule is most applicable to main group elements.
Noble gas configuration: Especially stable due to a filled valence shell (e.g., Ne: [2s2 2p6]).
Ionic Bonding
Ionic bonds form when electrons are transferred from one atom to another, resulting in oppositely charged ions held together by electrostatic attraction.
Alkali metals (Group 1A): Lose one electron to form cations (e.g., Na+).
Halogens (Group 7A): Gain one electron to form anions (e.g., Cl-).
Example: Sodium chloride (NaCl) forms from Na+ and Cl- ions.
Covalent Bonding
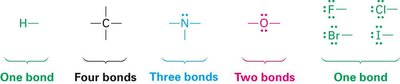
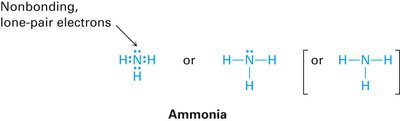
Covalent bonds involve the sharing of electron pairs between atoms to achieve noble gas configuration. Lewis structures (electron-dot structures) are used to represent covalent bonds and lone pairs.
Bonding pairs: Shared electrons between atoms.
Lone pairs: Valence electrons not involved in bonding; can act as nucleophiles.

Limitations of the Octet Rule
The octet rule is not universal and has several exceptions:
Applies mainly to first-row and p-block elements.
Fails for molecules like PCl5, NO, and BF3.
Does not predict bond strength or always the correct number of bonds.
Summary
Atoms consist of a nucleus (protons and neutrons) surrounded by electrons in orbitals.
Electron configuration is determined by quantum numbers and filling rules (Aufbau, Pauli, Hund).
The octet rule explains the tendency of atoms to achieve noble gas configurations through bonding.
Ionic bonds result from electron transfer; covalent bonds from electron sharing.
