 Back
BackBases, Aldehydes, Ketones, and Ethers: Structure, Reactivity, and Nomenclature
Study Guide - Smart Notes

Bases and Basicity in Organic Chemistry
Definition and Measurement of Basicity
Basicity in organic chemistry refers to the ability of a compound to accept a proton (H+) by donating a pair of electrons. The strength of a base is commonly measured using the pKa of its conjugate acid, rather than the pKb scale, which has largely been replaced in modern practice.
pKa: The negative logarithm of the acid dissociation constant (Ka), used to express acid strength. For bases, the pKa of the conjugate acid is used.
Strong bases have a large pKa (conjugate acid is weak), while weak bases have a small pKa (conjugate acid is strong).
Basic drugs are often administered as water-soluble salts, such as hydrochlorides.
Equation for base ionization in water:
Relationship between base strength and pKa:
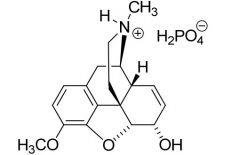
Electronic and Structural Features Influencing Basicity
The basicity of organic compounds is influenced by electronic effects such as inductive and resonance (delocalization) effects. Features that stabilize the conjugate base (such as electron delocalization) decrease basicity, while features that localize electrons increase basicity.
Inductive effects: Electron-withdrawing groups stabilize the conjugate base, making the compound less basic.
Resonance effects: Delocalization of electrons in the conjugate base reduces basicity.
Key Point: Compounds with delocalized electrons are generally weaker bases than those with localized electrons.
Common Basic Functional Groups
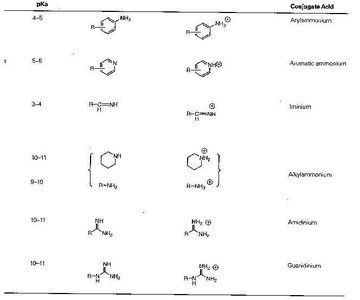
Organic bases include amines, imines, amidines, and guanidines, each with characteristic pKa values and conjugate acid forms.
pKa | Structure | Conjugate Acid |
|---|---|---|
4–5 | Arylamine | Arylammonium |
5–6 | Aromatic amine | Aromatic ammonium |
3–4 | Imine | Imine conjugate acid |
10–11 | Alkylamine | Alkylammonium |
8–10 | Amidine | Amidinium |
10–11 | Guanidine | Guanidinium |
Chemistry of Aldehydes and Ketones
Introduction and Biological Importance
Aldehydes (RCHO) and ketones (R2CO) are widespread in nature and industry. They are required by living organisms and serve as key intermediates in metabolism and as industrial solvents.
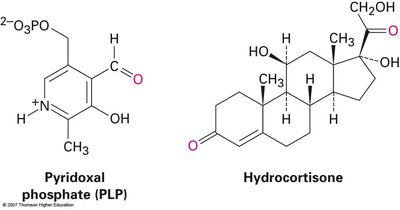
Pyridoxal phosphate (PLP): A coenzyme derived from vitamin B6, essential in amino acid metabolism.
Hydrocortisone: A steroid hormone secreted by the adrenal glands.
Industrial Importance
Formaldehyde (H2C=O): Used in insulation materials.
Acetone ((CH3)2C=O): Used as an industrial solvent.
Nomenclature of Aldehydes and Ketones
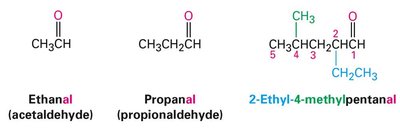
Aldehydes are named by replacing the terminal –e of the parent alkane with –al, and the parent chain must include the –CHO group, which is always numbered as C1. Ketones are named by replacing the terminal –e with –one, and the parent chain is the longest chain containing the carbonyl group, with numbering starting from the end nearest the carbonyl carbon.
Aldehyde example: Ethanal (acetaldehyde), Propanal (propionaldehyde)
Ketone example: Hexan-3-one, Hex-4-en-2-one, Hexane-2,4-dione
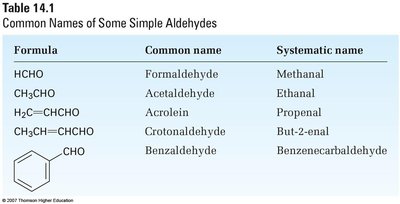
Formula | Common name | Systematic name |
|---|---|---|
HCHO | Formaldehyde | Methanal |
CH3CHO | Acetaldehyde | Ethanal |
H2C=CHCHO | Acrolein | Propenal |
CH3CH=CHCHO | Crotonaldehyde | But-2-enal |
| Benzaldehyde | Benzenecarbaldehyde |
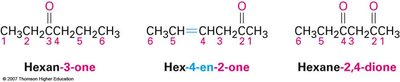
Preparation of Aldehydes and Ketones
Aldehydes can be prepared by oxidation of primary alcohols (using PCC) or by reduction of carboxylic esters (using DIBALH).
Ketones are typically prepared by oxidation of secondary alcohols using chromium-based reagents.
The Carbonyl Group: Structure and Reactivity
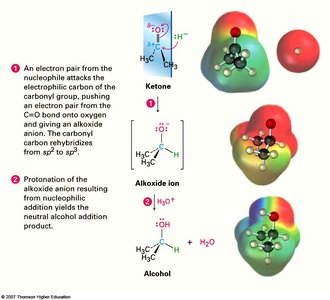
The carbonyl group (C=O) is central to the chemistry of aldehydes and ketones. The difference in electronegativity between carbon and oxygen creates a dipole, making the carbonyl carbon electrophilic and susceptible to nucleophilic attack.
The C=O bond consists of one sigma (σ) and one pi (π) bond.
Aldehydes are generally more reactive than ketones due to less stabilization of the partial positive charge on the carbonyl carbon.

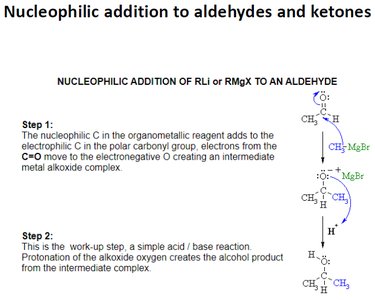
Nucleophilic Addition Reactions of Aldehydes and Ketones
Nucleophilic addition is the characteristic reaction of aldehydes and ketones. A nucleophile attacks the electrophilic carbonyl carbon, forming a tetrahedral alkoxide intermediate, which is then protonated to yield an alcohol.
Aldehydes are more reactive than ketones due to less steric hindrance and electronic effects.
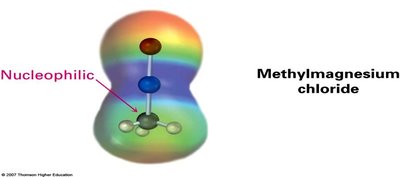
Grignard reagents (RMgX) are important carbon nucleophiles that add to the carbonyl group to form alcohols after protonation.
Chemistry of Ethers
Structure and Properties of Ethers
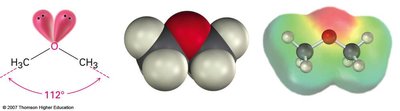
Ethers are compounds in which an oxygen atom is bonded to two alkyl or aryl groups (R–O–R'). Unlike alcohols, ethers cannot form hydrogen bonds with each other, resulting in lower boiling points. Ethers are generally unreactive and are widely used as solvents.
The bond angle in simple ethers (e.g., dimethyl ether) is approximately 112°, similar to water.
Ethers have a nearly tetrahedral geometry around the oxygen atom.
Nomenclature of Ethers
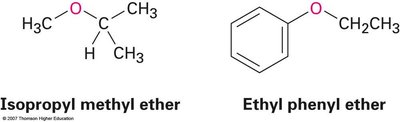
Simple ethers are named by identifying the two organic substituents and adding the word "ether." For more complex ethers, the alkoxy group is named as a substituent.
Examples: Isopropyl methyl ether, Ethyl phenyl ether
Preparation of Ethers: Williamson Ether Synthesis
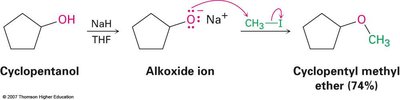
The Williamson ether synthesis is the most common method for preparing ethers. It involves the reaction of an alkoxide ion with a primary alkyl halide or tosylate in an SN2 reaction. The alkoxide ion is typically generated by reacting an alcohol with a strong base such as sodium hydride (NaH).
Example: Cyclopentanol reacts with NaH to form cyclopentoxide ion, which then reacts with methyl iodide to give cyclopentyl methyl ether.
Applications of Ethers: Polyethylene Glycol (PEG)
Polyethylene glycol (PEG) is an ether-based polymer with a wide range of applications, including use in skin creams, toothpaste, laxatives, and pharmaceutical formulations (tablets, capsules).
PEG is valued for its solubility, low toxicity, and chemical inertness.
